New-Drug-Classificat
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Clinical Study Is Nonmicronized Diosmin 600Mg As Effective As
Hindawi International Journal of Vascular Medicine Volume 2020, Article ID 4237204, 9 pages https://doi.org/10.1155/2020/4237204 Clinical Study Is Nonmicronized Diosmin 600mg as Effective as Micronized Diosmin 900mg plus Hesperidin 100mg on Chronic Venous Disease Symptoms? Results of a Noninferiority Study Marcio Steinbruch,1 Carlos Nunes,2 Romualdo Gama,3 Renato Kaufman,4 Gustavo Gama,5 Mendel Suchmacher Neto,6 Rafael Nigri,7 Natasha Cytrynbaum,8 Lisa Brauer Oliveira,9 Isabelle Bertaina,10 François Verrière,10 and Mauro Geller 3,6,9 1Hospital Albert Einstein (São Paulo-Brasil), R. Mauricio F Klabin 357/17, Vila Mariana, SP, Brazil 04120-020 2Instituto de Pós-Graduação Médica Carlos Chagas-Fundação Educacional Serra dos Órgãos-UNIFESO (Rio de Janeiro/Teresópolis- Brasil), Av. Alberto Torres 111, Teresópolis, RJ, Brazil 25964-004 3Fundação Educacional Serra dos Órgãos-UNIFESO (Teresópolis-Brasil), Av. Alberto Torres 111, Teresópolis, RJ, Brazil 25964-004 4Faculdade de Ciências Médicas, Universidade Estadual do Rio de Janeiro (UERJ) (Rio de Janeiro-Brazil), Av. N. Sra. De Copacapana, 664/206, Rio de Janeiro, RJ, Brazil 22050-903 5Fundação Educacional Serra dos Órgãos-UNIFESO (Teresópolis-Brasil), Rua Prefeito Sebastião Teixeira 400/504-1, Rio de Janeiro, RJ, Brazil 25953-200 6Instituto de Pós-Graduação Médica Carlos Chagas (Rio de Janeiro-Brazil), R. General Canabarro 68/902, Rio de Janeiro, RJ, Brazil 20271-200 7Department of Medicine, Rutgers New Jersey Medical School-USA, 185 S Orange Ave., Newark, NJ 07103, USA 8Hospital Universitário Pedro Ernesto, Universidade Estadual do Rio de Janeiro (UERJ) (Rio de Janeiro-Brazil), R. Hilário de Gouveia, 87/801, Rio de Janeiro, RJ, Brazil 22040-020 9Universidade Federal do Rio de Janeiro (UFRJ) (Rio de Janeiro-Brazil), Av. -
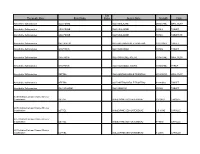
Therapeutic Class Brand Name P a Status Generic
P A Therapeutic Class Brand Name Status Generic Name Strength Form Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 250MG/5ML ORAL SUSP Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 500MG TABLET Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 500MG TABLET DR Absorbable Sulfonamides BACTRIM DS SULFAMETHOXAZOLE/TRIMETHO 800-160MG TABLET Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE 500MG TABLET Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE ACETYL 500MG/5ML ORAL SUSP Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE ACETYL 500MG/5ML SYRUP Absorbable Sulfonamides SEPTRA SULFAMETHOXAZOLE/TRIMETHO 200-40MG/5 ORAL SUSP Absorbable Sulfonamides SEPTRA SULFAMETHOXAZOLE/TRIMETHO 400-80MG TABLET Absorbable Sulfonamides SULFADIAZINE SULFADIAZINE 500MG TABLET ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 10-20MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 2.5-10MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-10MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-20MG CAPSULE P A Therapeutic Class Brand Name Status Generic Name Strength Form ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-40MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 10-40MG CAPSULE Acne Agents, Systemic ACCUTANE ISOTRETINOIN 10MG CAPSULE Acne Agents, Systemic ACCUTANE ISOTRETINOIN 20MG CAPSULE Acne Agents, Systemic ACCUTANE -

