Heat of Formation of Decaborane Walter H
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
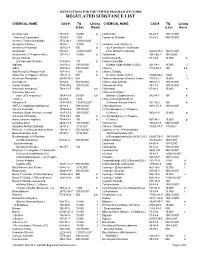
Regulated Substance List
INSTRUCTIONS FOR THE UNIFIED PROGRAM (UP) FORM REGULATED SUBSTANCE LIST CHEMICAL NAME CAS # TQ Listing CHEMICAL NAME CAS # TQ Listing (Lbs) Basis (Lbs) Basis Acetaldehyde 75-07-0 10,000 g Cantharidin 56-25-7 100/10,0001 * Acetone Cyanohydrin 75-86-5 1,000 Carbachol Chloride 51-83-2 500/10,0001 Acetone Thiosemicarbazide 1752-30-3 1,000/10,0001 Acetylene (Ethyne) 74-86-2 10,000 f Carbamic Acid, Methyl-,o- Acrolein (2-Propenal) 107-02-8 500 b (((2,4-Dimethyl-1,3-Dithiolan- Acrylamide 79-06-1 1,000/10,0001 2-YL) Methylene)Amino)- 26419-73-8 100/10,0001 Acrylonitrile (2- Propenenitrile) 107-13-1 10,000 b Carbofuran 1563-66-2 10/10,0001 Acrylyl Chloride Carbon Disulfide 75-15-0 10,000 b (2-Propenoyl Chloride) 814-68-6 100 b Carbon Oxysulfide Aldicarb 116-06-3 100/10,0001 (Carbon Oxide Sulfide (COS)) 463-58-1 10,000 f Aldrin 309-00-2 500/10,0001 Chlorine 7782-50-5 100 a,b Allyl Alcohol (2-Propen-1-ol) 107-18-6 1,000 b Chlorine Dioxide Allylamine (2-Propen-1-Amine) 107-11-9 500 b (Chlorine Oxide (ClO2)) 10049-04-4 1,000 c Aluminum Phosphide 20859-73-8 500 Chlorine Monoxide (Chlorine Oxide) 7791-21-1 10,000 f Aminopterin 54-62-6 500/10,0001 Chlormequat Chloride 999-81-5 100/10,0001 Amiton Oxalate 3734-97-2 100/10,0001 Chloroacetic Acid 79-11-8 100/10,0001 Ammonia, Anhydrous 2 7664-41-7 500 a,b Chloroform 67-66-3 10,000 b Ammonia, Aqueous Chloromethyl Ether (conc 20% or greater) 7664-41-7 20,000 a,b (Methane,Oxybis(chloro-) 542-88-1 100 b * Aniline 62-53-3 1,000 Chloromethyl Methyl Ether Antimycin A 1397-94-0 1,000/10,0001 (Chloromethoxymethane) -

Thermodynamic Hydricity of Small Borane Clusters and Polyhedral Closo-Boranes
molecules Article Thermodynamic Hydricity of Small Borane Clusters y and Polyhedral closo-Boranes Igor E. Golub 1,* , Oleg A. Filippov 1 , Vasilisa A. Kulikova 1,2, Natalia V. Belkova 1 , Lina M. Epstein 1 and Elena S. Shubina 1,* 1 A. N. Nesmeyanov Institute of Organoelement Compounds and Russian Academy of Sciences (INEOS RAS), 28 Vavilova St, 119991 Moscow, Russia; [email protected] (O.A.F.); [email protected] (V.A.K.); [email protected] (N.V.B.); [email protected] (L.M.E.) 2 Faculty of Chemistry, M.V. Lomonosov Moscow State University, 1/3 Leninskiye Gory, 119991 Moscow, Russia * Correspondence: [email protected] (I.E.G.); [email protected] (E.S.S.) Dedicated to Professor Bohumil Štibr (1940-2020), who unfortunately passed away before he could reach the y age of 80, in the recognition of his outstanding contributions to boron chemistry. Academic Editors: Igor B. Sivaev, Narayan S. Hosmane and Bohumír Gr˝uner Received: 6 June 2020; Accepted: 23 June 2020; Published: 25 June 2020 MeCN Abstract: Thermodynamic hydricity (HDA ) determined as Gibbs free energy (DG◦[H]−) of the H− detachment reaction in acetonitrile (MeCN) was assessed for 144 small borane clusters (up 2 to 5 boron atoms), polyhedral closo-boranes dianions [BnHn] −, and their lithium salts Li2[BnHn] (n = 5–17) by DFT method [M06/6-311++G(d,p)] taking into account non-specific solvent effect (SMD MeCN model). Thermodynamic hydricity values of diborane B2H6 (HDA = 82.1 kcal/mol) and its 2 MeCN dianion [B2H6] − (HDA = 40.9 kcal/mol for Li2[B2H6]) can be selected as border points for the range of borane clusters’ reactivity. -

