Read the Decision
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
APP203875 Submissions Compilation.Pdf
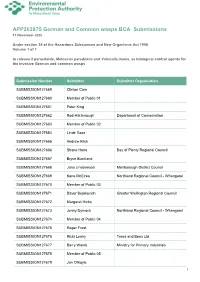
APP203875 German and Common wasps BCA Submissions 11 November 2020 Under section 34 of the Hazardous Substances and New Organisms Act 1996 Volume 1 of 1 to release 2 parasitoids, Metoecus paradoxus and Volucella inanis, as biological control agents for the invasive German and common wasps Submission Number Submitter Submitter Organisation SUBMISSION127659 Clinton Care SUBMISSION127660 Member of Public 01 SUBMISSION127661 Peter King SUBMISSION127662 Rod Hitchmough Department of Conservation SUBMISSION127663 Member of Public 02 SUBMISSION127664 Linde Rose SUBMISSION127665 Andrew Blick SUBMISSION127666 Shane Hona Bay of Plenty Regional Council SUBMISSION127667 Bryce Buckland SUBMISSION127668 Jono Underwood Marlborough District Council SUBMISSION127669 Kane McElrea Northland Regional Council - Whangarei SUBMISSION127670 Member of Public 03 SUBMISSION127671 Davor Bejakovich Greater Wellington Regional Council SUBMISSION127672 Margaret Hicks SUBMISSION127673 Jenny Dymock Northland Regional Council - Whangarei SUBMISSION127674 Member of Public 04 SUBMISSION127675 Roger Frost SUBMISSION127676 Ricki Leahy Trees and Bees Ltd SUBMISSION127677 Barry Wards Ministry for Primary Industries SUBMISSION127678 Member of Public 05 SUBMISSION127679 Jan O'Boyle 1 Submission Number Submitter Submitter Organisation Apiculture New Zealand Science and SUBMISSION127680 Sue Carter Research Focus Group SUBMISSION127681 Andrea Dorn SUBMISSION127682 Benita Wakefield Te Rūnanga o Ngāi Tahu SUBMISSION127683 Emma Edney-Browne Auckland Council SUBMISSION127684 David Hunter -

Elytra Reduction May Affect the Evolution of Beetle Hind Wings
Zoomorphology https://doi.org/10.1007/s00435-017-0388-1 ORIGINAL PAPER Elytra reduction may affect the evolution of beetle hind wings Jakub Goczał1 · Robert Rossa1 · Adam Tofilski2 Received: 21 July 2017 / Revised: 31 October 2017 / Accepted: 14 November 2017 © The Author(s) 2017. This article is an open access publication Abstract Beetles are one of the largest and most diverse groups of animals in the world. Conversion of forewings into hardened shields is perceived as a key adaptation that has greatly supported the evolutionary success of this taxa. Beetle elytra play an essential role: they minimize the influence of unfavorable external factors and protect insects against predators. Therefore, it is particularly interesting why some beetles have reduced their shields. This rare phenomenon is called brachelytry and its evolution and implications remain largely unexplored. In this paper, we focused on rare group of brachelytrous beetles with exposed hind wings. We have investigated whether the elytra loss in different beetle taxa is accompanied with the hind wing shape modification, and whether these changes are similar among unrelated beetle taxa. We found that hind wings shape differ markedly between related brachelytrous and macroelytrous beetles. Moreover, we revealed that modifications of hind wings have followed similar patterns and resulted in homoplasy in this trait among some unrelated groups of wing-exposed brachelytrous beetles. Our results suggest that elytra reduction may affect the evolution of beetle hind wings. Keywords Beetle · Elytra · Evolution · Wings · Homoplasy · Brachelytry Introduction same mechanism determines wing modification in all other insects, including beetles. However, recent studies have The Coleoptera order encompasses almost the quarter of all provided evidence that formation of elytra in beetles is less currently known animal species (Grimaldi and Engel 2005; affected by Hox gene than previously expected (Tomoyasu Hunt et al. -

