1 Neuronal Distribution Across the Cerebral Cortex of the Marmoset
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Neocortex and Allocortex Respond Differentially to Cellular Stress in Vitro and Aging in Vivo
Neocortex and Allocortex Respond Differentially to Cellular Stress In Vitro and Aging In Vivo Jessica M. Posimo, Amanda M. Titler, Hailey J. H. Choi, Ajay S. Unnithan, Rehana K. Leak* Division of Pharmaceutical Sciences, Mylan School of Pharmacy, Duquesne University, Pittsburgh, Pennsylvania, United States of America Abstract In Parkinson’s and Alzheimer’s diseases, the allocortex accumulates aggregated proteins such as synuclein and tau well before neocortex. We present a new high-throughput model of this topographic difference by microdissecting neocortex and allocortex from the postnatal rat and treating them in parallel fashion with toxins. Allocortical cultures were more vulnerable to low concentrations of the proteasome inhibitors MG132 and PSI but not the oxidative poison H2O2. The proteasome appeared to be more impaired in allocortex because MG132 raised ubiquitin-conjugated proteins and lowered proteasome activity in allocortex more than neocortex. Allocortex cultures were more vulnerable to MG132 despite greater MG132-induced rises in heat shock protein 70, heme oxygenase 1, and catalase. Proteasome subunits PA700 and PA28 were also higher in allocortex cultures, suggesting compensatory adaptations to greater proteasome impairment. Glutathione and ceruloplasmin were not robustly MG132-responsive and were basally higher in neocortical cultures. Notably, neocortex cultures became as vulnerable to MG132 as allocortex when glutathione synthesis or autophagic defenses were inhibited. Conversely, the glutathione precursor N-acetyl cysteine rendered allocortex resilient to MG132. Glutathione and ceruloplasmin levels were then examined in vivo as a function of age because aging is a natural model of proteasome inhibition and oxidative stress. Allocortical glutathione levels rose linearly with age but were similar to neocortex in whole tissue lysates. -

The Structural Model: a Theory Linking Connections, Plasticity, Pathology, Development and Evolution of the Cerebral Cortex
Brain Structure and Function https://doi.org/10.1007/s00429-019-01841-9 REVIEW The Structural Model: a theory linking connections, plasticity, pathology, development and evolution of the cerebral cortex Miguel Ángel García‑Cabezas1 · Basilis Zikopoulos2,3 · Helen Barbas1,3 Received: 11 October 2018 / Accepted: 29 January 2019 © Springer-Verlag GmbH Germany, part of Springer Nature 2019 Abstract The classical theory of cortical systematic variation has been independently described in reptiles, monotremes, marsupials and placental mammals, including primates, suggesting a common bauplan in the evolution of the cortex. The Structural Model is based on the systematic variation of the cortex and is a platform for advancing testable hypotheses about cortical organization and function across species, including humans. The Structural Model captures the overall laminar structure of areas by dividing the cortical architectonic continuum into discrete categories (cortical types), which can be used to test hypotheses about cortical organization. By type, the phylogenetically ancient limbic cortices—which form a ring at the base of the cerebral hemisphere—are agranular if they lack layer IV, or dysgranular if they have an incipient granular layer IV. Beyond the dysgranular areas, eulaminate type cortices have six layers. The number and laminar elaboration of eulaminate areas differ depending on species or cortical system within a species. The construct of cortical type retains the topology of the systematic variation of the cortex and forms the basis for a predictive Structural Model, which has successfully linked cortical variation to the laminar pattern and strength of cortical connections, the continuum of plasticity and stability of areas, the regularities in the distribution of classical and novel markers, and the preferential vulnerability of limbic areas to neurodegenerative and psychiatric diseases. -

