Dynamic Carboniferous Tropical Forests: New Views of Plant Function and Potential for Physiological Forcing of Climate
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
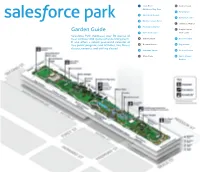
Salesforce Park Garden Guide
Start Here! D Central Lawn Children’s Play Area Garden Guide6 Palm Garden 1 Australian Garden Start Here! D Central Lawn Salesforce Park showcases7 California over Garden 50 species of Children’s Play Area 2 Mediterraneantrees and Basin over 230 species of understory plants. 6 Palm Garden -ã ¼ÜÊ ÊăØÜ ØÊèÜãE úØƀØÊèÃJapanese Maples ¼ÃØ Ê¢ 1 Australian Garden 3 Prehistoric¢ØÕ輫ÕØÊ£ØÂÜÃã«ó«ã«Üŧ¼«¹ĆãÃÜÜ Garden 7 California Garden ¼ÜÜÜŧÊÃØãÜŧÃØ¢ã«Ã£¼ÜÜÜũF Amphitheater Garden Guide 2 Mediterranean Basin 4 Wetland Garden Main Lawn E Japanese Maples Salesforce Park showcases over 50 species of 3 Prehistoric Garden trees and over 230 species of understory plants. A Oak Meadow 8 Desert Garden F Amphitheater It also offers a robust year-round calendar of 4 Wetland Garden Main Lawn free public programs and activities, like fitness B Bamboo Grove 9 Fog Garden Desert Garden classes, concerts, and crafting classes! A Oak Meadow 8 5 Redwood Forest 10 Chilean Garden B Bamboo Grove 9 Fog Garden C Main Plaza 11 South African 10 Chilean Garden Garden 5 Redwood Forest C Main Plaza 11 South African Garden 1 Children’s Australian Play Area Garden ABOUT THE GARDENS The botanist aboard the Endeavor, Sir Joseph Banks, is credited with introducing many plants from Australia to the western world, and many This 5.4 acre park has a layered soil system that plants today bear his name. balances seismic shifting, collects and filters storm- water, and irrigates the gardens. Additionally, the soil Native to eastern Australia, Grass Trees may grow build-up and dense planting help offset the urban only 3 feet in 100 years, and mature plants can be heat island effect by lowering the air temperature. -

A Comparative Study of the Primary Vascular System Of
Amer. J. Bot. 55(4): 464-472. 1!16'>. A COMPARATIVE STUDY OF THE PRIMARY VASCULAR SYSTE~1 OF CONIFERS. III. STELAR EVOLUTION IN GYMNOSPERMS 1 KADAMBARI K. NAMBOODIRI2 AND CHARLES B. BECK Department of Botany, University of Michigan, Ann Arbor ABST RAe T This paper includes a survey of the nature of the primary vascular system in a large number of extinct gymnosperms and progymnosperms. The vascular system of a majority of these plants resembles closely that of living conifers, being characterized, except in the most primitive forms which are protostelic, by a eustele consisting of axial sympodial bundles from which leaf traces diverge. The vascular supply to a leaf originates as a single trace with very few exceptions. It is proposed that the eustele in the gyrr.nosperms has evolved directly from the protostele by gradual medullation and concurrent separation of the peripheral conducting tissue into longitudinal sympodial bundles from which traces diverge radially. A subsequent modification results in divergence of traces in a tangential plane, The closed vascular system of conifers with opposite and whorled phyllotaxis, in which the vascular supply to a leaf originates as two traces which subsequently fuse, is considered to be derived from the open sympodial system characteristic of most gymnosperms. This hypothesis of stelar evolution is at variance with that of Jeffrey which suggests that the eustele of seed plants is derived by the lengthening and overlapping of leaf gaps in a siphonostele followed by further reduction in the resultant vascular bundles. This study suggests strongly that the "leaf gap" of conifers and other extant gymnosperms is not homologous with that of siphonostelic ferns and strengthens the validity of the view that Pterop sida is an unnatural group. -

