Chemical Inventory Packet / Calarp Lists / HFD / Dmg 2004 Table 1
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Transport of Dangerous Goods
ST/SG/AC.10/1/Rev.16 (Vol.I) Recommendations on the TRANSPORT OF DANGEROUS GOODS Model Regulations Volume I Sixteenth revised edition UNITED NATIONS New York and Geneva, 2009 NOTE The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. ST/SG/AC.10/1/Rev.16 (Vol.I) Copyright © United Nations, 2009 All rights reserved. No part of this publication may, for sales purposes, be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, electrostatic, magnetic tape, mechanical, photocopying or otherwise, without prior permission in writing from the United Nations. UNITED NATIONS Sales No. E.09.VIII.2 ISBN 978-92-1-139136-7 (complete set of two volumes) ISSN 1014-5753 Volumes I and II not to be sold separately FOREWORD The Recommendations on the Transport of Dangerous Goods are addressed to governments and to the international organizations concerned with safety in the transport of dangerous goods. The first version, prepared by the United Nations Economic and Social Council's Committee of Experts on the Transport of Dangerous Goods, was published in 1956 (ST/ECA/43-E/CN.2/170). In response to developments in technology and the changing needs of users, they have been regularly amended and updated at succeeding sessions of the Committee of Experts pursuant to Resolution 645 G (XXIII) of 26 April 1957 of the Economic and Social Council and subsequent resolutions. -

"U/^ ((*.U \ BIBLIOTHEEK STARINGGEBOUW Volatilization of Tri-Allate, Ethoprophos and Parathion Measured with Four Methods After Spraying on a Sandy Soil
"u/^ ((*.u \ BIBLIOTHEEK STARINGGEBOUW Volatilization of tri-allate, ethoprophos and parathion measured with four methods after spraying on a sandy soil G. Bor F. van den Berg J.H. Smelt R.A. Smidt A.E. van de Peppel-Groen M. Leistra Report 104 DLO Winand Staring Centre, Wageningen (The Netherlands), 1995 1 h FEB. 1998 0000' ABSTRACT Bor, G., F. van den Berg, J.H. Smelt, R.A. Smidt, A.E. van de Peppel-Groen, M. Leistra, 1995. Volatilization of tri-allate, ethoprophos and parathion measured withfour methods after spraying on a sandy soil. Wageningen (The Netherlands), DLO Winand Staring Centre. Report 104. 62 pp.; 9 Figs; 6 Tables; 14 Refs; 3 Annex. At about eleven times after application of tri-allate, ethoprophos and parathion to a sandy soil, their rates of volatilization were determined with the aerodynamic method (AD),th e Bowen-ratio method (BR), the theoretical-profile method (TP) and the Box method (B). The volatilization was highest for tri-allate and lowest for parathion. On the first day after application, the volatilization rate decreased sharply,bu tthereafte r the decreasewa smor egradual .Th edifference s involatilizatio n rate asdetermine d withth eAD ,B R andT Pmethod s werecomparativel y small.Th erate sdetermine d with the Box method were mostly lower than those determined with the other methods. Keywords: aerodynamic method, air quality, air sampling, Bowen ratio method, Box method, field experiment, gas chromatography, pesticide, polystyrene, theoretical-profile method, XAD ISSN 0927-4537 ©1995 DLO Winand Staring Centre for Integrated Land, Soil and Water Research (SC-DLO) P.O. -

Calarp) Program
California Accidental Release Prevention (CalARP) Program Administering Agency Guidance January 31, 2005 Preface This document provides general guidance to help Administering Agencies (AAs) implement and enforce the California Accidental Release Prevention (CalARP) Program. The intent is to identify the elements of the Program applicable to each regulated business, and assist AAs with oversight of the CalARP Program statutes and regulations. This document is not a substitute for the CalARP Program regulations; it does not impose legally binding requirements. About This Document This document follows the format of the California Code of Regulations, Title 19, Division 2, Chapter 4.5: California Accidental Release Prevention (CalARP) Program. The regulatory sections are presented in parentheses for ease of reference. Acknowledgements The California Emergency Management Agency (Cal EMA) would like to thank the following people for their valuable assistance in the preparation of this document: Howard Wines, Hazardous Materials Specialist, City of Bakersfield Fire Department Robert Distaso P.E., Fire Safety Engineer, Orange County Fire Authority Randall L. Sawyer, Supervisor, Accidental Release Prevention Programs, Contra Costa County Health Services Department Beronia Beniamine, Senior Hazardous Materials Specialist, Stanislaus County Environmental Resources Department Angie Proboszcz, Risk Management Program Coordinator, USEPA Region 9 Jon Christenson, Senior Environmental Health Specialist, Merced County Department of Public Health Teresa -

