The Trans-SNARE-Regulating Function of Munc18-1 Is Essential to Synaptic Exocytosis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
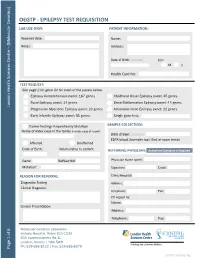
Oegtp - Epilepsy Test Requisition Lab Use Only: Patient Information
OEGTP - EPILEPSY TEST REQUISITION LAB USE ONLY: PATIENT INFORMATION: Received date: Name: Notes: Address: Date of Birth: YY/MM/DD Sex: M F Health Card No: TEST REQUEST: See page 2 for gene list for each of the panels below Epilepsy Comprehensive panel: 167 genes Childhood Onset Epilepsy panel: 45 genes Focal Epilepsy panel: 14 genes Brain Malformation Epilepsy panel: 44 genes London Health Sciences Centre – (Molecular Genetics) London Health Sciences Centre Progressive Myoclonic Epilepsy panel: 20 genes Actionable Gene Epilepsy panel: 22 genes Early Infantile Epilepsy panel: 51 genes Single gene test: Carrier Testing/ KnownFamily Mutation SAMPLE COLLECTION: Name of index case in the family (include copy of report) Date drawn: YY/MM/DD EDTA blood (lavender top) (5ml at room temp) Affected Unaffected Date of Birth: Relationship to patient: REFERRING PHYSICIAN: Authorized Signature is Required Gene: RefSeq:NM Physician Name (print): Mutation: Signature: Email: REASON FOR REFERRAL: Clinic/Hospital: Diagnostic Testing Address: Clinical Diagnosis: Telephone: Fax: CC report to: Name: Clinical Presentation: Address: Telephone: Fax: Molecular Genetics Laboratory Victoria Hospital, Room B10-123A 800 Commissioners Rd. E. London, Ontario | N6A 5W9 Pathology and Laboratory Medicine Ph: 519-685-8122 | Fax: 519-685-8279 Page 1 of 6 Page OEGTP (2021/05/28) OEGTP - EPILEPSY TEST PANELS Patient Identifier: COMPREHENSIVE EPILEPSY PANEL: 167 Genes ACTB, ACTG1, ADSL, AKT3, ALDH7A1, AMT, AP3B2, ARFGEF2, ARHGEF9, ARV1, ARX, ASAH1, ASNS, ATP1A3, ATP6V0A2, ATP7A, -

Epileptic Mechanisms Shared by Alzheimer's Disease
International Journal of Molecular Sciences Review Epileptic Mechanisms Shared by Alzheimer’s Disease: Viewed via the Unique Lens of Genetic Epilepsy Jing-Qiong Kang 1,2 1 Department of Neurology & Pharmacology, Vanderbilt University Medical Center, Nashville, TN 37232-8552, USA; [email protected]; Tel.: +1-615-936-8399; Fax: +1-615-322-5517 2 Vanderbilt Kennedy Center of Human Development, Vanderbilt Brain Institute, Vanderbilt University, Nashville, TN 37232-8522, USA Abstract: Our recent work on genetic epilepsy (GE) has identified common mechanisms between GE and neurodegenerative diseases including Alzheimer’s disease (AD). Although both disorders are seemingly unrelated and occur at opposite ends of the age spectrum, it is likely there are shared mechanisms and studies on GE could provide unique insights into AD pathogenesis. Neurodegen- erative diseases are typically late-onset disorders, but the underlying pathology may have already occurred long before the clinical symptoms emerge. Pathophysiology in the early phase of these diseases is understudied but critical for developing mechanism-based treatment. In AD, increased seizure susceptibility and silent epileptiform activity due to disrupted excitatory/inhibitory (E/I) balance has been identified much earlier than cognition deficit. Increased epileptiform activity is likely a main pathology in the early phase that directly contributes to impaired cognition. It is an enormous challenge to model the early phase of pathology with conventional AD mouse models due to the chronic disease course, let alone the complex interplay between subclinical nonconvulsive epileptiform activity, AD pathology, and cognition deficit. We have extensively studied GE, especially with gene mutations that affect the GABA pathway such as mutations in GABAA receptors and GABA transporter Citation: Kang, J.-Q. -

