Template for Electronic Submission to ACS Journals
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Status of Industry Efforts to Replace Halon Fire Extinguishing Agents
STATUS OF INDUSTRY EFFORTS TO REPLACE HALON FIRE EXTINGUISHING AGENTS Robert T. Wickham, P.E. March 16, 2002 WICKHAM ASSOCIATES NOTICE This document is disseminated under the sponsorship of the U.S. Environmental Protection Agency in the interest of information exchange. The views expressed in this report are those of the author and do not necessarily reflect those of the US Environmental Protection Agency. The US Environmental Protection Agency does not assume liability for the contents or use thereof and further the Agency does not endorse products or manufacturers. Trade or manufacturer's names appear herein solely because they are considered essential to the objective of this report. The following commercial products (requiring a trademark designation ® or ™) are mentioned in this report. Due to the frequency of usage, trademarks are not indicated. Mention of a product does not constitute endorsement or rejection of the product. Ansul FE-25 Argonite FE-36 Argotec FM-200 CEA-410 Halotron I CEA-614 Inergen CEA-308 NAF P-IV Envirogel NAF S-III FE-13 NN100 FE-227 Triodide FE-241 There are no restrictions on copying or distribution of this document. Additional copies of this report are available from ….. Wickham Associates 9 Winding Brook Drive Stratham, New Hampshire 03885 USA Tel: +603-772-3229 Fax: +603-772-7305 email: [email protected] The report is available at http://home.attbi.com/~wickham/downloads.htm for downloading in Adobe Acrobat portable document format (pdf). Preface This report is not intended to be a market study, but instead a snapshot of the progress industry and the government are making in employing non ozone depleting alternatives to halons in the fire protection sector. -

Shelf-Stable Electrophilic Trifluoromethylating Reagents: a Brief Historical Perspective
Shelf-stable electrophilic trifluoromethylating reagents: A brief historical perspective Norio Shibata*1, Andrej Matsnev1 and Dominique Cahard*2 Review Open Access Address: Beilstein J. Org. Chem. 2010, 6, No. 65. doi:10.3762/bjoc.6.65 1Department of Frontier Materials, Graduate School of Engineering, Nagoya Institute of Technology, Gokiso, Showa-ku, Nagoya Received: 15 March 2010 466-8555, Japan and 2UMR 6014 CNRS – C.O.B.R.A. Université et Accepted: 21 May 2010 INSA de Rouen, 1 rue Tesnière, 76130 Mont Saint Aignan, France Published: 16 June 2010 Email: Guest Editor: D. O'Hagan Norio Shibata* - [email protected]; Dominique Cahard* - [email protected] © 2010 Shibata et al; licensee Beilstein-Institut. License and terms: see end of document. * Corresponding author Keywords: asymmetric synthesis; electrophilic; fluorine; reagent; trifluoromethylation Abstract Since the discovery by Yagupolskii and co-workers that S-trifluoromethyl diarylsulfonium salts are effective for the trifluoro- methylation of thiophenolates, the design and synthesis of electrophilic trifluoromethylating reagents have been extensively researched in both academia and industry, due to the significant unique features that trifluoromethylated compounds have in phar- maceuticals, agricultural chemicals, and functional materials. Several effective reagents have been developed by the groups of Yagupolskii, Umemoto, Shreeve, Adachi, Magnier, Togni and Shibata. Due to the high stability and reactivity of these reagents, a series of Umemoto reagents, Togni reagent and Shibata reagent are now commercially available. In this review, we wish to briefly provide a historical perspective of the development of so-called “shelf-stable electrophilic trifluoromethylating reagents”, although this field is in constant development. Review The chemistry of fluoro-organic compounds is one of the areas [3]. -