Diborane (B2H6) 2 Heavier Boron Hydrides in the Form of Volatile Liquids Such As
Even in the absence of contamination, diborane is so reactive that it will slowly decompose by itself, forming H gas and Diborane (B2H6) 2 heavier boron hydrides in the form of volatile liquids such as Storage and Delivery B5H9 and sublimable solids such as B10H14. These heavier hydrides are delivered with the gas and can cause Air Products supplies a wide range of gases and chemicals contamination of the process and fouling of the delivery system that are used for integrated circuit, flat panel display and components. Figure 1 shows the internals of the first stage of photovoltaic device manufacturing. Understanding the proper a two-stage gas regulator that was fouled by B10H14 deposits. storage and handling requirements of these specialty materials is essential to obtain consistent results and to ensure the highest level of process safety. In this technical bulletin, we will provide the user with some of the knowledge required for its effective storage and use. Figure 1: Picture of the first stage of diborane mixture regulator Diborane (formula B2H6) is a colorless, spontaneously fouled with solid decomposition products. flammable (pyrophoric) gas. It burns rapidly in air and reacts vigorously with water with the evolution of heat. The gas has The gas-phase decomposition rate of diborane was shown to a distinct, noxious odor and is extremely toxic by inhalation or increase with concentration and with temperature. Pure by skin exposure. Because of these hazards, it is essential diborane is so reactive in its pure state that it may only be that all gas handling systems used for diborane be tightly stored or transported if surrounded with dry ice to prevent rapid sealed and carefully leak checked. -

276262828.Pdf
FUNDAMENTAL STUDIES OF CATALYTIC DEHYDROGENATION ON ALUMINA-SUPPORTED SIZE-SELECTED PLATINUM CLUSTER MODEL CATALYSTS by Eric Thomas Baxter A dissertation submitted to the faculty of The University of Utah in partial fulfillment of the requirements for the degree of Doctor of Philosophy Department of Chemistry The University of Utah May 2018 Copyright © Eric Thomas Baxter 2018 All Rights Reserved T h e U n iversity of Utah Graduate School STATEMENT OF DISSERTATION APPROVAL The dissertation of Eric Thomas Baxter has been approved by the following supervisory committee members: Scott L. Anderson , Chair 11/13/2017 Date Approved Peter B. Armentrout , Member 11/13/2017 Date Approved Marc D. Porter , Member 11/13/2017 Date Approved Ilya Zharov , Member 11/13/2017 Date Approved Sivaraman Guruswamy , Member 11/13/2017 Date Approved and by Cynthia J. Burrows , Chair/Dean of the Department/College/School of Chemistry and by David B. Kieda, Dean of The Graduate School. ABSTRACT The research presented in this dissertation focuses on the use of platinum-based catalysts to enhance endothermic fuel cooling. Chapter 1 gives a brief introduction to the motivation for this work. Chapter 2 presents fundamental studies on the catalytic dehydrogenation of ethylene by size-selected Ptn (n = 4, 7, 8) clusters deposited onto thin film alumina supports. The model catalysts were probed by a combination of experimental and theoretical techniques including; temperature-programmed desorption and reaction (TPD/R), low energy ion scattering spectroscopy (ISS), X-ray photoelectron spectroscopy (XPS), plane wave density-functional theory (PW-DFT), and statistical mechanical theory. It is shown that the Pt clusters dehydrogenated approximately half of the initially adsorbed ethylene, leading to deactivation of the catalyst via (coking) carbon deposition. -