Abstracts from the 51St European Society of Human Genetics Conference: Electronic Posters
European Journal of Human Genetics (2019) 27:870–1041 https://doi.org/10.1038/s41431-019-0408-3 MEETING ABSTRACTS Abstracts from the 51st European Society of Human Genetics Conference: Electronic Posters © European Society of Human Genetics 2019 June 16–19, 2018, Fiera Milano Congressi, Milan Italy Sponsorship: Publication of this supplement was sponsored by the European Society of Human Genetics. All content was reviewed and approved by the ESHG Scientific Programme Committee, which held full responsibility for the abstract selections. Disclosure Information: In order to help readers form their own judgments of potential bias in published abstracts, authors are asked to declare any competing financial interests. Contributions of up to EUR 10 000.- (Ten thousand Euros, or equivalent value in kind) per year per company are considered "Modest". Contributions above EUR 10 000.- per year are considered "Significant". 1234567890();,: 1234567890();,: E-P01 Reproductive Genetics/Prenatal Genetics then compared this data to de novo cases where research based PO studies were completed (N=57) in NY. E-P01.01 Results: MFSIQ (66.4) for familial deletions was Parent of origin in familial 22q11.2 deletions impacts full statistically lower (p = .01) than for de novo deletions scale intelligence quotient scores (N=399, MFSIQ=76.2). MFSIQ for children with mater- nally inherited deletions (63.7) was statistically lower D. E. McGinn1,2, M. Unolt3,4, T. B. Crowley1, B. S. Emanuel1,5, (p = .03) than for paternally inherited deletions (72.0). As E. H. Zackai1,5, E. Moss1, B. Morrow6, B. Nowakowska7,J. compared with the NY cohort where the MFSIQ for Vermeesch8, A. -

Supplementary Table 2
Supplementary Table 2. Differentially Expressed Genes following Sham treatment relative to Untreated Controls Fold Change Accession Name Symbol 3 h 12 h NM_013121 CD28 antigen Cd28 12.82 BG665360 FMS-like tyrosine kinase 1 Flt1 9.63 NM_012701 Adrenergic receptor, beta 1 Adrb1 8.24 0.46 U20796 Nuclear receptor subfamily 1, group D, member 2 Nr1d2 7.22 NM_017116 Calpain 2 Capn2 6.41 BE097282 Guanine nucleotide binding protein, alpha 12 Gna12 6.21 NM_053328 Basic helix-loop-helix domain containing, class B2 Bhlhb2 5.79 NM_053831 Guanylate cyclase 2f Gucy2f 5.71 AW251703 Tumor necrosis factor receptor superfamily, member 12a Tnfrsf12a 5.57 NM_021691 Twist homolog 2 (Drosophila) Twist2 5.42 NM_133550 Fc receptor, IgE, low affinity II, alpha polypeptide Fcer2a 4.93 NM_031120 Signal sequence receptor, gamma Ssr3 4.84 NM_053544 Secreted frizzled-related protein 4 Sfrp4 4.73 NM_053910 Pleckstrin homology, Sec7 and coiled/coil domains 1 Pscd1 4.69 BE113233 Suppressor of cytokine signaling 2 Socs2 4.68 NM_053949 Potassium voltage-gated channel, subfamily H (eag- Kcnh2 4.60 related), member 2 NM_017305 Glutamate cysteine ligase, modifier subunit Gclm 4.59 NM_017309 Protein phospatase 3, regulatory subunit B, alpha Ppp3r1 4.54 isoform,type 1 NM_012765 5-hydroxytryptamine (serotonin) receptor 2C Htr2c 4.46 NM_017218 V-erb-b2 erythroblastic leukemia viral oncogene homolog Erbb3 4.42 3 (avian) AW918369 Zinc finger protein 191 Zfp191 4.38 NM_031034 Guanine nucleotide binding protein, alpha 12 Gna12 4.38 NM_017020 Interleukin 6 receptor Il6r 4.37 AJ002942 -