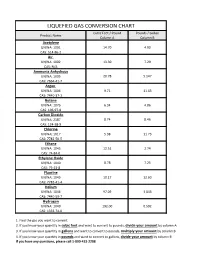
Liquefied Gas Conversion Chart
LIQUEFIED GAS CONVERSION CHART Cubic Feet / Pound Pounds / Gallon Product Name Column A Column B Acetylene UN/NA: 1001 14.70 4.90 CAS: 514-86-2 Air UN/NA: 1002 13.30 7.29 CAS: N/A Ammonia Anhydrous UN/NA: 1005 20.78 5.147 CAS: 7664-41-7 Argon UN/NA: 1006 9.71 11.63 CAS: 7440-37-1 Butane UN/NA: 1075 6.34 4.86 CAS: 106-97-8 Carbon Dioxide UN/NA: 2187 8.74 8.46 CAS: 124-38-9 Chlorine UN/NA: 1017 5.38 11.73 CAS: 7782-50-5 Ethane UN/NA: 1045 12.51 2.74 CAS: 74-84-0 Ethylene Oxide UN/NA: 1040 8.78 7.25 CAS: 75-21-8 Fluorine UN/NA: 1045 10.17 12.60 CAS: 7782-41-4 Helium UN/NA: 1046 97.09 1.043 CAS: 7440-59-7 Hydrogen UN/NA: 1049 192.00 0.592 CAS: 1333-74-0 1. Find the gas you want to convert. 2. If you know your quantity in cubic feet and want to convert to pounds, divide your amount by column A 3. If you know your quantity in gallons and want to convert to pounds, multiply your amount by column B 4. If you know your quantity in pounds and want to convert to gallons, divide your amount by column B If you have any questions, please call 1-800-433-2288 LIQUEFIED GAS CONVERSION CHART Cubic Feet / Pound Pounds / Gallon Product Name Column A Column B Hydrogen Chloride UN/NA: 1050 10.60 8.35 CAS: 7647-01-0 Krypton UN/NA: 1056 4.60 20.15 CAS: 7439-90-9 Methane UN/NA: 1971 23.61 3.55 CAS: 74-82-8 Methyl Bromide UN/NA: 1062 4.03 5.37 CAS: 74-83-9 Neon UN/NA: 1065 19.18 10.07 CAS: 7440-01-9 Mapp Gas UN/NA: 1060 9.20 4.80 CAS: N/A Nitrogen UN/NA: 1066 13.89 6.75 CAS: 7727-37-9 Nitrous Oxide UN/NA: 1070 8.73 6.45 CAS: 10024-97-2 Oxygen UN/NA: 1072 12.05 9.52 CAS: 7782-44-7 Propane UN/NA: 1075 8.45 4.22 CAS: 74-98-6 Sulfur Dioxide UN/NA: 1079 5.94 12.0 CAS: 7446-09-5 Xenon UN/NA: 2036 2.93 25.51 CAS: 7440-63-3 1. -
Ozone Depleting Substances
Ozone Depleting Substances What are they? Ozone depleting substances are chemicals that destroy the earth’s protective ozone layer. They include: • chlorofluorocarbons (CFCs) • halons • carbon tetrachloride (CCl4) • methyl chloroform (CH3CCl3) • hydrobromofluorocarbons (HBFCs) • hydrochlorofluorocarbons (HCFCs) • methyl bromide (CH3Br) • bromochloromethane (CH2BrCl) The use of these chemicals is controlled by the Montreal Protocol on Substances that Deplete the Ozone Layer (the Montreal Protocol). There are other ozone depleting substances, but their ozone depleting effects are very small, so they are not controlled by the Montreal Protocol. One kilogram of halon 1211 can destroy 50 tonnes of ozone What is ozone depleting potential? Ozone depleting potential is a measure of how much damage a chemical can cause to the ozone layer compared with a similar mass What did we use ozone depleting of trichlorofluoromethane (CFC-11). CFC-11, with an ozone depleting potential of 1.0, is substances for? used as the base figure for measuring ozone depletion potential. The higher the number, The main uses of ozone depleting substances the more damage a chemical can cause to include; CFCs and HCFCs in refrigerators and the ozone layer. Bromotrifluoromethane air conditioners, HCFCs and halons in fire (halon -1301) has an ozone depleting potential extinguishers, CFCs and HCFCs in foam, CFCs of 10.0. Carbon dioxide (CO2) is a naturally and HCFCs as aerosol propellants and methyl occurring greenhouse gas, but has an ozone bromide for fumigation of soil, structures and depleting potential of 0. goods to be imported or exported. Have we stopped using ozone Why do we still use some ozone depleting substances? depleting substances? Production of most ozone depleting Some substances with a high ozone depleting substances has been phased out under the potential are still used in quarantine and Montreal Protocol. -