Coleoptera: Tenebrionoidea)
ACTA ENTOMOLOGICA MUSEI NATIONALIS PRAGAE Published 30.vi.2010 Volume 50(1), pp. 157–166 ISSN 0374-1036 A review of Ripiphoridae in the Arabian Peninsula (Coleoptera: Tenebrionoidea) Jan BATELKA Nad Vodovodem 16, CZ-100 00 Praha 10, Czech Republic; e-mail: [email protected] Abstract. Distribution of the Ripiphoridae (Coleoptera: Tenebrionoidea) in the Arabian Peninsula is evaluated. Six species belonging to the genera Macrosiagon Hentz, 1830 and Ripiphorus Bosc, 1791 are fi gured and keyed, and the distribution of each species is mapped. Including new and previously published records, the Ripiphoridae are now reported from 19 localities of the Arabian Peninsula and offshore islands. Coordinates for each exact locality are given. Key words. Coleoptera, Tenebrionoidea, Ripiphoridae, Ripiphorinae, Macrosia- gon, Ripiphorus, faunistics, Arabian Peninsula, Palaearctic Region Introduction The Ripiphoridae (Coleoptera: Tenebrionoidea) are a cosmopolitan group of parasitoids, whose biogeography is only poorly understood. Only scarce distributional data are usually available because of their cryptic way of life in larval stages and short-lived adults. The Arabian Peninsula plays an important role in the understanding of their distribution in the Old World, as it is a transitional zone among three main zoogeographical realms: Afrotropical, Oriental and Palaearctic. The aim of this paper is to provide basis for further studies of the Ripiphoridae in this part of Asia and to make further research easier for those students who are not familiar with these rarely collected beetles. Each species is keyed and fi gured based on specimens collected in the Arabian Peninsula, with emphasis on colour variability and also on sexual dimorphism where appropriate. -

Wedge-Shaped Beetles (Suggested Common Name) Ripiphorus Spp. (Insecta: Coleoptera: Ripiphoridae)1 David Owens, Ashley N
EENY613 Wedge-Shaped Beetles (suggested common name) Ripiphorus spp. (Insecta: Coleoptera: Ripiphoridae)1 David Owens, Ashley N. Mortensen, Jeanette Klopchin, William Kern, and Jamie D. Ellis2 Introduction Ripiphoridae are a family of unusual parasitic beetles that are thought to be related to tumbling flower beetles (Coleoptera: Mordellidae) and blister beetles (Coleoptera: Meloidae). There is disagreement over the spelling of the family (Ripiphoridae) and genus (Ripiphorus) names. Here we use the original spelling that starts with only the letter Figure 1. Adult specimens of the two genera of Ripiphoridae. A) “R”; however, an initial “Rh” has also been used in the Macrosiagon Hentz, and B) Ripiphorus Bosc. scientific community (Rhipiphoridae and Rhipiphorus). Credits: Allen M. Boatman There are an estimated 35 nearctic species of Ripiphorus, Generally, the biology of the family Ripiphoridae is poorly two of which have been collected in Florida: Ripiphorus known. Ripiphorids parasitize bees and wasps (Hymenop- schwarzi LeConte (Figure 2A) and Ripiphorus fasciatus Say tera), roaches (Blattodea), and wood-boring beetles (Co- (Figure 2B). Due to limited information for both of these leoptera). However, the specific hosts for many ripiphorid species, the information presented below is characteristic species are unknown. Furthermore, only one sex (either of the genus Ripiphorus. Information specific to Ripiphorus male or female) has been described for several species, and fasciatus and Ripiphorus schwarzi is presented where the males and females of some species look different. detailed information is available. Two genera of Ripiphoridae infest hymenopteran (bee and wasp) nests: Macrosiagon Hentz (Figure 1A) and Ripiphorus Bosc (formerly Myodites Latreille) (Figure 1B). Species of Macrosiagon are parasites of a variety of hymenopteran families including: Halictidae, Vespidae, Tiphiidae, Apidae, Pompilidae, Crabronidae, and Sphecidae. -