STXBP1 Analysis for Early Infantile Epileptic Encephalopathy
STXBP1 Analysis for Early Infantile Epileptic Encephalopathy Clinical Features: Early infantile epileptic encephalopathy (EIEE) [MIM #308350], also known as Ohtahara syndrome, is a severe form of epilepsy characterized by frequent tonic spasms with onset in the first months of life. EEG reveals suppression-burst patterns, characterized by high-voltage bursts alternating with almost flat suppression phases. Seizures are medically intractable with evolution to West syndrome at 3-6 months of age and then Lennox- Gastaut syndrome at 1-3 years of age. EIEE represents less than 1% of all epilepsies occurring in children less than 15 years of age (1). Patients have severe developmental delay and poor prognosis. Molecular Genetics: Mutations of the Syntaxin binding protein 1 (STXBP1) [OMIM #602926] have been identified in patients with early infantile epileptic encephalopathy 4 (EIEE4) [MIM #612164]. The STXBP1 gene maps to 9q34.1 and has 19 coding exons. Syntaxin binding protein 1, more commonly referred to as MUNC18-1, is a neuron specific protein of the SEC1 family of membrane-trafficking proteins. MUNC18-1 is expressed throughout the brain and is a key component for calcium-dependent neurotransmitter release. Sequencing of STXBP1 detected mutations in 4 out of 106 patients with EIEE [2]. Earlier reports identified 4 heterozygous missense mutation in 13 patients with EIEE (2). Affected individuals with microdeletions involving STXBP1 have also been reported (3). Inheritance: STXBP1 mutations are inherited in an autosomal dominant pattern. Most cases are de novo and there has been one family reported with paternal mosaicism of a STXBP1 mutation (4). Resources: Aaron’s Ohtahara Foundation www.ohtahara.org Test methods: Comprehensive sequence coverage of the coding regions and splice junctions of the STXBP1 gene is performed. -

Deciphering the Molecular Profile of Plaques, Memory Decline And
ORIGINAL RESEARCH ARTICLE published: 16 April 2014 AGING NEUROSCIENCE doi: 10.3389/fnagi.2014.00075 Deciphering the molecular profile of plaques, memory decline and neuron loss in two mouse models for Alzheimer’s disease by deep sequencing Yvonne Bouter 1†,Tim Kacprowski 2,3†, Robert Weissmann4, Katharina Dietrich1, Henning Borgers 1, Andreas Brauß1, Christian Sperling 4, Oliver Wirths 1, Mario Albrecht 2,5, Lars R. Jensen4, Andreas W. Kuss 4* andThomas A. Bayer 1* 1 Division of Molecular Psychiatry, Georg-August-University Goettingen, University Medicine Goettingen, Goettingen, Germany 2 Department of Bioinformatics, Institute of Biometrics and Medical Informatics, University Medicine Greifswald, Greifswald, Germany 3 Department of Functional Genomics, Interfaculty Institute for Genetics and Functional Genomics, University Medicine Greifswald, Greifswald, Germany 4 Human Molecular Genetics, Department for Human Genetics of the Institute for Genetics and Functional Genomics, Institute for Human Genetics, University Medicine Greifswald, Ernst-Moritz-Arndt University Greifswald, Greifswald, Germany 5 Institute for Knowledge Discovery, Graz University of Technology, Graz, Austria Edited by: One of the central research questions on the etiology of Alzheimer’s disease (AD) is the Isidro Ferrer, University of Barcelona, elucidation of the molecular signatures triggered by the amyloid cascade of pathological Spain events. Next-generation sequencing allows the identification of genes involved in disease Reviewed by: Isidro Ferrer, University of Barcelona, processes in an unbiased manner. We have combined this technique with the analysis of Spain two AD mouse models: (1) The 5XFAD model develops early plaque formation, intraneu- Dietmar R. Thal, University of Ulm, ronal Ab aggregation, neuron loss, and behavioral deficits. (2)TheTg4–42 model expresses Germany N-truncated Ab4–42 and develops neuron loss and behavioral deficits albeit without plaque *Correspondence: formation. -