Phytotoxicity of Some Organophosphate Insecticides to Oughly with Air Dry, Sieved Soil, to Pro Duce Rates of 37.5, 75 and ISO Mg Aj
Plant Protection Quarterly VoI.7(1} 1992 23 -------------------------------- ties of each pesticide were shaken thor Phytotoxicity of some organophosphate insecticides to oughly with air dry, sieved soil, to pro duce rates of 37.5, 75 and ISO mg aj. L-' onions and carrots during germination and emergence soil. Each pot constituted one plot. If it is assumed that in a field situation, P.J. Sinclair, New South Wales Agriculture, Horticultural Research and band-in-furrow treatment would treat a Advisory Station, Griffith, New South Wales 2680, Australia. strip 50 mm wide by 20 mm deep, the rates tested would correspond to 37.5, 75 R.J. Neeson and P.A. Williams, New South Wales Agriculture, Agricultural and 150 mg a.i. m" row, or 0.5,1.0 and 2.0 institute, Yanco, New South Wales 2703, Australia. kg a.i. ha" at a row spacing of 75 cm. The low and medium rates are then compara Summary ble to ra tes used in the field by Getzin The phytotoxicity of some commonly phytotoxicity from carbofuran applied to (1973), Thompson et al. (1981) and used insecticides to onions (Allium cepa) onions as a seed dressing. Chlorpyrifos is Goodyer et al. (1989) . and carrots (Daucus carota) during es generally non-phytotoxic at recom Daily counts of emerged seedlings were tablishment was assessed in pot trials. mended rates and methods of application, made at 7 to 18 days and at 21 days after Terbufos, ethoprophos, phoxirn and but some crops are especially sensitive to first watering. These data were used to de carbofuran (all 10% a.L granular fonnu it during the seedling stage or if the termine the total number of seedlings lations) and chlorpyrifos (25% a.i. -

1,45%,562 UNITED SATES P All.‘ Bl If" Til?
Patented May 1, 1923. 1,45%,562 UNITED SATES P All.‘ bl if" til? . ROBERT E. WILSON, LEON ‘JV. PARSONS, AND STANLEY 1E. OHXSHOLIYI, OF WASHINGTON, DISTRICT OF COLUMBIA. PROCESS THE PRODUCTION OE‘ All‘HALLEAETELMLETAL PERTJIANGANATES. N0 Drawing". Application ?led September 27, 1918. Serial No. 255,975. To (all to]: am it may concern. .' manganate by oxidation or acidification, Be it known that we, Romain‘ E. lllinsou, metatheses into calcium pern'iangamite by LEON “7. Parsons, and STANLEY L. Unis treatment With calcium sulphate or milk at HoLM, citizens of the United States. and sta lime. tioned at ViTashington, District of Columbia, O'li' these four possible methods, (1.) is not 60 in the o?icc of the Director the Chemical a possible large scale method. on account l/Varfare Service, Research Division, have in of its use ot silver; (2) and are elec vented a Process for the ll’roduction oif Al trolytic methods Without a. great deal out kali-Earth-l\letal Permanpjanates, of which promise, and are to be considered elsewhere; ll) the ‘following is a speci?cation. (ll) the principal subject of this applica G3 in The present invention relates to the pro tion. duction oi? alkah» earth metal permangam Three distinct methods for preparing: ba~ nates and especially the permanganates of rium (or strontium) manganate have been calcium and magnesium as these have beenv here investigated. The ?rst of? these meth found to be very ellicient oxidizing agents ods involves heating together barium perox 70 for certain purposes, more e?icient even. than ide, hydroxide, or a salt, such as the nitrate the permanganates of the allmliearth metals. -