(12) United States Patent (10) Patent N0.: US 8,343,962 B2 Kisak Et Al
US008343962B2 (12) United States Patent (10) Patent N0.: US 8,343,962 B2 Kisak et al. (45) Date of Patent: *Jan. 1, 2013 (54) TOPICAL FORMULATION (58) Field of Classi?cation Search ............. .. 514/226.5, 514/334, 420, 557, 567 (75) Inventors: Edward T. Kisak, San Diego, CA (US); See application ?le fOr Complete Search history. John M. NeWsam, La Jolla, CA (US); _ Dominic King-Smith, San Diego, CA (56) References C‘ted (US); Pankaj Karande, Troy, NY (US); Samir Mitragotri, Goleta, CA (US) US' PATENT DOCUMENTS 5,602,183 A 2/1997 Martin et al. (73) Assignee: NuvoResearchOntano (CA) Inc., Mississagua, 6,328,979 2B1 12/2001 Yamashita et a1. 7,001,592 B1 2/2006 Traynor et a1. ( * ) Notice: Subject to any disclaimer, the term of this 7,795,309 B2 9/2010 Kisak eta1~ patent is extended or adjusted under 35 2002/0064524 A1 5/2002 Cevc U.S.C. 154(b) by 212 days. FOREIGN PATENT DOCUMENTS This patent is subject to a terminal dis- W0 WO 2005/009510 2/2005 claimer- OTHER PUBLICATIONS (21) APPI' NO‘, 12/848,792 International Search Report issued on Aug. 8, 2008 in application No. PCT/lB2007/0l983 (corresponding to US 7,795,309). _ Notice ofAlloWance issued on Apr. 29, 2010 by the Examiner in US. (22) Med Aug- 2’ 2010 Appl. No. 12/281,561 (US 7,795,309). _ _ _ Of?ce Action issued on Dec. 30, 2009 by the Examiner in US. Appl. (65) Prior Publication Data No, 12/281,561 (Us 7,795,309), Us 2011/0028460 A1 Feb‘ 3’ 2011 Primary Examiner * Raymond Henley, 111 Related U 5 Application Data (74) Attorney, Agent, or Firm * Foley & Lardner LLP (63) Continuation-in-part of application No. -

Appendix on Tariff Elimination Schedule for Mercosur
Trade part of the EU-Mercosur Association Agreement Without Prejudice Disclaimer: In view of the Commission's transparency policy, the Commission is publishing the texts of the Trade Part of the Agreement following the agreement in principle announced on 28 June 2019. The texts are published for information purposes only and may undergo further modifications including as a result of the process of legal revision. However, in view of the growing public interest in the negotiations, the texts are published at this stage of the negotiations for information purposes. These texts are without prejudice to the final outcome of the agreement between the EU and Mercosur. The texts will be final upon signature. The agreement will become binding on the Parties under international law only after completion by each Party of its internal legal procedures necessary for the entry into force of the Agreement (or its provisional application). AR applied BR applied PY applied UY applied Mercosur Final NCM Description Comments tariff tariff tariff tariff Offer 01012100 Pure-bred horses 0 0 0 0 0 01012900 Lives horses, except pure-bred breeding 2 2 2 2 0 01013000 Asses, pure-bred breeding 4 4 4 4 4 01019000 Asses, except pure-bred breeding 4 4 4 4 4 01022110 Purebred breeding cattle, pregnant or lactating 0 0 0 0 0 01022190 Other pure-bred cattle, for breeding 0 0 0 0 0 01022911 Other bovine animals for breeding,pregnant or lactating 2 2 2 2 0 01022919 Other bovine animals for breeding 2 2 2 2 4 01022990 Other live catlle 2 2 2 2 0 01023110 Pure-bred breeding buffalo, pregnant or lactating 0 0 0 0 0 01023190 Other pure-bred breeding buffalo 0 0 0 0 0 01023911 Other buffalo for breeding, ex. -

2D6 Substrates 2D6 Inhibitors 2D6 Inducers
Physician Guidelines: Drugs Metabolized by Cytochrome P450’s 1 2D6 Substrates Acetaminophen Captopril Dextroamphetamine Fluphenazine Methoxyphenamine Paroxetine Tacrine Ajmaline Carteolol Dextromethorphan Fluvoxamine Metoclopramide Perhexiline Tamoxifen Alprenolol Carvedilol Diazinon Galantamine Metoprolol Perphenazine Tamsulosin Amiflamine Cevimeline Dihydrocodeine Guanoxan Mexiletine Phenacetin Thioridazine Amitriptyline Chloropromazine Diltiazem Haloperidol Mianserin Phenformin Timolol Amphetamine Chlorpheniramine Diprafenone Hydrocodone Minaprine Procainamide Tolterodine Amprenavir Chlorpyrifos Dolasetron Ibogaine Mirtazapine Promethazine Tradodone Aprindine Cinnarizine Donepezil Iloperidone Nefazodone Propafenone Tramadol Aripiprazole Citalopram Doxepin Imipramine Nifedipine Propranolol Trimipramine Atomoxetine Clomipramine Encainide Indoramin Nisoldipine Quanoxan Tropisetron Benztropine Clozapine Ethylmorphine Lidocaine Norcodeine Quetiapine Venlafaxine Bisoprolol Codeine Ezlopitant Loratidine Nortriptyline Ranitidine Verapamil Brofaramine Debrisoquine Flecainide Maprotline olanzapine Remoxipride Zotepine Bufuralol Delavirdine Flunarizine Mequitazine Ondansetron Risperidone Zuclopenthixol Bunitrolol Desipramine Fluoxetine Methadone Oxycodone Sertraline Butylamphetamine Dexfenfluramine Fluperlapine Methamphetamine Parathion Sparteine 2D6 Inhibitors Ajmaline Chlorpromazine Diphenhydramine Indinavir Mibefradil Pimozide Terfenadine Amiodarone Cimetidine Doxorubicin Lasoprazole Moclobemide Quinidine Thioridazine Amitriptyline Cisapride -