Perfluoro-3-Ethyl-2,4-Dimethyl-3
Journal of Fluorine Chemistry 227 (2019) 109370 Contents lists available at ScienceDirect Journal of Fluorine Chemistry journal homepage: www.elsevier.com/locate/fluor Perfluoro-3-ethyl-2,4-dimethyl-3-pentyl persistent radical: A new reagent T for direct, metal-free radical trifluoromethylation and polymer initiation ⁎ ⁎ ⁎ Haibo Meia, Jianlin Hana, , Sarah Whiteb, , Greg Butlerb, Vadim A. Soloshonokc,d, a College of Chemical Engineering, Nanjing Forestry University, Nanjing, 210037, China b Oakwood Chemical, Inc. 730 Columbia Hwy. N, Estill, SC, 29918, USA c Department of Organic Chemistry I, Faculty of Chemistry, University of the Basque Country UPV/EHU, Paseo Manuel Lardizábal 3, 20018, San Sebastián, Spain d IKERBASQUE, Basque Foundation for Science, María Díaz de Haro 3, Plaza Bizkaia, 48013, Bilbao, Spain ARTICLE INFO ABSTRACT Keywords: This review comprehensively profiles perfluoro-3-ethyl-2,4-dimethyl-3-pentyl persistent radical (PPFR) asanew Fluorine reagent for radical trifluoromethylation, trifluoromethylation/fluorination and polymer initiation. The PPFRis Trifluoromethylation perfectly stable at ambient conditions, but at temperatures above 80 °C undergoes β-scission to generate tri- Persistent radical fluoromethyl radical. This property can be used to initiate various chain-polymerization or trifluoromethylation Fluorination reactions. The unique feature of this process, distinguishing it from all other known methods for ·CF3-radical Polymer generation, is that the radical is produced under neutral, inert and additive-free -

Halons Technical Options Committee 2018 AssessMent Report
Halons Technical Options Committee 2018 Assess ment Report Volume 1 Montreal Protocol on Substances that Deplete the Ozone Layer Ozone Secretariat MONTREAL PROTOCOL ON SUBSTANCES THAT DEPLETE THE OZONE LAYER REPORT OF THE HALONS TECHNICAL OPTIONS COMMITTEE DECEMBER 2018 VOLUME 1 2018 ASSESSMENT REPORT i Montreal Protocol on Substances that Deplete the Ozone Layer Report of the Halons Technical Options Committee December 2018 Volume 1 2018 ASSESSMENT REPORT The text of this report is composed in Times New Roman Co-ordination: Halons Technical Options Committee Composition of the report: Halons Technical Options Committee Reproduction: Ozone Secretariat Date: December 2018 Under certain conditions, printed copies of this report are available from: UNITED NATIONS ENVIRONMENT PROGRAMME Ozone Secretariat, P.O. Box 30552, Nairobi, Kenya This document is also available in portable document format from the Ozone Secretariat's website: https://ozone.unep.org/sites/default/files/Assessment_Panel/Assessment_Panels/TEAP/R eports/HTOC/HTOC_assessment_2018.pdf No copyright involved. This publication may be freely copied, abstracted and cited, with acknowledgement of the source of the material. ISBN: 978-9966-076-48-9 iii Disclaimer The United Nations Environment Programme (UNEP), the Technology and Economic Assessment Panel (TEAP) Co-chairs and members, the Technical Options Committees Co-chairs and members, the TEAP Task Forces Co-chairs and members, and the companies and organisations that employ them do not endorse the performance, worker safety, or environmental acceptability of any of the technical options discussed. Every industrial operation requires consideration of worker safety and proper disposal of contaminants and waste products. Moreover, as work continues - including additional toxicity evaluation - more information on health, environmental and safety effects of alternatives and replacements will become available for use in selecting among the options discussed in this document. -