Minutesbasel Approved
Approved minutes, Basel 2018 International Union of Pure and Applied Chemistry Division VIII Chemical Nomenclature and Structure Representation Approved minutes for Division Committee Meeting Basel, Switzerland, 13–14 August, 2018 1. Welcome, introductory remarks and housekeeping announcements Alan Hutton (ATH) welcomed everybody to the meeting, extending a special welcome to those who were attending the Division Committee (DC) meeting for the first time, and noting the presence of three former Presidents of the Division, as well as the current Secretary General of IUPAC. He described the house rules and arrangements for the course of the meeting. 2. Attendance and apologies Present: Alan T. Hutton (President, ATH), Karl-Heinz Hellwich (Past-President, KHH), Risto S. Laitinen (Secretary, RSL), Michael A. Beckett (MAB), Edwin C. Constable (ECC), Ture Damhus (TD), Richard M. Hartshorn (RMH), Elisabeth Mansfield (EM), Gerry P. Moss (GPM), Michelle M. Rogers (MMR), Molly A. Strausbaugh (MAS), Clare Tovee (CT), Andrey Yerin (AY) (see also Appendix 1) Apologies: Fabio Aricò (FA), Maria Atanasova Petrova (MA), Neil Burford (NB), (JC), Ana Maria da Costa Ferreira (ACF), Safiye Erdem (SE), Rafał Kruszyński (RK), Robin Macaluso (RM), , Ladda Meesuk (LM), Ebbe Nordlander (EN), Amelia P. Rauter (APR), Erik Szabó (ES), Keith T. Taylor (KTT), Jiří Vohlídal (JV) Invited observer: G. Jeffery Leigh (GJL) No replies: József Nagy, Sangho Koo 3. Introduction of attendees A short round of introductions was made. A new titular member, Prof. Edwin C. Constable and a new Associate Member, Dr. Clare Tovee, were attending the meeting of the Division Committee for the first time in this function. KHH informed the meeting that the following persons had passed away during recent years: Peter A. -

Identification of Diborane(4) with Bridging B–H–B Bonds
Chemical Science View Article Online EDGE ARTICLE View Journal | View Issue Identification of diborane(4) with bridging B–H–B bonds† Cite this: Chem. Sci.,2015,6, 6872 Sheng-Lung Chou,a Jen-Iu Lo,a Yu-Chain Peng,a Meng-Yeh Lin,a Hsiao-Chi Lu,a Bing-Ming Cheng*a and J. F. Ogilvieb The irradiation of diborane(6) dispersed in solid neon at 3 K with tunable far-ultraviolet light from a Received 17th July 2015 synchrotron yielded a set of IR absorption lines, the pattern of which implies a carrier containing two Accepted 14th August 2015 boron atoms. According to isotope effects and quantum-chemical calculations, we identified this new DOI: 10.1039/c5sc02586a species as diborane(4), B2H4, possessing two bridging B–H–B bonds. Our work thus establishes a new www.rsc.org/chemicalscience prototype, diborane(4), for bridging B–H–B bonds in molecular structures. Introduction The B–H–B linkage in these compounds is considered to be an atypical electron-decient covalent chemical bond.7 Creative Commons Attribution 3.0 Unported Licence. The structure that B2H6 adopts in its electronic ground state According to calculations, diborane species possessing less and most stable conformation contains two bridging B–H–B than 6 hydrogen atoms and bridging B–H–Bbondscan 8–13 bonds, and four terminal B–H bonds. Although Bauer, from exist, but all possible candidates are transient species, experiments with the electron diffraction of gaseous samples in difficult to prepare and to identify. Since the pioneering work the laboratory of Pauling and Brockway, favoured a structure of involving absorption spectra of samples at temperatures less 14,15 diborane(6) with a central B–B bond analogous to that of than 100 K, many free radicals and other unstable 16–20 ethane,1 Longuet-Higgins deduced the bridged structure for compounds dispersed in inert hosts have been detected. -

UC Riverside Electronic Theses and Dissertations
UC Riverside UC Riverside Electronic Theses and Dissertations Title Carboranes: Building Blocks for Materials and Ligand Development Permalink https://escholarship.org/uc/item/2vp9m2z6 Author Estrada, Jess Steven Publication Date 2017 Peer reviewed|Thesis/dissertation eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA RIVERSIDE Carboranes: Building Blocks for Materials and Ligand Development A Dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Chemistry by Jess Steven Estrada September 2017 Dissertation Committee: Dr. Vincent Lavallo, Chairperson Dr. Richard Hooley Dr. Pingyun Feng Copyright by Jess Steven Estrada 2017 The Dissertation Jess Steven Estrada is approved: Committee Chairperson University of California, Riverside Acknowledgements I would like to thank Dr. Vincent Lavallo for giving me the opportunity to join his lab and for all of his help and support throughout graduate school. I owe a lot of my success to you. I would also like to thank the amazing faculty at UCR for all of their help and knowledge they have provided. I truly enjoyed every class I took here at UCR. In addition to the faculty, the staff in the chemistry department has been amazing over the last five years and played a huge role in my success as well so I would specifically like to thank Dr. Borchardt, the NMR genius, Dr. Fook for all of his help with my great looking X-ray structures, Christina Youhas for answering literally every question I ever had and being so kind about answering. My lab mates, who created the most unique group of people probably in the history of UCR and that I’ve ever had the pleasure of working with. -