Assessment of Portable HAZMAT Sensors for First Responders
The author(s) shown below used Federal funds provided by the U.S. Department of Justice and prepared the following final report: Document Title: Assessment of Portable HAZMAT Sensors for First Responders Author(s): Chad Huffman, Ph.D., Lars Ericson, Ph.D. Document No.: 246708 Date Received: May 2014 Award Number: 2010-IJ-CX-K024 This report has not been published by the U.S. Department of Justice. To provide better customer service, NCJRS has made this Federally- funded grant report available electronically. Opinions or points of view expressed are those of the author(s) and do not necessarily reflect the official position or policies of the U.S. Department of Justice. Assessment of Portable HAZMAT Sensors for First Responders DOJ Office of Justice Programs National Institute of Justice Sensor, Surveillance, and Biometric Technologies (SSBT) Center of Excellence (CoE) March 1, 2012 Submitted by ManTech Advanced Systems International 1000 Technology Drive, Suite 3310 Fairmont, West Virginia 26554 Telephone: (304) 368-4120 Fax: (304) 366-8096 Dr. Chad Huffman, Senior Scientist Dr. Lars Ericson, Director UNCLASSIFIED This project was supported by Award No. 2010-IJ-CX-K024, awarded by the National Institute of Justice, Office of Justice Programs, U.S. Department of Justice. The opinions, findings, and conclusions or recommendations expressed in this publication are those of the author(s) and do not necessarily reflect those of the Department of Justice. This document is a research report submitted to the U.S. Department of Justice. This report has not been published by the Department. Opinions or points of view expressed are those of the author(s) and do not necessarily reflect the official position or policies of the U.S. -

Safety Practices in Chemistry Laboratories
4/24/2012 Safety Practices in Chemistry Laboratory Sam Tung, HSE Specialist, CIH, RSO Health Safety and Environment Office, Hong Kong University of Science and Technology Contents 1. Introduction to Legislations related to Chemistry Laboratories 2. Introduction to Chemical Hazards 3. Criteria of Design for Laboratory Ventilation 4. Operations and Maintenance of Laboratory Fume hood 5. Case Studies of Laboratory Accidents 6. Handling of Chemical Spills 7. Emergency Response Equipment 1 4/24/2012 Importance of Health and Safety To protect your health and safety (and environment) To protect your colleagues’ and students’ health and safety Case Study: Laboratory Fire Kills UCLA Researcher A 23-year-old research assistant working at UCLA who was seriously burned in a lab fire in December 2008 recently died from her injuries. She was trying to transfer up to 2 ounces (~50ml) of t-butyl lithium (pyrophoric chemical), which was dissolved in pentane from one sealed container to another by a 50 ml syringe. The barrel of the syringe was either ejected or pulled out of the syringe, causing liquid to be released. A flash fire set her clothing ablaze and spread second- and third-degree burns over 43% of her body. 2 4/24/2012 Root Causes of the Accident Poor technique and improper method Use a 50ml syringe to transfer~50 ml pyrophoric chemical Should use a 100ml syringe Should use Cannula Method for transfer of pyrophoric chemical > 50 ml Lack of proper training No safety training record Lack of supervision No follow up actions had been -