Advances in Catalytic Enantioselective Fluorination, Mono‑,Di‑, and Trifluoromethylation, and Trifluoromethylthiolation Reactions ‡ ‡ † Xiaoyu Yang, Tao Wu, Robert J
Review pubs.acs.org/CR Advances in Catalytic Enantioselective Fluorination, Mono‑,Di‑, and Trifluoromethylation, and Trifluoromethylthiolation Reactions ‡ ‡ † Xiaoyu Yang, Tao Wu, Robert J. Phipps, and F. Dean Toste* Department of Chemistry, University of California, Berkeley, California 94720, United States 6. Catalytic Enantioselective Trifluoromethylthiola- tion 865 7. Summary and Outlook 866 Author Information 866 Corresponding Author 866 Present Address 866 Author Contributions 866 Notes 866 CONTENTS Biographies 866 Acknowledgments 867 1. Introduction 826 References 867 2. Catalytic Enantioselective Fluorination 827 2.1. Electrophilic Fluorination 827 2.1.1. Metal-Catalyzed Fluorination Involving 1. INTRODUCTION Enolates 827 Despite being largely absent from natural products and 2.1.2. Metal-Catalyzed Fluorination Not In- biological processes, fluorine plays a conspicuous and volving Enolates 834 increasingly important role within pharmaceuticals and agro- 2.1.3. Organocatalytic Electrophilic Fluorina- − chemicals, as well as in materials science.1a c Indeed, as many as tion 835 35% of agrochemicals and 20% of pharmaceuticals on the 2.1.4. Fluorination Using Multiple Catalysts 846 market contain fluorine.1d Fluorine is the most electronegative 2.1.5. One-Pot and Tandem Processes 848 element in the periodic table, and the introduction of one or 2.2. Nucleophilic Fluorination 850 more fluorine atoms into a molecule can result in greatly 3. Catalytic Enantioselective Trifluoromethylation perturbed properties. Fluorine substituents can potentially and Perfluoroalkylation 853 impact a number of variables, such as the acidity or basicity of 3.1. Asymmetric Nucleophilic Trifluoromethyla- neighboring groups, dipole moment, and properties such as tion 853 lipophilicity, metabolic stability, and bioavailability. The 3.1.1. Overview of Nucleophilic Trifluorome- multitude of effects that can arise from the introduction of thylation 853 fluorine in small molecules in the context of medicinal 3.1.2. -

CV-PSB-August 2020
Curriculum Vitae Phil S. Baran Appointment: Scripps Research Professor, Department of Chemistry 10550 North Torrey Pines Road, BCC-436 La Jolla, California 92037 Telephone: (858) 784-7373 Facsimile: (858) 784-7575 Email: [email protected] Website: www.scripps.edu/chem/baran/ January, 2013 Darlene Shiley Professor of Chemistry April, 2009 Member, Skaggs Institute for Chemical Biology June, 2008 Professor of Chemistry July, 2006 Associate Professor of Chemistry (with Tenure) June, 2003 Assistant Professor of Chemistry Date/Place of Birth: 10 Aug 1977 / Denville, NJ, USA Citizenship: United States Education 2001 – 2003 Postdoctoral Associate Advisor: Professor E.J. Corey Harvard University, Cambridge, Massachusetts 1997 – 2001 Ph.D. Graduate Student in Chemistry Advisor: Professor K.C. Nicolaou The Scripps Research Institute, La Jolla, California 1995 – 1997 B.S. with Honors in Chemistry Advisor: Professor D.I. Schuster New York University, New York, New York 1991 – 1995 Simultaneous high school graduation from Mt. Dora High School and A.A. degree with honors, Lake Sumter Community College, Florida Awards • Inhoffen Medal, 2019 • Manchot Research Professorship, 2017 • Member, The National Academy of Sciences, 2017 • Emanuel Merck Lectureship, 2017 • Blavatnik National Laureate in Chemistry, 2016 • ACS Elias J. Corey Award, 2016 • Member, American Academy of Arts and Sciences, 2015 • College of Arts and Science Alumni Distinguished Service Award, New York University, 2015 • Reagent of the Year Award (EROS), 2015 • Mukaiyama Award, 2014 • -