The Evolution and Genomic Basis of Beetle Diversity
The evolution and genomic basis of beetle diversity Duane D. McKennaa,b,1,2, Seunggwan Shina,b,2, Dirk Ahrensc, Michael Balked, Cristian Beza-Bezaa,b, Dave J. Clarkea,b, Alexander Donathe, Hermes E. Escalonae,f,g, Frank Friedrichh, Harald Letschi, Shanlin Liuj, David Maddisonk, Christoph Mayere, Bernhard Misofe, Peyton J. Murina, Oliver Niehuisg, Ralph S. Petersc, Lars Podsiadlowskie, l m l,n o f l Hans Pohl , Erin D. Scully , Evgeny V. Yan , Xin Zhou , Adam Slipinski , and Rolf G. Beutel aDepartment of Biological Sciences, University of Memphis, Memphis, TN 38152; bCenter for Biodiversity Research, University of Memphis, Memphis, TN 38152; cCenter for Taxonomy and Evolutionary Research, Arthropoda Department, Zoologisches Forschungsmuseum Alexander Koenig, 53113 Bonn, Germany; dBavarian State Collection of Zoology, Bavarian Natural History Collections, 81247 Munich, Germany; eCenter for Molecular Biodiversity Research, Zoological Research Museum Alexander Koenig, 53113 Bonn, Germany; fAustralian National Insect Collection, Commonwealth Scientific and Industrial Research Organisation, Canberra, ACT 2601, Australia; gDepartment of Evolutionary Biology and Ecology, Institute for Biology I (Zoology), University of Freiburg, 79104 Freiburg, Germany; hInstitute of Zoology, University of Hamburg, D-20146 Hamburg, Germany; iDepartment of Botany and Biodiversity Research, University of Wien, Wien 1030, Austria; jChina National GeneBank, BGI-Shenzhen, 518083 Guangdong, People’s Republic of China; kDepartment of Integrative Biology, Oregon State -

White Admiral Newsletter
W h i t e A d m i r a l Newsletter 88 Summer 2014 Suffolk Naturalists’ Society C o n te n t s E d i t or i a l Ben Heather 1 Another new fungus for Suffolk Neil Mahler 2 The battle continues…the fight Matt Holden 4 against invasive alien plants in the Stour Valley! Nesting Materials Richard Stewart 8 How you can help monitor Suffolk’s Su e H o ot on 8 b a t s ? Records please! Rosemary Leaf Beetle Ben Heather 10 Stratiomys longicornis – a fly a long Peter Vincent 11 way from home! Periglacial Landforms in Breckland Caroline Markham 13 Are some roadside plants on the Dr. Anne Kell and 15 verge of extinction? Dennis Kell Where has all the road kill gone? Tom Langton 21 Back on the Hopper Trail in 2013 Colin Lucas & 22 Tricia Taylor Volucella zonaria – an impressive Peter Vincent 24 b e a s t Suffolk Show Wildlife H a w k H on e y 27 Suffolk’s Nature Strategy Nick Collinson 29 Species ‘Re - introductions’ Nick Miller 32 ISSN 0959-8537 Published by the Suffolk Naturalists’ Society c/o Ipswich Museum, High Street, Ipswich, Suffolk IP1 3QH Registered Charity No. 206084 © Suffolk Naturalists’ Society Front cover: Alder spittle bug - Aphrophora alni by Ben Heather Newsletter 88 - Summer 2014 Thank you to all those who have contributed to this full issue of the White Admiral newsletter. This issue covers a wide range of topics from roadside verges to an observation on the lack of roadkill on our roads. -

Appendix 1 Appendix 2
Oikos OIK-07259 de Manincor, N., Hautekeete, N., Piquot, Y., Schatz, B., Vanappelghem, C. and Massol, F. 2020. Does phenology explain plant–pollinator interactions at different latitudes? An assessment of its explanatory power in plant–hoverfly networks in French calcareous grasslands. – Oikos doi: 10.1111/oik.07259 Appendix 1 The following Supplementary material is available for this article: Figure A1. Sites location in France. Figure A2. Block clustering provided by LBM in the site of Falaises (FAL, Normandie), overlaid on a heatmap of species phenology overlap. Figure A3. Block clustering provided by LBM in the site of Bois de Fontaret (BF, Occitanie), overlaid on a heatmap of species phenology overlap. Figure A4. Block clustering provided by LBM in the site of Larris (LAR, Hauts-de-France), overlaid on a heatmap of species phenology overlap. Figure A5. Block clustering provided by LBM in the site of Riez (R, Hauts-de-France), overlaid on a heatmap of species phenology overlap. Table A1. Table of transformed plant abundances. Table A2. Table of hoverfly and plant species names and abbreviations used in the LBM Figures. Table A3. Table of model accuracy. Appendix 2 Appendix 2.1. Script modularity and latent block model analysis (LBM). Appendix 2.2. Model code. Appendix 2.3. Model script for the 16 models. 1 Appendix 1 Figure A1. Site location in France: in blue the French départements Pas-de-Calais and Somme (Hauts-de-France region), in green the départements Eure and Seine Maritime (Normandie region), in orange the départment Gard (Occitanie region). The six sites correspond to the red dots (with the sites of Fourches and Bois de Fontaret represented by the same dot due to their closeness). -