United States Patent Office Patented Jan
3,489,812 United States Patent Office Patented Jan. 13, 1970 2 3,489,812 and sufficient dialkyl sulfide to permit convenient stirring METHOD OF PREPARING BIS-(DIETHYL of the reactant mixture. The reaction should be carried SULFIDE)-DECABORANE out in the absence of air or other oxidizing gas, suitably Mervin D. Marshall, Fombell, and Richard M. Hunt and by using a vacuum or nonoxidizing cover gas such as Gerald T. Heiferan, Butler, Pa., assignors to Mine nitrogen or argon. Safety Appliances Company, a corporation of Pennsyl vania tion:The following examples are illustrative of this inven No Drawing. Filed Jan. 15, 1968, Ser. No. 697,596 Int, C. C07f 5/02 EXAMPLE I U.S. C. 260-606.5 1 Claim O A glass reactor equipped with a magnetic stirrer and connected to a glass vacuum apparatus was charged with ABSTRACT OF THE DISCLOSURE 3.513 g. of (NH4)2BioHo (22.6 millimols) and 40 ml. of diethyl sulfide, cooled to about 4° C. and evacuated. Bis-(diethyl sulfide)-decaborane is prepared by reac Measured aliquots of anhydrous hydrogen chloride (92. tion of ammonium decahydrodecarborate, HCl and di 15 immoles) were added in two equal increments. This reac ethyl sulfide. tion mixture was allowed to warm to room temperature and stirred for 72 hours. A white precipitate formed which Bis-(dialkyl sulfide)-decaboranes, such as bis-(dimethyl was filtered from the diethyl sulfide solution. The evapora sulfide)-decaborane, BioH12S (CH3)2)2, and bis-(diethyl tion of excess diethyl sulfide from the solution gave 4.596 sulfide)-decaborane, BioH12S (C2H5)2)2, have been used g. -

Nite States Patent O?Ice Patented Apr
3,030,423 nite States Patent O?ice Patented Apr. 17, 1962 1 2 cuprous chloride catalyst employed also can be varied 3,030,423 REACTION PRODUCTS 0F ACETYLENES widely, generally being in the range of from 0.005 to 0.5 AND DECABORANE mole of cuprous chloride per mole of decaborane and Starling K. Alley, Beverly Hills, Calif., and Otto Fuchs, preferably from 0.01 to 0.2 mole of cuprous chloride per Tonawanzia, N.Y., assignors to Olin Mathieson Chemi- 5 mole of decaborane. The reaction temperature can vary cal Corporation, a corporation of Virginia from 100 to 350° C. and preferably from 150 to 250° C. No Drawing. Filed Feb. 13, 1959, Ser. No. 793,211 The pressure can vary from atmospheric pressure to about 3 Claims. (Cl. 260-6065) 700 p.s.i.g., although closed system reactions are usually conducted at about 100 to 600 p.s.i.g. The reaction gen This invention relates to solid products obtained by 10 erally requires about 1 to 10 hours depending upon the the reaction of acetylene or methylacetylene with deca~ ratio of reactants and the temperature and pressure em borane. ployed. The solid products of this invention, when incorporated Although the reaction will proceed in the absence of a with suitable oxidizers such as ammonium perchlorate, solvent, a solvent common for the reactants but inert with potassium perchlorate, sodium perchlorate, etc., yield 15 respect to the reactants under the reaction conditions is solid propellants generally suitable for rocket power usually employed. Such solvents include aliphatic hydro plants and other jet propelled devices. -

Chapter 9 Hydrogen
Chapter 9 Hydrogen Diborane, B2 H6• is the simplest member of a large class of compounds, the electron-deficient boron hydrid Like all boron hydrides, it has a positive standard free energy of formation, and so cannot be prepared direct from boron and hydrogen. The bridge B-H bonds are longer and weaker than the tenninal B-H bonds (13: vs. 1.19 A). 89.1 Reactions of hydrogen compounds? (a) Ca(s) + H2Cg) ~ CaH2Cs). This is the reaction of an active s-me: with hydrogen, which is the way that saline metal hydrides are prepared. (b) NH3(g) + BF)(g) ~ H)N-BF)(g). This is the reaction of a Lewis base and a Lewis acid. The product I Lewis acid-base complex. (c) LiOH(s) + H2(g) ~ NR. Although dihydrogen can behave as an oxidant (e.g., with Li to form LiH) or reductant (e.g., with O2 to form H20), it does not behave as a Br0nsted or Lewis acid or base. It does not r with strong bases, like LiOH, or with strong acids. 89.2 A procedure for making Et3MeSn? A possible procedure is as follows: ~ 2Et)SnH + 2Na 2Na+Et3Sn- + H2 Ja'Et3Sn- + CH3Br ~ Et3MeSn + NaBr 9.1 Where does Hydrogen fit in the periodic chart? (a) Hydrogen in group 1? Hydrogen has one vale electron like the group 1 metals and is stable as W, especially in aqueous media. The other group 1 m have one valence electron and are quite stable as ~ cations in solution and in the solid state as simple 1 salts. -