ACE Tips for Filing EPA ODS
Automated Commercial Environment ACE Tips for Filing EPA ODS June, 2018 Ozone-depleting substances (ODS) deplete the stratospheric ozone layer when the chlorine and bromine atoms that they contain come into contact with ozone molecules. One chlorine atom can destroy over 100,000 ozone molecules before it is removed from the stratosphere. ODS that release chlorine include chlorofluorocarbons (CFCs), hydrochlorofluorocarbons (HCFCs), carbon tetrachloride, and methyl chloroform. ODS that release bromine include halons and methyl bromide. HTS code Ozone-Depleting Substance 2903.14.0000 Carbon tetrachloride What commodities 2903.19.6010 Methylchloroform (1,1,1-Trichloroethane) are ODS used in? 2903.39.1520 Methyl bromide 2903.71.0000 Chlorodifluoromethane (HCFC-22) ODS have been used 2903.72.0020 Dichlorotrifluoroethane (HCFC-123) for refrigeration, air 2903.73.0000 Dichlorofluoroethanes (HCFC-141, 141b) conditioning, insulation, 2903.74.0000 Chlorodifluoroethanes (HCFC-142, 142b) solvents, aerosol 2903.75.0000 Dichloropentafluoropropanes propellants, and in other (HCFC-225, 225ca, 225cb) sectors. Currently, the United 2903.76.0010 Bromotrifluoromethane (Halon 1301) States is in the 2903.76.0050 Bromochlorodifluoromethane (Halon 1211), process of phasing bromotrifluoromethane (Halon 1301), and out HCFCs, but in the dibromotetrafluoroethanes (Halon 2402), Other interim limited imports are 2903.77.0010 Trichlorofluoromethane (CFC-11) allowed. CFCs, 2903.77.0020 Trichlorotrifluoroethanes (CFC-113, CFC-113a) methyl bromide, halon, 2903.77.0030 -

List of Lists
United States Office of Solid Waste EPA 550-B-10-001 Environmental Protection and Emergency Response May 2010 Agency www.epa.gov/emergencies LIST OF LISTS Consolidated List of Chemicals Subject to the Emergency Planning and Community Right- To-Know Act (EPCRA), Comprehensive Environmental Response, Compensation and Liability Act (CERCLA) and Section 112(r) of the Clean Air Act • EPCRA Section 302 Extremely Hazardous Substances • CERCLA Hazardous Substances • EPCRA Section 313 Toxic Chemicals • CAA 112(r) Regulated Chemicals For Accidental Release Prevention Office of Emergency Management This page intentionally left blank. TABLE OF CONTENTS Page Introduction................................................................................................................................................ i List of Lists – Conslidated List of Chemicals (by CAS #) Subject to the Emergency Planning and Community Right-to-Know Act (EPCRA), Comprehensive Environmental Response, Compensation and Liability Act (CERCLA) and Section 112(r) of the Clean Air Act ................................................. 1 Appendix A: Alphabetical Listing of Consolidated List ..................................................................... A-1 Appendix B: Radionuclides Listed Under CERCLA .......................................................................... B-1 Appendix C: RCRA Waste Streams and Unlisted Hazardous Wastes................................................ C-1 This page intentionally left blank. LIST OF LISTS Consolidated List of Chemicals -

1 HUMAN HEALTH SAFETY EVALUATION of HALON REPLACEMENT CANDIDATES Darol E. Dodd1, Gary W. Jepson1, and Joseph A. Macko, Jr.2 1Air