International Organisation of Palaeobotany IOP NEWSLETTER
INTERNATIONAL UNION OF BIOLOGIC A L S C IENC ES S ECTION FOR P A L A EOBOTANY International Organisation of Palaeobotany IOP NEWSLETTER 110 August 2016 CONTENTS FROM THE SECRETARY/TREASURER IPC XIV/IOPC X 2016 IOPC 2020 IOP MEMBERSHIP IOP EXECUTIVE COMMITTEE ELECTIONS IOP WEBMASTER POSITION WHAT HAPPENED TO THE OUPH COLLECTIONS? THE PALAEOBOTANY OF ITALY UPCOMING MEETINGS CALL FOR NEWS and NOTES The views expressed in the newsletter are those of its correspondents, and do not necessarily reflect the policy of IOP. Please send us your contributions for the next edition of our newsletter (June 2016) by M ay 30th, 2016. President: Johanna Eder-Kovar (G ermany) Vice Presidents: Bob Spicer (Great Britain), Harufumi Nishida (Japan), M ihai Popa (Romania) M embers at Large: Jun W ang (China), Hans Kerp (Germany), Alexej Herman (Russia) Secretary/Treasurer/Newsletter editor: M ike Dunn (USA) Conference/Congress Chair: Francisco de Assis Ribeiro dos Santos IOP Logo: The evolution of plant architecture (© by A. R. Hemsley) I OP 110 2 August 2016 FROM THE In addition, please send any issues that you think need to be addressed at the Business SECRETARY/TREASURER meeting. I will add those to the Agenda. Dear IOP Members, Respectfully, Mike I am happy to report, that IOP seems to be on track and ready for a new Executive Council to take over. The elections are IPC XIV/IOPC X 2016 progressing nicely and I will report the results in the September/October Newsletter. The one area that is still problematic is the webmaster position. We really to talk amongst ourselves, and find someone who is willing and able to do the job. -

Cycad Species List
Mailing Address: E mail: [email protected] 3233 Brant St. Phone: (619) 291-4605 San Diego CA, 92103-5503 Jungle Music Fax: (619) 574-1595 Nursery Location: 450 Ocean View Ave. Palms & Cycads We’re on the Web! Encinitas CA www.junglemusic.net The palm and cycad source since 1977 Cycad Availability SPRING/SUMMER 2006 Directions Leucadia Blvd Take Freeway 5 to Encinitas (10 minutes south of Oceanside, 30 minutes north of San Diego). N Ocean View Ave Ave View Ocean e Exit Leucadia Blvd West (toward ocean). v A W E s Immediate left on Orpheus Ave, u e h left on Union St, p S r Right on Ocean View Ave O Union St Location: 450 Ocean View Ave, Encinitas, CA (next to the “Monarch Program”) The following is a listing of species available as of APRIL 2006. Call for sizes and prices available. Many species are extremely limited in availability. Some species may have sold out by the time you get this listing. Some cycad species are given as a “locality”. For visits to the nursery, always call first. Mail orders are possible to most areas within the United States with minimum order of $200 plus s/h. Thank you for your interest. Cycads are for U.S. domestic use only and cannot be shipped internationally. Phil Bergman, Owner The purpose of this listing is to give palm and cycad enthusiasts a list of the species offered at this time. Sizes and prices are not included herein as availability changes frequently and creating such a listing would be far too cumber- some. -

Prepared in Cooperation with the Lllinois State Museum, Springfield
Prepared in cooperation with the lllinois State Museum, Springfield Richard 1. Leary' and Hermann W. Pfefferkorn2 ABSTRACT The Spencer Farm Flora is a compression-impression flora of early Pennsylvanian age (Namurian B, or possibly Namurian C) from Brown County, west-central Illinois. The plant fossils occur in argillaceous siltstones and sand- stones of the Caseyville Formation that were deposited in a ravine eroded in Mississippian carbonate rocks. The plant-bearing beds are the oldest deposits of Pennsylva- nian age yet discovered in Illinois. They were formed be- fore extensive Pennsylvanian coal swamps developed. The flora consists of 29 species and a few prob- lematical forms. It represents an unusual biofacies, in which the generally rare genera Megalopteris, Lesleya, Palaeopteridium, and Lacoea are quite common. Noegger- athiales, which are seldom present in roof-shale floras, make up over 20 percent of the specimens. The Spencer Farm Flora is an extrabasinal (= "upland1') flora that was grow- ing on the calcareous soils in the vicinity of the ravine in which they were deposited. It is suggested here that the Noeggerathiales may belong to the Progymnosperms and that Noeggerathialian cones might be derived from Archaeopteris-like fructifica- tions. The cone genus Lacoea is intermediate between Noeggerathiostrobus and Discini tes in its morphology. Two new species, Lesleya cheimarosa and Rhodeop- teridi urn phillipsii , are described, and Gulpenia limbur- gensis is reported from North America for the first time. INTRODUCTION The Spencer Farm Flora (table 1) differs from other Pennsylvanian floras of the Illinois Basin. Many genera and species in the Spencer Farm Flora either have not been found elsewhere in the basin or are very l~uratorof Geology, Illinois State Museum, Springfield. -

