Drosophila Nicotinamidase and Sir2 in Longevity Regulation and Oxidative Stress Response, and the Mplici Ations for Aging Theory Gregory Miller Wayne State University
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Mycobacterium Haemophilum Sp. Nov., a New Pathogen of Humanst
0020-7713/78/0028-0067$02.0/0 INTERNATIONALJOURNAL OF SYSTEMATICBACTERIOLOGY, Jan. 1978, p. 67-75 Vol. 28, No. 1 Copyright 0 1978 International Association of Microbiological Societies Printed in U.S. A. Mycobacterium haemophilum sp. nov., a New Pathogen of Humanst DAVID SOMPOLINSKY,’v2 ANNIE LAGZIEL,’ DAVID NAVEH,3 AND TULI YANKILEVITZ3 Department of Microbwlogy, Asaf Harofe Government Hospital, Zerifin‘; Rapaport Laboratories, Bar-Ilan University, Ramat-Gan2;and Department of Internal Medicine “A,”Meir Hospital, Kfar Saba,3 Israel A patient under immunosuppressive treatment of Hodgkin’s disease developed generalized skin granulomata and subcutaneous abscesses. Several aspirated pus samples yielded acid-fast rods with the following properties: temperature opti- mum, about 30°C with no growth at 37°C; slow growth (2 to 4 weeks); nonchrom- ogenic; hemoglobin or hemin requirement for growth; catalase negative; pyrazin- amidase and nicotinamidase positive; and urease negative. The guanine-plus- cytosine content of the deoxyribonucleic acid was calculated from the melting temperature to be 66.0 mol%. It is concluded that these isolates belong to a new species, for which the name Mycobacterium haemophilum is proposed. The type strain of this species is strain 1 (= ATCC 29548). The new species is related to M. marinum and M. ulcerans. Granulomatous skin diseases of humans CASE HISTORY caused by mycobacteria other than Mycobacte- After World War 11, a 27-year-old woman was di- rium tuberculosis and M. leprae are well known. agnosed as having tuberculosis. She received treat- The two organisms most often involved etiolog- ment until 1951. In February 1969, at the age of 51, ically are M. -

A Review and Appraisal of the DNA Damage Theory of Ageing
Mutation Research 728 (2011) 12–22 Contents lists available at ScienceDirect Mutation Research/Reviews in Mutation Research jo urnal homepage: www.elsevier.com/locate/reviewsmr Co mmunity address: www.elsevier.com/locate/mutres Review A review and appraisal of the DNA damage theory of ageing a,b a, Alex A. Freitas , Joa˜o Pedro de Magalha˜es * a Integrative Genomics of Ageing Group, Institute of Integrative Biology, University of Liverpool, Biosciences Building, Crown Street, Liverpool, L69 7ZB, UK b School of Computing and Centre for BioMedical Informatics, University of Kent, Canterbury, CT2 7NF, UK A R T I C L E I N F O A B S T R A C T Article history: Given the central role of DNA in life, and how ageing can be seen as the gradual and irreversible Received 10 February 2011 breakdown of living systems, the idea that damage to the DNA is the crucial cause of ageing remains a Received in revised form 2 May 2011 powerful one. DNA damage and mutations of different types clearly accumulate with age in mammalian Accepted 3 May 2011 tissues. Human progeroid syndromes resulting in what appears to be accelerated ageing have been Available online 10 May 2011 linked to defects in DNA repair or processing, suggesting that elevated levels of DNA damage can accelerate physiological decline and the development of age-related diseases not limited to cancer. Keywords: Higher DNA damage may trigger cellular signalling pathways, such as apoptosis, that result in a faster Ageing depletion of stem cells, which in turn contributes to accelerated ageing. -