HUMAN HEALTH SAFETY EVALUATION OF HALON REPLACEMENT CANDIDATES Darol E. Dodd1, Gary W. Jepson1, and Joseph A. Macko, Jr.2 1Air Force Research Laboratory/Operational Toxicology Branch (AFRL/HEST) – ManTech Environmental Technology, Inc. Wright-Patterson Air Force Base, Ohio 2Toxicology Directorate (MCHB-TS-T)/U.S. Army Center for Health Promotion & Preventive Medicine Aberdeen Proving Ground, Maryland Next-Generation Fire Suppression Technology Program (NGP) Project 3B/1/89 Final Report – Section II of II This research is part of the Department of Defense’s Next-Generation Fire Suppression Technology Program, funded by the DoD Strategic Environmental Research and Development Program (SERDP) 1 TABLE OF CONTENTS Page INTRODUCTION……………………………...………………………………………………….4 PHASE 1 – TOXICITY SCREENING METHODS……………………...……………………….5 Chemical/physical properties……………………………………………….…………….6 Existing toxicity literature…………………………………………………..…………….6 Preliminary “specific use” scenarios………………………………………..…………….7 Qualitative/quantitative structure activity relationships……………………..……………7 In vitro screens……………………………………………………………...…………….8 Acute irritation tests………………………………………………………………………9 Acute toxicity tests…………………………………………………………..…………..10 The “limit test”…………………………………………………………………..11 The “specific use” scenario test……………………………………..…………..12 Genotoxicity tests (Phase 1)………………………………………………..……………13 DECISION POINT 1……………………………………………………………………14 Concentration x time (C x T) relationships……………………….……………18 Stop or continue testing…………………………………………..…………….18 PHASE 2 – TOXICITY -

Federal Register/Vol. 84, No. 230/Friday, November 29, 2019
Federal Register / Vol. 84, No. 230 / Friday, November 29, 2019 / Proposed Rules 65739 are operated by a government LIBRARY OF CONGRESS 49966 (Sept. 24, 2019). The Office overseeing a population below 50,000. solicited public comments on a broad Of the impacts we estimate accruing U.S. Copyright Office range of subjects concerning the to grantees or eligible entities, all are administration of the new blanket voluntary and related mostly to an 37 CFR Part 210 compulsory license for digital uses of increase in the number of applications [Docket No. 2019–5] musical works that was created by the prepared and submitted annually for MMA, including regulations regarding competitive grant competitions. Music Modernization Act Implementing notices of license, notices of nonblanket Therefore, we do not believe that the Regulations for the Blanket License for activity, usage reports and adjustments, proposed priorities would significantly Digital Uses and Mechanical Licensing information to be included in the impact small entities beyond the Collective: Extension of Comment mechanical licensing collective’s potential for increasing the likelihood of Period database, database usability, their applying for, and receiving, interoperability, and usage restrictions, competitive grants from the Department. AGENCY: U.S. Copyright Office, Library and the handling of confidential of Congress. information. Paperwork Reduction Act ACTION: Notification of inquiry; To ensure that members of the public The proposed priorities do not extension of comment period. have sufficient time to respond, and to contain any information collection ensure that the Office has the benefit of SUMMARY: The U.S. Copyright Office is requirements. a complete record, the Office is extending the deadline for the extending the deadline for the Intergovernmental Review: This submission of written reply comments program is subject to Executive Order submission of written reply comments in response to its September 24, 2019 to no later than 5:00 p.m.