Nomina Histologica Veterinaria, First Edition
NOMINA HISTOLOGICA VETERINARIA Submitted by the International Committee on Veterinary Histological Nomenclature (ICVHN) to the World Association of Veterinary Anatomists Published on the website of the World Association of Veterinary Anatomists www.wava-amav.org 2017 CONTENTS Introduction i Principles of term construction in N.H.V. iii Cytologia – Cytology 1 Textus epithelialis – Epithelial tissue 10 Textus connectivus – Connective tissue 13 Sanguis et Lympha – Blood and Lymph 17 Textus muscularis – Muscle tissue 19 Textus nervosus – Nerve tissue 20 Splanchnologia – Viscera 23 Systema digestorium – Digestive system 24 Systema respiratorium – Respiratory system 32 Systema urinarium – Urinary system 35 Organa genitalia masculina – Male genital system 38 Organa genitalia feminina – Female genital system 42 Systema endocrinum – Endocrine system 45 Systema cardiovasculare et lymphaticum [Angiologia] – Cardiovascular and lymphatic system 47 Systema nervosum – Nervous system 52 Receptores sensorii et Organa sensuum – Sensory receptors and Sense organs 58 Integumentum – Integument 64 INTRODUCTION The preparations leading to the publication of the present first edition of the Nomina Histologica Veterinaria has a long history spanning more than 50 years. Under the auspices of the World Association of Veterinary Anatomists (W.A.V.A.), the International Committee on Veterinary Anatomical Nomenclature (I.C.V.A.N.) appointed in Giessen, 1965, a Subcommittee on Histology and Embryology which started a working relation with the Subcommittee on Histology of the former International Anatomical Nomenclature Committee. In Mexico City, 1971, this Subcommittee presented a document entitled Nomina Histologica Veterinaria: A Working Draft as a basis for the continued work of the newly-appointed Subcommittee on Histological Nomenclature. This resulted in the editing of the Nomina Histologica Veterinaria: A Working Draft II (Toulouse, 1974), followed by preparations for publication of a Nomina Histologica Veterinaria. -

031609.Phitchcock.Ce
Author(s): Peter Hitchcock, PH.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution–Non-commercial–Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/ We have reviewed this material in accordance with U.S. Copyright Law and have tried to maximize your ability to use, share, and adapt it. The citation key on the following slide provides information about how you may share and adapt this material. Copyright holders of content included in this material should contact [email protected] with any questions, corrections, or clarification regarding the use of content. For more information about how to cite these materials visit http://open.umich.edu/education/about/terms-of-use. Any medical information in this material is intended to inform and educate and is not a tool for self-diagnosis or a replacement for medical evaluation, advice, diagnosis or treatment by a healthcare professional. Please speak to your physician if you have questions about your medical condition. Viewer discretion is advised: Some medical content is graphic and may not be suitable for all viewers. Citation Key for more information see: http://open.umich.edu/wiki/CitationPolicy Use + Share + Adapt { Content the copyright holder, author, or law permits you to use, share and adapt. } Public Domain – Government: Works that are produced by the U.S. Government. (USC 17 § 105) Public Domain – Expired: Works that are no longer protected due to an expired copyright term. Public Domain – Self Dedicated: Works that a copyright holder has dedicated to the public domain. -

The Evolutionary Development of the Brain As It Pertains to Neurosurgery
Open Access Original Article DOI: 10.7759/cureus.6748 The Evolutionary Development of the Brain As It Pertains to Neurosurgery Jaafar Basma 1 , Natalie Guley 2 , L. Madison Michael II 3 , Kenan Arnautovic 3 , Frederick Boop 3 , Jeff Sorenson 3 1. Neurological Surgery, University of Tennessee Health Science Center, Memphis, USA 2. Neurological Surgery, University of Arkansas for Medical Sciences, Little Rock, USA 3. Neurological Surgery, Semmes-Murphey Clinic, Memphis, USA Corresponding author: Jaafar Basma, [email protected] Abstract Background Neuroanatomists have long been fascinated by the complex topographic organization of the cerebrum. We examined historical and modern phylogenetic theories pertaining to microneurosurgical anatomy and intrinsic brain tumor development. Methods Literature and history related to the study of anatomy, evolution, and tumor predilection of the limbic and paralimbic regions were reviewed. We used vertebrate histological cross-sections, photographs from Albert Rhoton Jr.’s dissections, and original drawings to demonstrate the utility of evolutionary temporal causality in understanding anatomy. Results Phylogenetic neuroanatomy progressed from the substantial works of Alcmaeon, Herophilus, Galen, Vesalius, von Baer, Darwin, Felsenstein, Klingler, MacLean, and many others. We identified two major modern evolutionary theories: “triune brain” and topological phylogenetics. While the concept of “triune brain” is speculative and highly debated, it remains the most popular in the current neurosurgical literature. Phylogenetics inspired by mathematical topology utilizes computational, statistical, and embryological data to analyze the temporal transformations leading to three-dimensional topographic anatomy. These transformations have shaped well-defined surgical planes, which can be exploited by the neurosurgeon to access deep cerebral targets. The microsurgical anatomy of the cerebrum and the limbic system is redescribed by incorporating the dimension of temporal causality. -