Downregulation of Carnitine Acyl-Carnitine Translocase by Mirnas
Page 1 of 288 Diabetes 1 Downregulation of Carnitine acyl-carnitine translocase by miRNAs 132 and 212 amplifies glucose-stimulated insulin secretion Mufaddal S. Soni1, Mary E. Rabaglia1, Sushant Bhatnagar1, Jin Shang2, Olga Ilkayeva3, Randall Mynatt4, Yun-Ping Zhou2, Eric E. Schadt6, Nancy A.Thornberry2, Deborah M. Muoio5, Mark P. Keller1 and Alan D. Attie1 From the 1Department of Biochemistry, University of Wisconsin, Madison, Wisconsin; 2Department of Metabolic Disorders-Diabetes, Merck Research Laboratories, Rahway, New Jersey; 3Sarah W. Stedman Nutrition and Metabolism Center, Duke Institute of Molecular Physiology, 5Departments of Medicine and Pharmacology and Cancer Biology, Durham, North Carolina. 4Pennington Biomedical Research Center, Louisiana State University system, Baton Rouge, Louisiana; 6Institute for Genomics and Multiscale Biology, Mount Sinai School of Medicine, New York, New York. Corresponding author Alan D. Attie, 543A Biochemistry Addition, 433 Babcock Drive, Department of Biochemistry, University of Wisconsin-Madison, Madison, Wisconsin, (608) 262-1372 (Ph), (608) 263-9608 (fax), [email protected]. Running Title: Fatty acyl-carnitines enhance insulin secretion Abstract word count: 163 Main text Word count: 3960 Number of tables: 0 Number of figures: 5 Diabetes Publish Ahead of Print, published online June 26, 2014 Diabetes Page 2 of 288 2 ABSTRACT We previously demonstrated that micro-RNAs 132 and 212 are differentially upregulated in response to obesity in two mouse strains that differ in their susceptibility to obesity-induced diabetes. Here we show the overexpression of micro-RNAs 132 and 212 enhances insulin secretion (IS) in response to glucose and other secretagogues including non-fuel stimuli. We determined that carnitine acyl-carnitine translocase (CACT, Slc25a20) is a direct target of these miRNAs. -

Structural Analysis of De Novo STXBP1 Mutation in Complex with Syntaxin 1A
bioRxiv preprint doi: https://doi.org/10.1101/642702; this version posted May 20, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Structural analysis of de novo STXBP1 mutation in complex with syntaxin 1A reveals a major alteration in the interaction interface in a child with developmental delay and spasticity Authors: Ehud Banne1, Tzipora Falik-Zaccai2,3, Esther Brielle4,5, Limor Kalfon2, Hagay Ladany2, Danielle Klinger4, Dina Schneidman-Duhovny4,6 and Michal Linial4 1 The Genetics Institute, Kaplan Medical Center, Rehovot, affiliated to the Hebrew University and Hadassah Medical School, Jerusalem, 2 Institute of Human Genetics, Galilee Medical Center, Naharia, Israel. 3 Azrieli Faculty of Medicine in the Galilee, Bar Ilan University, Safed, Israel. 4 Department of Biological Chemistry, Institute of Life Sciences, The Hebrew University of Jerusalem, Jerusalem, Israel 5 The Alexander Grass Center for Bioengineering, The Hebrew University of Jerusalem, Jerusalem, Israel 6 The Rachel and Selim Benin School of Computer Science and Engineering, The Hebrew University of Jerusalem, Jerusalem, Israel Email: EB [email protected] DS [email protected] ML [email protected] Keywords: SNARE proteins, Syntaxin, Munc-18, Molecular dynamics, Neurotransmitter release, Synaptic pathology, Epileptic encephalopathies bioRxiv preprint doi: https://doi.org/10.1101/642702; this version posted May 20, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. -