Muscarinic Acetylcholine Receptor
mAChR Muscarinic acetylcholine receptor mAChRs (muscarinic acetylcholine receptors) are acetylcholine receptors that form G protein-receptor complexes in the cell membranes of certainneurons and other cells. They play several roles, including acting as the main end-receptor stimulated by acetylcholine released from postganglionic fibersin the parasympathetic nervous system. mAChRs are named as such because they are more sensitive to muscarine than to nicotine. Their counterparts are nicotinic acetylcholine receptors (nAChRs), receptor ion channels that are also important in the autonomic nervous system. Many drugs and other substances (for example pilocarpineand scopolamine) manipulate these two distinct receptors by acting as selective agonists or antagonists. Acetylcholine (ACh) is a neurotransmitter found extensively in the brain and the autonomic ganglia. www.MedChemExpress.com 1 mAChR Inhibitors & Modulators (+)-Cevimeline hydrochloride hemihydrate (-)-Cevimeline hydrochloride hemihydrate Cat. No.: HY-76772A Cat. No.: HY-76772B Bioactivity: Cevimeline hydrochloride hemihydrate, a novel muscarinic Bioactivity: Cevimeline hydrochloride hemihydrate, a novel muscarinic receptor agonist, is a candidate therapeutic drug for receptor agonist, is a candidate therapeutic drug for xerostomia in Sjogren's syndrome. IC50 value: Target: mAChR xerostomia in Sjogren's syndrome. IC50 value: Target: mAChR The general pharmacol. properties of this drug on the The general pharmacol. properties of this drug on the gastrointestinal, urinary, and reproductive systems and other… gastrointestinal, urinary, and reproductive systems and other… Purity: >98% Purity: >98% Clinical Data: No Development Reported Clinical Data: No Development Reported Size: 10mM x 1mL in DMSO, Size: 10mM x 1mL in DMSO, 1 mg, 5 mg 1 mg, 5 mg AC260584 Aclidinium Bromide Cat. No.: HY-100336 (LAS 34273; LAS-W 330) Cat. -

The In¯Uence of Medication on Erectile Function
International Journal of Impotence Research (1997) 9, 17±26 ß 1997 Stockton Press All rights reserved 0955-9930/97 $12.00 The in¯uence of medication on erectile function W Meinhardt1, RF Kropman2, P Vermeij3, AAB Lycklama aÁ Nijeholt4 and J Zwartendijk4 1Department of Urology, Netherlands Cancer Institute/Antoni van Leeuwenhoek Hospital, Plesmanlaan 121, 1066 CX Amsterdam, The Netherlands; 2Department of Urology, Leyenburg Hospital, Leyweg 275, 2545 CH The Hague, The Netherlands; 3Pharmacy; and 4Department of Urology, Leiden University Hospital, P.O. Box 9600, 2300 RC Leiden, The Netherlands Keywords: impotence; side-effect; antipsychotic; antihypertensive; physiology; erectile function Introduction stopped their antihypertensive treatment over a ®ve year period, because of side-effects on sexual function.5 In the drug registration procedures sexual Several physiological mechanisms are involved in function is not a major issue. This means that erectile function. A negative in¯uence of prescrip- knowledge of the problem is mainly dependent on tion-drugs on these mechanisms will not always case reports and the lists from side effect registries.6±8 come to the attention of the clinician, whereas a Another way of looking at the problem is drug causing priapism will rarely escape the atten- combining available data on mechanisms of action tion. of drugs with the knowledge of the physiological When erectile function is in¯uenced in a negative mechanisms involved in erectile function. The way compensation may occur. For example, age- advantage of this approach is that remedies may related penile sensory disorders may be compen- evolve from it. sated for by extra stimulation.1 Diminished in¯ux of In this paper we will discuss the subject in the blood will lead to a slower onset of the erection, but following order: may be accepted. -