The Enthalpy of Formation of Organic Compounds with “Chemical Accuracy”
chemengineering Article Group Contribution Revisited: The Enthalpy of Formation of Organic Compounds with “Chemical Accuracy” Robert J. Meier Pro-Deo Consultant, 52525 Heinsberg, North-Rhine Westphalia, Germany; [email protected] Abstract: Group contribution (GC) methods to predict thermochemical properties are of eminent importance to process design. Compared to previous works, we present an improved group contri- bution parametrization for the heat of formation of organic molecules exhibiting chemical accuracy, i.e., a maximum 1 kcal/mol (4.2 kJ/mol) difference between the experiment and model, while, at the same time, minimizing the number of parameters. The latter is extremely important as too many parameters lead to overfitting and, therewith, to more or less serious incorrect predictions for molecules that were not within the data set used for parametrization. Moreover, it was found to be important to explicitly account for common chemical knowledge, e.g., geminal effects or ring strain. The group-related parameters were determined step-wise: first, alkanes only, and then only one additional group in the next class of molecules. This ensures unique and optimal parameter values for each chemical group. All data will be made available, enabling other researchers to extend the set to other classes of molecules. Keywords: enthalpy of formation; thermodynamics; molecular modeling; group contribution method; quantum mechanical method; chemical accuracy; process design Citation: Meier, R.J. Group Contribution Revisited: The Enthalpy of Formation of Organic Compounds with “Chemical Accuracy”. 1. Introduction ChemEngineering 2021, 5, 24. To understand chemical reactivity and/or chemical equilibria, knowledge of thermo- o https://doi.org/10.3390/ dynamic properties such as gas-phase standard enthalpy of formation DfH gas is a necessity. -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -
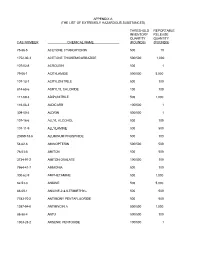
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

Hazardous Material Inventory Statement
City of Brooklyn Park FIRE DEPARTMENT 5200 - 85th Avenue North Brooklyn Park MN 55443 Phone: (763)493-8020 Fax: (763) 493-8391 Hazardous Materials Inventory Statement Users Guide A separate inventory statement shall be provided for each building. An amended inventory statement shall be provided within 30 days of the storage of any hazardous materials or plastics that changes or adds a hazard class or which is sufficient in quantity to cause an increase in the quantity which exceeds 5 percent for any hazard class. The hazardous materials inventory statement shall list by hazard class categories. Each grouping shall provide the following information for each hazardous material listed for that group including a total quantity for each group of hazard class. 1. Hazard class. (See attached Hazardous Materials Categories Listing) 2. Common or trade name. 3. Chemical Abstract Service Number (CAS number) found in 29 Code of Federal Regulations (C.F.R.). 4. Whether the material is pure or a mixture, and whether the material is a solid, liquid or gas 5. Maximum aggregate quantity stored at any one time. 6. Maximum aggregate quantity In-Use (Open to atmosphere) at any one time. 7. Maximum aggregate quantity In-Use (Closed to atmosphere) at any one time. 8. Storage conditions related to the storage type, high-pile, encapsulated, non-encapsulated. Attached is a listing of categories that all materials need to be organized to. Definitions of these categories are also attached for your use. At the end of this packet are blank forms for completing this project. For questions regarding Hazardous Materials Inventory Statement contact the Fire Department at 763-493-8020. -

United States Patent O Fice Patented Dec
2,816,144 United States Patent O fice Patented Dec. 10, 1957 2. conventional manner and the hydrolysis mass is heated until completely free of any side chain chlorine deriva 2,816,144 tives. This can be determined by the use of alcoholic silver nitrate, for example, which will form a cloudy pre PROBEDUCTION OF RBENZALDEHYDE cipitate upon contact with compounds containing a side Robert W. Harris, Silsborough Township, Somerset chain chlorine group. County, N. S. The benzaldehyde which is obtained upon hydrolysis Separates from the aqueous layer and upon washing and No Drawing. Application August 4, 1955, fractionating yields a substantially pure compound which Seria No. 526,574. 0 is totally free from chlorine. 8 Claims. (C. 260-599) The chlorination of toluene is essentially a step-wise reaction and thus the chlorination can be terminated be fore more than two or three percent is converted to the The present invention relates to a process for the pro 5 tri-chloro derivative. This will correspond to a benzal duction of benzaldehyde from toluene. More particu chloride content of no more than about 35%, depending larly, it relates to a process wherein toluene is reacted upon the particular conditions of the reaction, the balance with chlorine and hydrolyzed to produce benzaldehyde being benzyl chloride with a minor amount of unreacted free from chlorine. toluene. Consequently, the reaction is terminated far The chlorination of toluene will result in the introduc 20 short of the maximum formation of benzal chloride so tion of one, two or three chlorine atoms in the methyl that substantially none of the toluene is lost through the side chain.