“This Is Getting Really Old . . . ” the Genetics of Aging
“This is getting really old . ” The Genetics of Aging Prof. Mike Kuchka Department of Biological Sciences OBJECTIVES • Explain how mutations in genes can increase lifespan in various organisms (METHUSELAH gene of Drosophila) • Relate chromosome length with aging (TELOMERE SHORTENING) • Understand how alteration of intracelluar signaling pathway impacts aging (INSULIN-LIKE GROWTH FACTOR) • Relate caloric restriction with aging (Role of SIRTUIN proteins) • Describe accelerated aging disorders in humans (WERNER’S SYNDROME, HUTCHINSON-GILFORD PROGERIA) Aging – the decline in survival and fecundity with advancing age, caused by the accumulation of damage to macromolecules, intracellular organelles, cells, tissues, organs. SOME INTRODUCTORY POINTS • Natural selection does not select for genes that cause aging or determine lifespan. Rather, aging occurs as a result of the pleiotropic effects of genes that specify other processes [Christensen et al. (2006)]. • Genes that influence longevity are involved in stress response and nutrient sensing, generally, intracellular signaling pathways. • In the past century, mean life expectancy in Western countries increased from ~50 to 75 – 80. • Twin studies (human) suggest that 25% of variation in lifespan is caused by genetic differences. • Manipulation of >100 genes in experimental animal models increases longevity. • Most of these genes are also present in the human genome. • Gene manipulations that increase longevity also postpone age-related diseases. Nematode Worm (C. elegans) as a Model Experimental Organism For the Study of Aging OLD YOUNG MUTANT 2 weeks old 2 days old 2 weeks old From: Hopkin, K. (2004) Scientific American, 14: 12 – 17. Mouse (Mus musculus) as a Model Experimental Organism For the Study of Aging From: Hampton, K. -

Calorie Restriction and Sirtuins Revisited
Downloaded from genesdev.cshlp.org on September 30, 2021 - Published by Cold Spring Harbor Laboratory Press REVIEW Calorie restriction and sirtuins revisited Leonard Guarente1 Department of Biology, Glenn Laboratory for the Science of Aging, Massachusetts Institute of Technology, Cambridge, Massachusetts 02139, USA Calorie or dietary restriction (CR) has attracted attention Toward the resolution of discordances because it is the oldest and most robust way to extend rodent life span. The idea that the nutrient sensors, termed Sirtuins and aging sirtuins, might mediate effects of CR was proposed 13 years Perhaps the greatest challenge to the idea that sirtuins ago and has been challenged in the intervening years. This mediate effects of CR was a study describing the failure to review addresses these challenges and draws from a great observe extension of life span in worms or flies transgenic body of new data in the sirtuin field that shows a systematic for the corresponding SIR2 orthologs after controlling for redirection of mammalian physiology in response to diet by genetic background (Burnett et al. 2011). These findings sirtuins. The prospects for drugs that can deliver at least contradicted other, earlier studies that showed extension a subset of the benefits of CR seems very real. of life span in transgenic worms (Tissenbaum and Guarente 2001; Viswanathan et al. 2005; Berdichevsky et al. 2006) A review published previously in Genes & Development and flies (Rogina and Helfand 2004; Wood et al. 2004; (Guarente 2000) stated the hypothesis that calorie re- Bauer et al. 2009). In fact, a subset of these earlier studies striction (CR) slowed aging and the accompanying de- did control for genetic background (e.g., Bauer et al. -

Calorie Restriction and the Exercise of Chromatin
Downloaded from genesdev.cshlp.org on October 3, 2021 - Published by Cold Spring Harbor Laboratory Press REVIEW Calorie restriction and the exercise of chromatin Alejandro Vaquero1,4 and Danny Reinberg2,3 1Chromatin Biology Laboratory, Cancer Epigenetics and Biology Program (PEBC), ICREA, and IDIBELL, L’Hospitalet de Llobregat, Barcelona 08907, Spain; 2Howard Hughes Medical Institute, Department of Biochemistry, New York University-Medical School, New York, New York 10016, USA Since the earliest stages of evolution, organisms have (Masoro2005).Inahallmarkstudyin1935,McCayetal. faced the challenge of sensing and adapting to environ- (1935) demonstrated that, compared with rats fed with mental changes for their survival under compromising a standard diet, rats fed under CR lived longer, weighed less, conditions such as food depletion or stress. Implicit in showed heart hypertrophy, and had smaller livers, which these responses are mechanisms developed during evolu- they explained at the time as an effect of growth retarda- tion that include the targeting of chromatin to allow or tion. Since then, considerable research efforts have revealed prevent expression of fundamental genes and to protect that, surprisingly, the effects of CR on life span are highly genome integrity. Among the different approaches to similar among diverse eukaryotes (e.g., yeast, insects, fish, study these mechanisms, the analysis of the response to and mammals) (Kennedy et al. 2007). Although we can only a moderate reduction of energy intake, also known as speculate on these findings, they suggest that the follow- calorie restriction (CR), has become one of the best sources ing general survival strategy has been conserved through- of information regarding the factors and pathways in- out eukaryotic evolution: In low-nutrient conditions, volved in metabolic adaptation from lower to higher metabolism is adjusted to enable more efficient use of eukaryotes. -