Classification of Medicinal Drugs and Driving: Co-Ordination and Synthesis Report
Project No. TREN-05-FP6TR-S07.61320-518404-DRUID DRUID Driving under the Influence of Drugs, Alcohol and Medicines Integrated Project 1.6. Sustainable Development, Global Change and Ecosystem 1.6.2: Sustainable Surface Transport 6th Framework Programme Deliverable 4.4.1 Classification of medicinal drugs and driving: Co-ordination and synthesis report. Due date of deliverable: 21.07.2011 Actual submission date: 21.07.2011 Revision date: 21.07.2011 Start date of project: 15.10.2006 Duration: 48 months Organisation name of lead contractor for this deliverable: UVA Revision 0.0 Project co-funded by the European Commission within the Sixth Framework Programme (2002-2006) Dissemination Level PU Public PP Restricted to other programme participants (including the Commission x Services) RE Restricted to a group specified by the consortium (including the Commission Services) CO Confidential, only for members of the consortium (including the Commission Services) DRUID 6th Framework Programme Deliverable D.4.4.1 Classification of medicinal drugs and driving: Co-ordination and synthesis report. Page 1 of 243 Classification of medicinal drugs and driving: Co-ordination and synthesis report. Authors Trinidad Gómez-Talegón, Inmaculada Fierro, M. Carmen Del Río, F. Javier Álvarez (UVa, University of Valladolid, Spain) Partners - Silvia Ravera, Susana Monteiro, Han de Gier (RUGPha, University of Groningen, the Netherlands) - Gertrude Van der Linden, Sara-Ann Legrand, Kristof Pil, Alain Verstraete (UGent, Ghent University, Belgium) - Michel Mallaret, Charles Mercier-Guyon, Isabelle Mercier-Guyon (UGren, University of Grenoble, Centre Regional de Pharmacovigilance, France) - Katerina Touliou (CERT-HIT, Centre for Research and Technology Hellas, Greece) - Michael Hei βing (BASt, Bundesanstalt für Straßenwesen, Germany). -

Thoroughbred Racing
178CSR1 Title 178 Legislative Rule West Virginia Racing Commission Series 1 Thoroughbred Racing Effective: July 9, 2014 West Virginia Racing Commission 900 Pennsylvania Avenue Suite 533 Charleston WV 25302 305.558.2150 Fax 304.558.6319 Web Site: www.racing.wv.gov 178CSR1 Table of Contents SERIES 1 THOROUGHBRED RACING ____________________________________________________________________ 1 §178-1-1. General. ____________________________________________________________________________________ 1 PART 1. DEFINITIONS. _________________________________________________________________________________ 1 §178-1-2. Definitions. _________________________________________________________________________________ 1 PART 2. GENERAL AUTHORITY. ________________________________________________________________________ 9 §178-1-3. General Authority of the Racing Commission. ______________________________________________________ 9 §178-1-4. Power Of Entry. ______________________________________________________________________________ 9 §178-1-5. Racing Commission personnel. __________________________________________________________________ 9 §178-1-6. Ejection/Exclusion. __________________________________________________________________________ 12 PART 3. RACING OFFICIALS. __________________________________________________________________________ 12 §178-1-7. General Provisions. __________________________________________________________________________ 12 §178-1-8. Stewards. __________________________________________________________________________________ 14 -