Cortical and Subcortical Anatomy: Basics and Applied
43rd Congress of the Canadian Neurological Sciences Federation Basic mechanisms of epileptogenesis and principles of electroencephalography Cortical and subcortical anatomy: basics and applied J. A. Kiernan MB, ChB, PhD, DSc Department of Anatomy & Cell Biology, The University of Western Ontario London, Canada LEARNING OBJECTIVES Know and understand: ! Two types of principal cell and five types of interneuron in the cerebral cortex. ! The layers of the cerebral cortex as seen in sections stained to show either nucleic acids or myelin. ! The types of corrtex: allocortex and isocortex. ! Major differences between extreme types of isocortex. As seen in primary motor and primary sensory areas. ! Principal cells in different layers give rise to association, commissural, projection and corticothalamic fibres. ! Cortical neurons are arranged in columns of neurons that share the same function. ! Intracortical circuitry provides for neurons in one column to excite one another and to inhibit neurons in adjacent columns. ! The general plan of neuronal connections within nuclei of the thalamus. ! The location of motor areas of the cerebral cortex and their parallel and hierarchical projections to the brain stem and spinal cord. ! The primary visual area and its connected association areas, which have different functions. ! Somatotopic representation in the primary somatosensory and motor areas. ! Cortical areas concerned with perception and expression of language, and the anatomy of their interconnections. DISCLOSURE FORM This disclosure form must be included as the third page of your Course Notes and the third slide of your presentation. It is the policy of the Canadian Neurological Sciences Federation to insure balance, independence, objectivity and scientific rigor in all of its education programs. -

History, Anatomical Nomenclature, Comparative Anatomy and Functions of the Hippocampal Formation
Bratisl Lek Listy 2006; 107 (4): 103106 103 TOPICAL REVIEW History, anatomical nomenclature, comparative anatomy and functions of the hippocampal formation El Falougy H, Benuska J Institute of Anatomy, Faculty of Medicine, Comenius University, Bratislava, [email protected] Abstract The complex structures in the cerebral hemispheres is included under one term, the limbic system. Our conception of this system and its special functions rises from the comparative neuroanatomical and neurophysiological studies. The components of the limbic system are the hippocampus, gyrus parahippocampalis, gyrus dentatus, gyrus cinguli, corpus amygdaloideum, nuclei anteriores thalami, hypothalamus and gyrus paraterminalis Because of its unique macroscopic and microscopic structure, the hippocampus is a conspicuous part of the limbic system. During phylogenetic development, the hippocampus developed from a simple cortical plate in amphibians into complex three-dimensional convoluted structure in mammals. In the last few decades, structures of the limbic system were extensively studied. Attention was directed to the physi- ological functions and pathological changes of the hippocampus. Experimental studies proved that the hippocampus has a very important role in the process of learning and memory. Another important functions of the hippocampus as a part of the limbic system is its role in regulation of sexual and emotional behaviour. The term hippocampal formation is defined as the complex of six structures: gyrus dentatus, hippocampus proprius, subiculum proprium, presubiculum, parasubiculum and area entorhinalis In this work we attempt to present a brief review of knowledge about the hippocampus from the point of view of history, anatomical nomenclature, comparative anatomy and functions (Tab. 1, Fig. 2, Ref. -
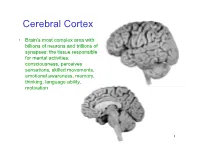
Cerebral Cortex
Cerebral Cortex • Brain’s most complex area with billions of neurons and trillions of synapses: the tissue responsible for mental activities: consciousness, perceives sensations, skilled movements, emotional awareness, memory, thinking, language ability, motivation 1 John Lorber, Science 210:1232 (1980) “There a young student at this university who has an IQ of 126, has gained a first-class honors degree in mathematics, and is socially completely normal. And yet the boy has virtually no brain.” “The student’s physician at the university noticed that the student had slightly larger than normal head… When we did a brain scan on him we saw that instead of the normal 4.5 cm thickness of brain tissue there was just a millimeter or so. His cranium is filled with CSF.” How is this possible? What does it tell us? Do you think this would be OK if it happened to an adult? To a 15 year old? To a 5 year old? To a neonate? 3 Types of Cerebral Cortex • Neocortex – Newest in evolution – About 90% of total – 6 layers, most complex • Paleocortex – Associated with olfactory system, the parahippocampal gyrus, uncus – fewer than 6 layers • Archicortex – Hippocampal formation; limbic system – 3 layers, most primitive • Mesocortex – Cingulate gyrus, insular cortex – Transitional between archicortex and neocortex 5 The perks of having a neocortex • The words used to describe the higher mental capacities of animals with a large neocortex include: – CONSCIOUSNESS – FREE WILL – INTELLIGENCE – INSIGHT • Animals with much simpler brains learn well, so LEARNING should not be among these capacities (Macphail 1982). • A species could have genetically determined mechanisms, acquired through evolutionary selection, for taking advantage of the regular features of the environment, or they could have learned through direct experience. -