STXBP1 Disorders Scientific
WAYS TO SUPPORT Register Understanding the natural history of STXBP1 is essential to discovering improved treatments. Whether it is a repur- posed drug or a novel therapy, validation in a controlled manner is paramount to understanding efficacy and conse- quently improving the lives of our STXBP1 patients. Families can register with Simons Searchlight by visiting simonssearchlight.org. Caregivers need only supply a ge- netic report confirming diagnosis to get started. Participate in Research Participating in or initiating research is essential to finding better therapies. Research onSTXBP1 is grossly un- derfunded; your support is critical to changing the current Lead paradigm. the Spread Awareness STXBP1 is rare and our families often go for years Charge without the correct diagnosis. Your support in understand- ing the need for genetic analysis and communicating with For a your colleagues and patients is important to getting our families the information they so desperately need. Cure Contribute Whether it is your time or your money, this rare disor- der represents an opportunity to make a meaningful and profound impact on STXBP1 patients’ lives. We hope you will support us today! stxbp1disorders.org stxbp1disorders.org [email protected] [email protected] WHAT IS STXBP1 DISORDER? Diagnosis STXBP1 disorder is an autosomal dominant disease, STXBP1 diagnosis is made through molecular genetic test- resulting from de novo mutations in the STXBP1 gene, ing, through a panel test, exome testing or chromosom- which affects the brain and nervous system, due to impair- al microarray analysis. The genetic testing results would ment of transmission between nerve cells. Patients with the identify a pathogenic heterozygous variant in STXBP1, or disorder typically have some of these symptoms: early onset a contiguous gene deletion that includes STXBP1 and pos- epilepsy, global delay, cognitive impairment (mild to pro- sibly adjacent genes. -

De Novo STXBP1 Mutations in Mental Retardation and Nonsyndromic
14. Post MJ, Yiannoutsos C, Simpson D, et al. Progressive multi- focal leukoencephalopathy in AIDS: are there any MR findings De Novo STXBP1 Mutations useful to patient management and predictive of patient survival? AIDS Clinical Trials Group, 243 Team. AJNR Am J Neurora- in Mental Retardation and diol 1999;20:1896–1906. 15. Shintaku M, Matsumoto R, Sawa H, Nagashima K. Infection Nonsyndromic Epilepsy with JC virus and possible dysplastic ganglion-like transforma- 1 2 tion of the cerebral cortical neurons in a case of progressive Fadi F. Hamdan, PhD, Ame´lie Piton, PhD, Julie Gauthier, PhD,2 Anne Lortie, MD,1 multifocal leukoencephalopathy. J Neuropathol Exp Neurol 3 2 2000;59:921–929. Franc¸ois Dubeau, MD, Sylvia Dobrzeniecka, MSc, Dan Spiegelman, MSc,2 Anne Noreau, MSc,2 16. Del Valle L, Delbue S, Gordon J, et al. Expression of JC virus 1 2 T-antigen in a patient with MS and glioblastoma multiforme. Ste´phanie Pellerin, RN, Me´lanie Coˆte´, BSc, Edouard Henrion, MSc,2 E´ric Fombonne, MD,4 Neurology 2002;58:895–900. 5 17. Raj GV, Khalili K. Transcriptional regulation: lessons from the hu- Laurent Mottron, MD, PhD, Claude Marineau, MSc, MBA,2 Pierre Drapeau, PhD,6 man neurotropic polyomavirus, JCV. Virology 1995;213:283–291. 2 7 18. Messam CA, Hou J, Gronostajski RM, Major EO. Lineage Ronald G. Lafrenie`re, PhD, Jean Claude Lacaille, PhD, Guy A. Rouleau, MD, PhD,2 and pathway of human brain progenitor cells identified by JC virus 1 susceptibility. Ann Neurol 2003;53:636–646. Jacques L. Michaud, MD 19. -