Bees and Wasps of the East Sussex South Downs
A SURVEY OF THE BEES AND WASPS OF FIFTEEN CHALK GRASSLAND AND CHALK HEATH SITES WITHIN THE EAST SUSSEX SOUTH DOWNS Steven Falk, 2011 A SURVEY OF THE BEES AND WASPS OF FIFTEEN CHALK GRASSLAND AND CHALK HEATH SITES WITHIN THE EAST SUSSEX SOUTH DOWNS Steven Falk, 2011 Abstract For six years between 2003 and 2008, over 100 site visits were made to fifteen chalk grassland and chalk heath sites within the South Downs of Vice-county 14 (East Sussex). This produced a list of 227 bee and wasp species and revealed the comparative frequency of different species, the comparative richness of different sites and provided a basic insight into how many of the species interact with the South Downs at a site and landscape level. The study revealed that, in addition to the character of the semi-natural grasslands present, the bee and wasp fauna is also influenced by the more intensively-managed agricultural landscapes of the Downs, with many species taking advantage of blossoming hedge shrubs, flowery fallow fields, flowery arable field margins, flowering crops such as Rape, plus plants such as buttercups, thistles and dandelions within relatively improved pasture. Some very rare species were encountered, notably the bee Halictus eurygnathus Blüthgen which had not been seen in Britain since 1946. This was eventually recorded at seven sites and was associated with an abundance of Greater Knapweed. The very rare bees Anthophora retusa (Linnaeus) and Andrena niveata Friese were also observed foraging on several dates during their flight periods, providing a better insight into their ecology and conservation requirements. -

HOVERFLY NEWSLETTER Dipterists
HOVERFLY NUMBER 41 NEWSLETTER SPRING 2006 Dipterists Forum ISSN 1358-5029 As a new season begins, no doubt we are all hoping for a more productive recording year than we have had in the last three or so. Despite the frustration of recent seasons it is clear that national and international study of hoverflies is in good health, as witnessed by the success of the Leiden symposium and the Recording Scheme’s report (though the conundrum of the decline in UK records of difficult species is mystifying). New readers may wonder why the list of literature references from page 15 onwards covers publications for the year 2000 only. The reason for this is that for several issues nobody was available to compile these lists. Roger Morris kindly agreed to take on this task and to catch up for the missing years. Each newsletter for the present will include a list covering one complete year of the backlog, and since there are two newsletters per year the backlog will gradually be eliminated. Once again I thank all contributors and I welcome articles for future newsletters; these may be sent as email attachments, typed hard copy, manuscript or even dictated by phone, if you wish. Please do not forget the “Interesting Recent Records” feature, which is rather sparse in this issue. Copy for Hoverfly Newsletter No. 42 (which is expected to be issued with the Autumn 2006 Dipterists Forum Bulletin) should be sent to me: David Iliff, Green Willows, Station Road, Woodmancote, Cheltenham, Glos, GL52 9HN, (telephone 01242 674398), email: [email protected], to reach me by 20 June 2006. -

Helminth Parasites of the Common Grackle Quiscalus Quiscula Versicolor Vieillot in Indiana
This dissertation has been 62—3609 microfilmed exactly as received WELKER, George William, 1923- HELMINTH PARASITES OF THE COMMON GRACKLE QUISCALUS QUISCULA VERSICOLOR VIEILLOT IN INDIANA. The Ohio State University, Ph.D., 1962 Zoology University Microfilms, Inc., Ann Arbor, Michigan HELMINTH PARASITES OP THE COMMON GRACKLE QUISCALU5 QUISCULA VERSICOLOR VIEILLOT IN INDIANA DISSERTATION Presented in Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy in the Graduate School of The Ohio State University By George William Welker, B. S., M. A. u _ u u u The Ohio State University 1962 Approved by: 1'XJijdJi ~7 Adviser urtameenhtt of Zoology and Entomology Dedicated as a tribute of appreciation and admiration to ELLEN ANN, my wife, for her help and for the sacrifices which she made during the four years covered by this study. ii ACKNOWLEDGMENTS The author wishes to express his sincere appreciation for all the help and cooperation which he has received from many people during the course of this study: Dr. Joseph Jones, Jr. of St. Augustine's College, Raleigh, North Carolina; Dr. Donal Myer, Southern Illinois university; Dr. E. J. Robinson, Jr., Kenyon College, Gambier, Ohio; Dr. Martin J. Ulmer, Iowa State University, Ames, Iowa; and Dr. A. Carter Broad and Dr. Carl Reese of the reading committee who helped in checking the paper for errors. Special acknowledgment goes to two persons whose help and influence are most deeply appreciated. To Professor Robert H. Cooper, Head of the Department of Science at Ball State Teachers College, whose sincere and continuous interest, encouragement and help made possible the completion of the work; and to Professor Joseph N. -