Behavioral Neuroanatomy: Large-Scale Networks, Association
M. - MAR S E L M E SU LAM Faced with an anatomical fact proven beyond doubt, any physiological result that stands in contradiction to it loses all its meaning. ... So, first anatomy and then physiology; but if first physiology, then not without ana tomy. —BERNHARD VON GUDDEN(1824-1886), QUOTED BY KORBINIAN BRODMANN, IN LAU RENCE GAREY ‘S TRANSLATION I. INTRODUCTION The human brain displays marked regional variations in architecture, connectivity, neurochemistrv, and physiology. This chapter explores the relevance of these re- gional variations to cognition and behavior. Some topics have been included mostly for the sake of completeness and continuity. Their coverage is brief, either because the available information is limited or because its relevance to behavior and cog- nition is tangential. Other subjects, such as the processing of visual information, are reviewed in extensive detail, both because a lot is known and also because the information helps to articulate general principles relevant to all other domains of behavior. Experiments on laboratory primates will receive considerable emphasis, espe- cially in those areas of cerebral connectivity and physiology where relevant infor- mation is not yet available in the human. Structural homologies across species are always incomplete, and many complex behaviors, particularly those that are of greatest interest to the clinician and cognitive neuroscientist, are either rudimentary or absent in other animals. Nonetheless, the reliance on animal data in this chapter is unlikely to be too misleading since the focus will be on principles rather than specifics and since principles of organization are likely to remain relatively stable across closely related species. -

NIH Public Access Author Manuscript Neuroimage
NIH Public Access Author Manuscript Neuroimage. Author manuscript; available in PMC 2014 January 01. Published in final edited form as: Neuroimage. 2013 January 1; 64C: 32–42. doi:10.1016/j.neuroimage.2012.08.071. Predicting the Location of Human Perirhinal Cortex, Brodmann's area 35, from MRI $watermark-text $watermark-text $watermark-text Jean C. Augustinacka,#, Kristen E. Hubera, Allison A. Stevensa, Michelle Roya, Matthew P. Froschb, André J.W. van der Kouwea, Lawrence L. Walda, Koen Van Leemputa,f, Ann McKeec, Bruce Fischla,d,e, and The Alzheimer's Disease Neuroimaging Initiative* aAthinoula A Martinos Center, Dept. of Radiology, MGH, 149 13th Street, Charlestown MA 02129 USA bC.S. Kubik Laboratory for Neuropathology, Pathology Service, MGH, 55 Fruit St., Boston MA 02115 USA cDepartment of Pathology, Boston University School of Medicine, Bedford Veterans Administration Medical Center, MA 01730 USA dMIT Computer Science and AI Lab, Cambridge MA 02139 USA eMIT Division of Health Sciences and Technology, Cambridge MA 02139 USA fDepartment of Informatics and Mathematical Modeling, Technical University of Denmark, Copenhagen, Denmark Abstract The perirhinal cortex (Brodmann's area 35) is a multimodal area that is important for normal memory function. Specifically, perirhinal cortex is involved in detection of novel objects and manifests neurofibrillary tangles in Alzheimer's disease very early in disease progression. We scanned ex vivo brain hemispheres at standard resolution (1 mm × 1 mm × 1 mm) to construct pial/white matter surfaces in FreeSurfer and scanned again at high resolution (120 μm × 120 μm × 120 μm) to determine cortical architectural boundaries. After labeling perirhinal area 35 in the high resolution images, we mapped the high resolution labels to the surface models to localize area 35 in fourteen cases. -