“This Is Getting Really Old . . . ” the Genetics of Aging
“This is getting really old . ” The Genetics of Aging Prof. Mike Kuchka Department of Biological Sciences OBJECTIVES • Explain how mutations in genes can increase lifespan in various organisms (METHUSELAH gene of Drosophila) • Relate chromosome length with aging (TELOMERE SHORTENING) • Understand how alteration of intracellular signaling pathway impacts aging (INSULIN-LIKE GROWTH FACTOR) • Relate caloric restriction with aging (Role of SIRTUIN proteins) • Describe accelerated aging disorders in humans (WERNER’S SYNDROME, HUTCHINSON-GILFORD PROGERIA) Aging – the decline in survival and fecundity with advancing age, caused by the accumulation of damage to macromolecules, intracellular organelles, cells, tissues, organs. SOME INTRODUCTORY POINTS • Natural selection does not select for genes that cause aging or determine lifespan. Rather, aging occurs as a result of the pleiotropic effects of genes that specify other processes [Christensen et al. (2006)]. • Genes that influence longevity are involved in stress response and nutrient sensing, generally, intracellular signaling pathways. • In the past century, mean life expectancy in Western countries increased from ~50 to 75 – 80. • Twin studies (human) suggest that 25% of variation in lifespan is caused by genetic differences. • Manipulation of >100 genes in experimental animal models increases longevity. • Most of these genes are also present in the human genome. • Gene manipulations that increase longevity also postpone age-related diseases. Nematode Worm (C. elegans) as a Model Experimental Organism For the Study of Aging OLD YOUNG MUTANT 2 weeks old 2 days old 2 weeks old From: Hopkin, K. (2004) Scientific American, 14: 12 – 17. Mouse (Mus musculus) as a Model Experimental Organism For the Study of Aging From: Hampton, K. -

Metallochaperones Are Needed for Mycobacterium Tuberculosis And
RESEARCH ARTICLE crossm Metallochaperones Are Needed for Mycobacterium tuberculosis and Escherichia coli Nicotinamidase-Pyrazinamidase Activity Downloaded from Patricia Sheen,a Anuntxi Monsalve,a Jhanina Campos,a Rodolfo Huerta,a Ricardo Antiparra,a Héctor Arteaga,a Patricia Duran,a Carlos Bueno,a Daniela E. Kirwan,b Robert H. Gilman,c Mirko Zimica aLaboratory of Bioinformatics and Molecular Biology, Department of Cellular and Molecular Sciences, School of Science, Universidad Peruana Cayetano Heredia, Lima, Peru bInfection and Immunity Research Institute, St. George’s, University of London, London, United Kingdom c Department of International Health, School of Public Health, Johns Hopkins University, Baltimore, Maryland, USA http://jb.asm.org/ ABSTRACT Mycobacterium tuberculosis nicotinamidase-pyrazinamidase (PZAse) is a metalloenzyme that catalyzes conversion of nicotinamide-pyrazinamide to nicotinic acid-pyrazinoic acid. This study investigated whether a metallochaperone is required for optimal PZAse activity. M. tuberculosis and Escherichia coli PZAses (PZAse-MT and PZAse-EC, respectively) were inactivated by metal depletion (giving PZAse-MT–Apo and PZAse-EC–Apo). Reactivation with the E. coli metallochaperone ZnuA or Rv2059 (the M. tuberculosis analog) was measured. This was repeated following proteolytic on January 16, 2020 at ST GEORGE'S LIBRARY and thermal treatment of ZnuA and Rv2059. The CDC1551 M. tuberculosis reference strain had the Rv2059 coding gene knocked out, and PZA susceptibility and the pyr- azinoic acid (POA) efflux rate were measured. ZnuA (200 M) achieved 65% PZAse- EC–Apo reactivation. Rv2059 (1 M) and ZnuA (1 M) achieved 69% and 34.3% PZAse-MT–Apo reactivation, respectively. Proteolytic treatment of ZnuA and Rv2059 and application of three (but not one) thermal shocks to ZnuA significantly reduced the capacity to reactivate PZAse-MT–Apo. -