Encyclopedia of Social Insects
G Guests of Social Insects resources and homeostatic conditions. At the same time, successful adaptation to the inner envi- Thomas Parmentier ronment shields them from many predators that Terrestrial Ecology Unit (TEREC), Department of cannot penetrate this hostile space. Social insect Biology, Ghent University, Ghent, Belgium associates are generally known as their guests Laboratory of Socioecology and Socioevolution, or inquilines (Lat. inquilinus: tenant, lodger). KU Leuven, Leuven, Belgium Most such guests live permanently in the host’s Research Unit of Environmental and nest, while some also spend a part of their life Evolutionary Biology, Namur Institute of cycle outside of it. Guests are typically arthropods Complex Systems, and Institute of Life, Earth, associated with one of the four groups of eusocial and the Environment, University of Namur, insects. They are referred to as myrmecophiles Namur, Belgium or ant guests, termitophiles, melittophiles or bee guests, and sphecophiles or wasp guests. The term “myrmecophile” can also be used in a broad sense Synonyms to characterize any organism that depends on ants, including some bacteria, fungi, plants, aphids, Inquilines; Myrmecophiles; Nest parasites; and even birds. It is used here in the narrow Symbionts; Termitophiles sense of arthropods that associated closely with ant nests. Social insect nests may also be parasit- Social insect nests provide a rich microhabitat, ized by other social insects, commonly known as often lavishly endowed with long-lasting social parasites. Although some strategies (mainly resources, such as brood, retrieved or cultivated chemical deception) are similar, the guests of food, and nutrient-rich refuse. Moreover, nest social insects and social parasites greatly differ temperature and humidity are often strictly regu- in terms of their biology, host interaction, host lated. -

Coleoptera: Ripiphoridae: Ripiphorinae)
Eur. J. Entomol. 101: 577–581, 2004 ISSN 1210-5759 Two new wedge-shaped beetles in Albo-Cenomanian ambers of France (Coleoptera: Ripiphoridae: Ripiphorinae) VINCENT PERRICHOT1, ANDRÉ NEL2* and DIDIER NÉRAUDEAU1 1 Géosciences Rennes and CNRS UMR 6118, Université Rennes 1, bât. 15, 263, avenue du Général Leclerc, 35042 Rennes Cedex, France; e-mails: [email protected], [email protected] 2 Entomologie and CNRS UMR 5143, Muséum National d’Histoire Naturelle, 45, rue Buffon, F-75005, Paris, France; e-mail: [email protected] Key words. Coleoptera, Ripiphoridae, Ripiphorinae, new genus, new species, amber fossil, Albian, Cenomanian, Archingeay, Salignac, France Abstract. Paleoripiphorus deploegi gen. n., sp. n. and Macrosiagon ebboi sp. n., described from two French Albo-Cenomanian ambers (mid Cretaceous), are the oldest definitely identified representatives of the Ripiphoridae: Ripiphorinae. They belong to or are closely related to extant genera of this coleopteran subfamily. Together with Myodites burmiticus Cockerell, 1917 from the Albian Burmese amber, they demonstrate that the group is distinctly older than suggested by the hitherto available fossil record. By infer- ence after the biology of the extant Ripiphorinae, Macrosiagon ebboi may have been parasitic on wasps and Paleoripiphorus deploegi on bees, suggesting that Apoidea may have been present in the Lower Cretaceous. INTRODUCTION FAMILY RIPIPHORIDAE The Ripiphoridae is a small family of parasitic beetles Subfamily Ripiphorinae rather poorly represented in the fossil record, mainly by Genus Paleoripiphorus gen. n. species from the Eocene Baltic amber (Spahr, 1981) and the Paleogene of Florissant (Colorado, USA) (Scudder, Type species. Paleoripiphorus deploegi sp. n. 1890; Meyer, 2003).