PDF Hosted at the Radboud Repository of the Radboud University Nijmegen
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/113728 Please be advised that this information was generated on 2021-10-11 and may be subject to change. THE HIPPOCAMPAL FORMATION IN NORMAL AGING AND ALZHEIMER'S DISEASE A MORPHOMETRIC STUDY HARKE DE VRIES THE HXX'X'OCÄJVLE'AX. FOFtlVITVrX CMST I IST JsiORjyrzvx. ÍVGXISÍ<3 ÎVISTD г х^ZH:E:xiynE:R » s XiXSEASE г^. ІУІОЬІ^НОІУЕЕТІІХС; STXXDY THE HIPPOCAMPAL FORMATION IN NORMAL· AGING AND ALZHEIMER'S DISEASE A MORPHOMETRIC STUDY een wetenschappelijke proeve op het gebied van de geneeskunde en tandheeIkunde Proefschrift ter verkrijging van de graad van doctor aan de Katholieke Universiteit te Nijmegen volgens besluit van het college van decanen in het openbaar te verdedigen op woensdag 21 februari 1990 des namiddags te 1.30 uur precies door geboren op 27 augustu199s 0195 7 te Badhocvedorp Drukkerij Leijn te Nijmegen Promotores: Prof. dr. R. Nieuwenhuys Prof. dr. G.J. ZwaniJOcen Co-promotor: Dr. J. Meek ...In the beginner's mind there is no thought "I have attained something"... (Shunryu Suzuki, in: Zen Mind, Beginner's Mind) Dedicated to my mother, In memory of my father. ISBN 90-9003283-5 CONTENTS CHAPTER 1 GENERAL INTRODUCTION 2 1.1 Epidemiology of Alzheimer's disease 3 1.2 Diagnosis of Alzheimer's disease 4 1.3 Hypotheses on the etiology of Alzheimer's disease....4 1.4 Infectious agents 5 1. 5 Neurotoxic agents 6 1.6 Dysfunction of the blood-brain barrier 6 1.7 Neurotransmitter and receptor deficiencies, neurotransmitter cytotoxicity 7 1. -

Limbic System)
Telencephalon Structure of telencephalon Gray matter Cortex (pallium) Basal ganglia (striatum) White matter - pathways Projection Commissural Association Cerebral cortex ALLOCORTEX 3-5 layers a) palleocortex (rhinencephalon) archicortex b) archicortex (limbic system) MESOCORTEX = palleocortex peripaleocortex, periarchicortex NEOCORTEX (ISOCORTEX) 6 layers Rhinencephalon Limbic lobe Bulbus olfactorius Gyrus cinguli Tractus olfactorius Gyrus parahippocampalis Tuberculum olf. Indusium griseum Stria olf. med. et lat. HippocampalARCHICORTEX complex: Hippocampus (cornu ammonis, CA) Gyrus dentatus Subiculum Fornix Limbic system – classic conception Papez‘s circuit (James Papez 1939) without specific function ncl. anterior thalami tr. mammilo-thalamicus ncl. mamillaris gyrus cinguli fornix gyrus parahippocampalis hippocampus RECENT CONCEPTION OF LIMBIC FOREBRAIN • basomedial telencephalon, structures of diencephalon and mesencephalon for emotion and motivation of our behavior Regular structures • g. cinguli, g. parahippocampalis, hippocampus, insular cortex, neocortical regions of forebrain - basal frontotemporal regions, orbital cortex • area septalis, amygdalar ncll., ventral striatum (pallidum) • ncl. anterior et medialis dorsalis thalami, habenulla • hypothalamus (ncl. mammillaris) Limbic system – classic conception Papez‘s circuit (James Papez 1939) Image of tooth pain Image of fear Reminiscence of music hearing Brodman’s map (cytoarchitectonic map of cortex) 11 regiones 52 areae Functional regions of cortex Primary motor c. (a 4), primary