STXBP1 Encephalopathy with Epilepsy
STXBP1 encephalopathy with epilepsy Description STXBP1 encephalopathy with epilepsy is a condition characterized by recurrent seizures (epilepsy), abnormal brain function (encephalopathy), and intellectual disability. The signs and symptoms of this condition typically begin in infancy but can first appear later in childhood or early adulthood. In many affected individuals, seizures stop in early childhood with the other neurological problems continuing throughout life. However, some people with STXBP1 encephalopathy with epilepsy have seizures that persist. The most common seizures in STXBP1 encephalopathy with epilepsy are infantile spasms, which occur before age 1 and consist of involuntary muscle contractions. Other seizure types that can occur in infants with this condition include involuntary muscle twitches (myoclonic seizures), sudden episodes of weak muscle tone (atonic seizures), partial or complete loss of consciousness (absence seizures), or loss of consciousness with muscle rigidity and convulsions (tonic-clonic seizures). Most people with STXBP1 encephalopathy with epilepsy have more than one type of seizure. In about one-quarter of individuals, the seizures are described as refractory because they do not respond to therapy with anti-epileptic medications. Other signs and symptoms of STXBP1 encephalopathy with epilepsy include intellectual disability that is often moderate to profound. Affected individuals also have delayed development of speech and walking; in some, these skills never fully develop. Movement and behavior disorders may also occur. Many affected individuals have feeding difficulties. In some cases, areas of brain tissue loss (atrophy) have been found on medical imaging. Frequency The prevalence of STXBP1 encephalopathy with epilepsy is unknown. At least 200 individuals with this condition have been described in the medical literature. -

Single-Cell Rnaseq Reveals Cell Adhesion Molecule Profiles in Electrophysiologically Defined Neurons
Single-cell RNAseq reveals cell adhesion molecule profiles in electrophysiologically defined neurons Csaba Földya,b,1, Spyros Darmanisc, Jason Aotoa,d, Robert C. Malenkae, Stephen R. Quakec,f, and Thomas C. Südhofa,f,1 aDepartment of Molecular and Cellular Physiology, Stanford University, Stanford, CA 94305; bBrain Research Institute, University of Zürich, 8057 Zurich, Switzerland; cDepartment of Bioengineering, Stanford University, Stanford, CA 94305; dDepartment of Pharmacology, University of Colorado Denver, Aurora, CO 80045; eNancy Pritzker Laboratory, Stanford University, Stanford, CA 94305; and fHoward Hughes Medical Institute, Stanford University, Stanford, CA 94305 Contributed by Thomas C. Südhof, July 10, 2016 (sent for review May 21, 2016; reviewed by Thomas Biederer, Tamas F. Freund, and Li-Huei Tsai) In brain, signaling mediated by cell adhesion molecules defines the neurexin (Nrxn1 and Nrxn3; presynaptic cell adhesion mole- identity and functional properties of synapses. The specificity of cules) isoforms were expressed cell type-specifically, with re- presynaptic and postsynaptic interactions that is presumably medi- markable consistency in respective cell types (9). We also found ated by cell adhesion molecules suggests that there exists a logic that that genetic deletion of neuroligin-3 (Nlgn3) (postsynaptic cell could explain neuronal connectivity at the molecular level. Despite its adhesion molecule) in PYR cells disabled tonic, cannabinoid importance, however, the nature of such logic is poorly understood, type 1 receptor-mediated, endocannabinoid signaling in RS CCK and even basic parameters, such as the number, identity, and single- synapses, but had no detectable phenotype in FS PV synapses cell expression profiles of candidate synaptic cell adhesion molecules, (10). Thus, although no systematic assessment of cell adhesion are not known.