)&F1y3x PHARMACEUTICAL APPENDIX to THE
)&f1y3X PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE )&f1y3X PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 3 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABAMECTIN 65195-55-3 ACTODIGIN 36983-69-4 ABANOQUIL 90402-40-7 ADAFENOXATE 82168-26-1 ABCIXIMAB 143653-53-6 ADAMEXINE 54785-02-3 ABECARNIL 111841-85-1 ADAPALENE 106685-40-9 ABITESARTAN 137882-98-5 ADAPROLOL 101479-70-3 ABLUKAST 96566-25-5 ADATANSERIN 127266-56-2 ABUNIDAZOLE 91017-58-2 ADEFOVIR 106941-25-7 ACADESINE 2627-69-2 ADELMIDROL 1675-66-7 ACAMPROSATE 77337-76-9 ADEMETIONINE 17176-17-9 ACAPRAZINE 55485-20-6 ADENOSINE PHOSPHATE 61-19-8 ACARBOSE 56180-94-0 ADIBENDAN 100510-33-6 ACEBROCHOL 514-50-1 ADICILLIN 525-94-0 ACEBURIC ACID 26976-72-7 ADIMOLOL 78459-19-5 ACEBUTOLOL 37517-30-9 ADINAZOLAM 37115-32-5 ACECAINIDE 32795-44-1 ADIPHENINE 64-95-9 ACECARBROMAL 77-66-7 ADIPIODONE 606-17-7 ACECLIDINE 827-61-2 ADITEREN 56066-19-4 ACECLOFENAC 89796-99-6 ADITOPRIM 56066-63-8 ACEDAPSONE 77-46-3 ADOSOPINE 88124-26-9 ACEDIASULFONE SODIUM 127-60-6 ADOZELESIN 110314-48-2 ACEDOBEN 556-08-1 ADRAFINIL 63547-13-7 ACEFLURANOL 80595-73-9 ADRENALONE -

Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017
Q UO N T FA R U T A F E BERMUDA PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 BR 111 / 2017 The Minister responsible for health, in exercise of the power conferred by section 48A(1) of the Pharmacy and Poisons Act 1979, makes the following Order: Citation 1 This Order may be cited as the Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017. Repeals and replaces the Third and Fourth Schedule of the Pharmacy and Poisons Act 1979 2 The Third and Fourth Schedules to the Pharmacy and Poisons Act 1979 are repealed and replaced with— “THIRD SCHEDULE (Sections 25(6); 27(1))) DRUGS OBTAINABLE ONLY ON PRESCRIPTION EXCEPT WHERE SPECIFIED IN THE FOURTH SCHEDULE (PART I AND PART II) Note: The following annotations used in this Schedule have the following meanings: md (maximum dose) i.e. the maximum quantity of the substance contained in the amount of a medicinal product which is recommended to be taken or administered at any one time. 1 PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 mdd (maximum daily dose) i.e. the maximum quantity of the substance that is contained in the amount of a medicinal product which is recommended to be taken or administered in any period of 24 hours. mg milligram ms (maximum strength) i.e. either or, if so specified, both of the following: (a) the maximum quantity of the substance by weight or volume that is contained in the dosage unit of a medicinal product; or (b) the maximum percentage of the substance contained in a medicinal product calculated in terms of w/w, w/v, v/w, or v/v, as appropriate. -

Non-Pharmacologic Therapies and Airway Clearance Techniques in Bronchiectasis
Division of Pulmonary, Critical Care and Sleep Medicine NON-PHARMACOLOGIC THERAPIES AND AIRWAY CLEARANCE TECHNIQUES IN BRONCHIECTASIS Ashwin Basavaraj, MD, FCCP Associate Director, NYU Bronchiectasis Program NTM Patient Education Program DC 11/24/2019 October 30, 2019 Financial Disclosure • Insmed - Consultant, Advisory Board (Active) • Hill-Rom – Consultant, Principal investigator on a clinical trial (Active) • COPD foundation grant on airway clearance 2 Division of Pulmonary, Critical Care and Sleep Medicine DC 11/24/2019 Case presentation • 66 year-old female with a history of prior pneumonia 15 years ago presents with productive cough. • She has mild shortness of breath. No fevers, no hemoptysis. She has gained two pounds over the year. • No other prior medical history, and currently not taking any medications • Initial workup including autoimmune serologies and quantitative immunoglobulin levels were negative. • You check AFB, bacterial and fungal sputum cultures. • She has 2 out 3 cultures positive for MAC. 3 Division of Pulmonary, Critical Care and Sleep Medicine DC 11/24/2019 4 Division of Pulmonary, Critical Care and Sleep Medicine DC 11/24/2019 What’s the next best step in management? A) Start 3 drug antibiotic therapy for MAC B) Initiate airway clearance with nebulized hypertonic saline and a positive expiratory pressure device C) Start antibiotics for MAC and initiate airway clearance D) Closely monitor without initiation of treatment 5 Division of Pulmonary, Critical Care and Sleep Medicine DC 11/24/2019 GOALS OF AIRWAY CLEARANCE Short term goals Long term goals • Provide more effective sputum • Reduce further airway damage by clearance that improves ventilation halting the vicious cycle • Reduce cough and breathlessness • Reduce pulmonary exacerbations • Improve quality of life O’Neill, et al.