New Paleobotanical Data on the Portuguese Pennsylvanian (Douro Carboniferous Basin, NW Portugal)
Versão online: http://www.lneg.pt/iedt/unidades/16/paginas/26/30/185 Comunicações Geológicas (2014) 101, Especial I, 409-414 IX CNG/2º CoGePLiP, Porto 2014 ISSN: 0873-948X; e-ISSN: 1647-581X New paleobotanical data on the Portuguese Pennsylvanian (Douro Carboniferous Basin, NW Portugal) Novos dados paleobotânicos do Pensilvaniano português (Bacia Carbonífera do Douro, NW Portugal) P. Correia1*, Z. Šimůnek2, J. Pšenička3, A. A. Sá4,5, R. Domingos6, A. Carneiro7, D. Flores1,7 Artigo Curto Short Article © 2014 LNEG – Laboratório Nacional de Geologia e Energia IP Abstract: This paper describes nine new macrofloral taxa from 1. Introduction Douro Carboniferous Basin (lower Gzhelian) of Portugal. The plant assemblage is mainly composed by pteridophylls (Sphenopteriss The fossil flora of Carboniferous of Portugal is still little arberi Kidston, Sphenopteris fayoli Zeiller, Sphenopteris tenuis known. The new megafloral occurrences recently found in Schenk, Odontopteris schlotheimii Brongniart), sphenopsids the Upper Pennsylvanian strata of Douro Carboniferous (Annularia spicata Gutbier, Stellotheca robusta (Feistmantel) Basin (DCB) provide new and important data about Surange and Prakash, Calamostachys grandis Zeiller (Jongmans) and Calamostachys calathifera Sterzel) besides the gymnosperm paleobotanical richness and diversity of the Paleozoic Cordaites foliolatus Grand`Eury. The new data provide a better floras of Portugal offering more information to previous understating of the knowledge of Late Carboniferous floras of researches reported from diverse localities and by different Portugal, showing the high plant diversity of Gzhelian floras, when authors (e.g. Wenceslau de Lima, Bernardino António considerable changes in paleogeography and climate dynamics are Gomes, Carlos Ribeiro, Carríngton da Costa, Carlos evidenced in Euramerican floristic assemblages. -

Tansley Review Evolution of Development of Vascular Cambia and Secondary Growth
New Phytologist Review Tansley review Evolution of development of vascular cambia and secondary growth Author for correspondence: Rachel Spicer1 and Andrew Groover2 Andrew Groover 1The Rowland Institute at Harvard, Cambridge, MA, USA; 2Institute of Forest Genetics, Pacific Tel: +1 530 759 1738 Email: [email protected] Southwest Research Station, USDA Forest Service, Davis, CA, USA Received: 29 December 2009 Accepted: 14 February 2010 Contents Summary 577 V. Evolution of development approaches for the study 587 of secondary vascular growth I. Introduction 577 VI. Conclusions 589 II. Generalized function of vascular cambia and their 578 developmental and evolutionary origins Acknowledgements 589 III. Variation in secondary vascular growth in angiosperms 581 References 589 IV. Genes and mechanisms regulating secondary vascular 584 growth and their evolutionary origins Summary New Phytologist (2010) 186: 577–592 Secondary growth from vascular cambia results in radial, woody growth of stems. doi: 10.1111/j.1469-8137.2010.03236.x The innovation of secondary vascular development during plant evolution allowed the production of novel plant forms ranging from massive forest trees to flexible, Key words: forest trees, genomics, Populus, woody lianas. We present examples of the extensive phylogenetic variation in sec- wood anatomy, wood formation. ondary vascular growth and discuss current knowledge of genes that regulate the development of vascular cambia and woody tissues. From these foundations, we propose strategies for genomics-based research in the evolution of development, which is a next logical step in the study of secondary growth. I. Introduction this pattern characterizes most extant forest trees, significant variation exists among taxa, ranging from extinct woody Secondary vascular growth provides a means of radially lycopods and horsetails with unifacial cambia (Cichan & thickening and strengthening plant axes initiated during Taylor, 1990; Willis & McElwain, 2002), to angiosperms primary, or apical growth. -