Safety Data Sheet
SAFETY DATA SHEET Revision Date 14-February-2020 Revision Number 2 1. Identification Product Name Decaborane Cat No. : 87892 CAS-No 17702-41-9 Synonyms No information available Recommended Use Laboratory chemicals. Uses advised against Food, drug, pesticide or biocidal product use. Details of the supplier of the safety data sheet Company Importer/Distributor Manufacturer Fisher Scientific Alfa Aesar 112 Colonnade Road, Thermo Fisher Scientific Chemicals, Inc. Ottawa, ON K2E 7L6, 30 Bond Street, Ward Hill, MA 01835-8099 Canada Tel: 800-343-0660 Fax: 800-322-4757 Tel: 1-800-234-7437 Email: [email protected] www.alfa.com Emergency Telephone Number During normal business hours (Monday-Friday, 8am-7pm EST), call (800) 343-0660. After normal business hours, call Carechem 24 at (800) 579-7421. 2. Hazard(s) identification Classification WHMIS 2015 Classification Classified as hazardous under the Hazardous Products Regulations (SOR/2015-17) Flammable solids Category 2 Acute oral toxicity Category 3 Acute dermal toxicity Category 3 Acute Inhalation Toxicity Category 2 Physical Hazards Not Otherwise Classified Category 1 Risk of explosion if heated under confinement Label Elements Signal Word Danger Hazard Statements Flammable solid Fatal if inhaled Toxic if swallowed or in contact with skin ______________________________________________________________________________________________ Page 1 / 7 Decaborane Revision Date 14-February-2020 ______________________________________________________________________________________________ Risk of explosion if heated under confinement Precautionary Statements Manufacturer Alfa Aesar Thermo Fisher Scientific Chemicals, Inc. 30 Bond Street, Ward Hill, MA 01835-8099 Tel: 800-343-0660 Fax: 800-322-4757 Email: [email protected] www.alfa.com Prevention Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. -

Tuning Photophysical Properties in Closo-Decaborane Cluster Derivatives
TUNING PHOTOPHYSICAL PROPERTIES IN CLOSO-DECABORANE CLUSTER DERIVATIVES by Mustapha B. Abdulmojeed A Thesis Submitted in Partial Fulfillment of the Requirements for the Degree of Master of Science in Chemistry Middle Tennessee State University 2020 Thesis Committee: Dr. Andrienne C. Friedli, Major Advisor Dr. Piotr Kaszynski, Committee Member Dr. Kevin Bicker, Committee Member Dr. Justin Miller, Committee Member ii Tuning Photophysical Properties in Closo-Decaborane Cluster Derivatives by Mustapha B. Abdulmojeed Approved: _____________________________________________ Dr. Andrienne C. Friedli, Committee Chair _____________________________________________ Dr. Piotr Kaszynski, Committee Member _____________________________________________ Dr. Kevin L. Bicker, Committee Member _____________________________________________ Dr. Justin M. Miller, Committee Member _____________________________________________ Dr. Paul G. Van Patten, Chair, Department of Chemistry _____________________________________________ Dr. David L. Butler, Dean, College of Graduate Studies ii iii ACKNOWLEDGEMENTS All thanks belong to God who has made it possible to complete this piece of work. Thanks to my wife for her love, care and understanding, taking care of our kids while I was away. Special thanks to my advisor, Dr. Andrienne Friedli for her support, guide and patiently delivered instructions through-out the course of this project. Dziękuję to my co- advisor, Dr. Piotr Kaszynski whose constant push and motivation from across the Atlantic Ocean kept me going, no amount of words could express my gratitude to both of you. Sincere appreciation also goes to Dr. Szymon Kapuscinski who is not just an excellent senior colleague but a great personality whose help in this work cannot be measured. I also want to thank MTSU chemistry department for the funding opportunity. I extend my gratitude to all faculty members in the department for creating a conducive and welcoming environment to learn and train as a chemist.