Supplementary Information
Supplementary information (a) (b) Figure S1. Resistant (a) and sensitive (b) gene scores plotted against subsystems involved in cell regulation. The small circles represent the individual hits and the large circles represent the mean of each subsystem. Each individual score signifies the mean of 12 trials – three biological and four technical. The p-value was calculated as a two-tailed t-test and significance was determined using the Benjamini-Hochberg procedure; false discovery rate was selected to be 0.1. Plots constructed using Pathway Tools, Omics Dashboard. Figure S2. Connectivity map displaying the predicted functional associations between the silver-resistant gene hits; disconnected gene hits not shown. The thicknesses of the lines indicate the degree of confidence prediction for the given interaction, based on fusion, co-occurrence, experimental and co-expression data. Figure produced using STRING (version 10.5) and a medium confidence score (approximate probability) of 0.4. Figure S3. Connectivity map displaying the predicted functional associations between the silver-sensitive gene hits; disconnected gene hits not shown. The thicknesses of the lines indicate the degree of confidence prediction for the given interaction, based on fusion, co-occurrence, experimental and co-expression data. Figure produced using STRING (version 10.5) and a medium confidence score (approximate probability) of 0.4. Figure S4. Metabolic overview of the pathways in Escherichia coli. The pathways involved in silver-resistance are coloured according to respective normalized score. Each individual score represents the mean of 12 trials – three biological and four technical. Amino acid – upward pointing triangle, carbohydrate – square, proteins – diamond, purines – vertical ellipse, cofactor – downward pointing triangle, tRNA – tee, and other – circle. -

Appl. Microbiol. Biotechnol. 78: 293-307
Appl Microbiol Biotechnol (2008) 78:293–307 DOI 10.1007/s00253-007-1308-y APPLIED GENETICS AND MOLECULAR BIOTECHNOLOGY PA2663 (PpyR) increases biofilm formation in Pseudomonas aeruginosa PAO1 through the psl operon and stimulates virulence and quorum-sensing phenotypes Can Attila & Akihiro Ueda & Thomas K. Wood Received: 19 October 2007 /Revised: 26 November 2007 /Accepted: 28 November 2007 / Published online: 22 December 2007 # Springer-Verlag 2007 Abstract Previously, we identified the uncharacterized PA2130, and PA4549) and for small molecule transport predicted membrane protein PA2663 of Pseudomonas (PA0326, PA1541, PA1632, PA1971, PA2214, PA2215, aeruginosa PAO1 as a virulence factor using a poplar tree PA2678, and PA3407). Phenotype arrays also showed that model; PA2663 was induced in the poplar rhizosphere and, PA2663 represses growth on D-gluconic acid, D-mannitol, upon inactivation, it caused 20-fold lower biofilm forma- and N-phthaloyl-L-glutamic acid. Hence, the PA2663 gene tion (Attila et al., Microb Biotechnol, 2008). Here, we product increases biofilm formation by increasing the psl- confirmed that PA2663 is related to biofilm formation by operon-derived exopolysaccharides and increases pyover- restoring the wild-type phenotype by complementing the dine synthesis, PQS production, and elastase activity while PA2663 mutation in trans and investigated the genetic basis reducing swarming and swimming motility. We speculate of its influence on biofilm formation through whole- that PA2663 performs these myriad functions as a novel transcriptome and -phenotype studies. Upon inactivating membrane sensor. PA2663 by transposon insertion, the psl operon that encodes a galactose- and mannose-rich exopolysaccharide Keywords Biofilm . Pseudomonas aeruginosa . was highly repressed (verified by RT-PCR). -

Longevity, Genes, and Aging: a View Provided by a Genetic Model System1
Experimental Gerontology, Vol. 34, No. 1, pp. 1–6, 1999 Copyright © 1999 Elsevier Science Inc. Printed in the USA. All rights reserved 0531-5565/99 $–see front matter PII S0531-5565(98)00053-9 LONGEVITY, GENES, AND AGING: A VIEW PROVIDED BY A GENETIC MODEL SYSTEM1 1 S. MICHAL JAZWINSKI Department of Biochemistry and Molecular Biology, Louisiana State University Medical Center, Box P7-2, 1901 Perdido Street, New Orleans, Louisiana 70112 Abstract—The genetic analysis of aging in the yeast Saccharomyces cerevisiae has revealed the importance of metabolic capacity, resistance to stress, integrity of gene regulation, and genetic stability for longevity. A balance between these life maintenance processes is sustained by the RAS2 gene, which channels cellular resources among them. This gene cooperates with mitochondria and PHB1 in metabolic adjustments important for longevity. It also modulates stress responses. Transcriptional silencing of heterochromatic regions of the genome is lost during aging, suggesting that gene dysregulation accompanies the aging process. There is evidence that this age change plays a causal role. Aging possesses features of a nonlinear process, and it is likely that application of nonlinear system methodology to aging will be productive. © 1999 Elsevier Science Inc. All rights reserved. Key words: metabolism, stress responses, gene dysregulation, nonlinear system INTRODUCTION IF WE WERE to design an organism, we would endow it with sufficient lifetime metabolic capacity, stress resistance, integrity of gene regulation, and genetic stability. Given a limit to the resources available, we would need to strike a balance between these processes and reproduc- tion. Genetic analysis of the aging process has confirmed the soundness of these engineering principles (Jazwinski, 1996). -

Generated by SRI International Pathway Tools Version 25.0, Authors S
Authors: Pallavi Subhraveti Ron Caspi Peter Midford Peter D Karp An online version of this diagram is available at BioCyc.org. Biosynthetic pathways are positioned in the left of the cytoplasm, degradative pathways on the right, and reactions not assigned to any pathway are in the far right of the cytoplasm. Transporters and membrane proteins are shown on the membrane. Ingrid Keseler Periplasmic (where appropriate) and extracellular reactions and proteins may also be shown. Pathways are colored according to their cellular function. Gcf_004758865Cyc: Acinetobacter baumannii CIAT758 Cellular Overview Connections between pathways are omitted for legibility. -
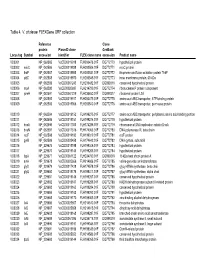
Table 4. V. Cholerae Flexgene ORF Collection
Table 4. V. cholerae FLEXGene ORF collection Reference Clone protein PlasmID clone GenBank Locus tag Symbol accession identifier FLEX clone name accession Product name VC0001 NP_062585 VcCD00019918 FLH200476.01F DQ772770 hypothetical protein VC0002 mioC NP_062586 VcCD00019938 FLH200506.01F DQ772771 mioC protein VC0003 thdF NP_062587 VcCD00019958 FLH200531.01F DQ772772 thiophene and furan oxidation protein ThdF VC0004 yidC NP_062588 VcCD00019970 FLH200545.01F DQ772773 inner membrane protein, 60 kDa VC0005 NP_062589 VcCD00061243 FLH236482.01F DQ899316 conserved hypothetical protein VC0006 rnpA NP_062590 VcCD00025697 FLH214799.01F DQ772774 ribonuclease P protein component VC0007 rpmH NP_062591 VcCD00061229 FLH236450.01F DQ899317 ribosomal protein L34 VC0008 NP_062592 VcCD00019917 FLH200475.01F DQ772775 amino acid ABC transporter, ATP-binding protein VC0009 NP_062593 VcCD00019966 FLH200540.01F DQ772776 amino acid ABC transproter, permease protein VC0010 NP_062594 VcCD00019152 FLH199275.01F DQ772777 amino acid ABC transporter, periplasmic amino acid-binding portion VC0011 NP_062595 VcCD00019151 FLH199274.01F DQ772778 hypothetical protein VC0012 dnaA NP_062596 VcCD00017363 FLH174286.01F DQ772779 chromosomal DNA replication initiator DnaA VC0013 dnaN NP_062597 VcCD00017316 FLH174063.01F DQ772780 DNA polymerase III, beta chain VC0014 recF NP_062598 VcCD00019182 FLH199319.01F DQ772781 recF protein VC0015 gyrB NP_062599 VcCD00025458 FLH174642.01F DQ772782 DNA gyrase, subunit B VC0016 NP_229675 VcCD00019198 FLH199346.01F DQ772783 hypothetical protein