Two Stages of Late Carboniferous to Triassic Magmatism in the Strandja
Geological Magazine Two stages of Late Carboniferous to Triassic www.cambridge.org/geo magmatism in the Strandja Zone of Bulgaria and Turkey ł ń 1,2 3 1 1 Original Article Anna Sa aci ska , Ianko Gerdjikov , Ashley Gumsley , Krzysztof Szopa , David Chew4, Aleksandra Gawęda1 and Izabela Kocjan2 Cite this article: Sałacińska A, Gerdjikov I, Gumsley A, Szopa K, Chew D, Gawęda A, and 1Institute of Earth Sciences, Faculty of Natural Sciences, University of Silesia in Katowice, Będzińska 60, 41-200 Kocjan I. Two stages of Late Carboniferous to 2 3 Triassic magmatism in the Strandja Zone of Sosnowiec, Poland; Institute of Geological Sciences, Polish Academy of Sciences, Warsaw, Poland; Faculty of ‘ ’ Bulgaria and Turkey. Geological Magazine Geology and Geography, Sofia University St. Kliment Ohridski , 15 Tzar Osvoboditel Blvd., 1504 Sofia, Bulgaria 4 https://doi.org/10.1017/S0016756821000650 and Department of Geology, School of Natural Sciences, Trinity College Dublin, Dublin, Ireland Received: 9 February 2021 Abstract Revised: 3 June 2021 Accepted: 8 June 2021 Although Variscan terranes have been documented from the Balkans to the Caucasus, the southeastern portion of the Variscan Belt is not well understood. The Strandja Zone along Keywords: the border between Bulgaria and Turkey encompasses one such terrane linking the Strandja Zone; Sakar unit; U–Pb zircon dating; Izvorovo Pluton Balkanides and the Pontides. However, the evolution of this terrane, and the Late Carboniferous to Triassic granitoids within it, is poorly resolved. Here we present laser ablation Author for correspondence: – inductively coupled plasma – mass spectrometry (LA-ICP-MS) U–Pb zircon ages, coupled ł ń Anna Sa aci ska, with petrography and geochemistry from the Izvorovo Pluton within the Sakar Unit Email: [email protected] (Strandja Zone). -

The Geologic Time Scale Is the Eon
Exploring Geologic Time Poster Illustrated Teacher's Guide #35-1145 Paper #35-1146 Laminated Background Geologic Time Scale Basics The history of the Earth covers a vast expanse of time, so scientists divide it into smaller sections that are associ- ated with particular events that have occurred in the past.The approximate time range of each time span is shown on the poster.The largest time span of the geologic time scale is the eon. It is an indefinitely long period of time that contains at least two eras. Geologic time is divided into two eons.The more ancient eon is called the Precambrian, and the more recent is the Phanerozoic. Each eon is subdivided into smaller spans called eras.The Precambrian eon is divided from most ancient into the Hadean era, Archean era, and Proterozoic era. See Figure 1. Precambrian Eon Proterozoic Era 2500 - 550 million years ago Archaean Era 3800 - 2500 million years ago Hadean Era 4600 - 3800 million years ago Figure 1. Eras of the Precambrian Eon Single-celled and simple multicelled organisms first developed during the Precambrian eon. There are many fos- sils from this time because the sea-dwelling creatures were trapped in sediments and preserved. The Phanerozoic eon is subdivided into three eras – the Paleozoic era, Mesozoic era, and Cenozoic era. An era is often divided into several smaller time spans called periods. For example, the Paleozoic era is divided into the Cambrian, Ordovician, Silurian, Devonian, Carboniferous,and Permian periods. Paleozoic Era Permian Period 300 - 250 million years ago Carboniferous Period 350 - 300 million years ago Devonian Period 400 - 350 million years ago Silurian Period 450 - 400 million years ago Ordovician Period 500 - 450 million years ago Cambrian Period 550 - 500 million years ago Figure 2. -

Earliest Record of Megaphylls and Leafy Structures, and Their Initial Diversification
Review Geology August 2013 Vol.58 No.23: 27842793 doi: 10.1007/s11434-013-5799-x Earliest record of megaphylls and leafy structures, and their initial diversification HAO ShouGang* & XUE JinZhuang Key Laboratory of Orogenic Belts and Crustal Evolution, School of Earth and Space Sciences, Peking University, Beijing 100871, China Received January 14, 2013; accepted February 26, 2013; published online April 10, 2013 Evolutionary changes in the structure of leaves have had far-reaching effects on the anatomy and physiology of vascular plants, resulting in morphological diversity and species expansion. People have long been interested in the question of the nature of the morphology of early leaves and how they were attained. At least five lineages of euphyllophytes can be recognized among the Early Devonian fossil plants (Pragian age, ca. 410 Ma ago) of South China. Their different leaf precursors or “branch-leaf com- plexes” are believed to foreshadow true megaphylls with different venation patterns and configurations, indicating that multiple origins of megaphylls had occurred by the Early Devonian, much earlier than has previously been recognized. In addition to megaphylls in euphyllophytes, the laminate leaf-like appendages (sporophylls or bracts) occurred independently in several dis- tantly related Early Devonian plant lineages, probably as a response to ecological factors such as high atmospheric CO2 concen- trations. This is a typical example of convergent evolution in early plants. Early Devonian, euphyllophyte, megaphyll, leaf-like appendage, branch-leaf complex Citation: Hao S G, Xue J Z. Earliest record of megaphylls and leafy structures, and their initial diversification. Chin Sci Bull, 2013, 58: 27842793, doi: 10.1007/s11434- 013-5799-x The origin and evolution of leaves in vascular plants was phology and evolutionary diversification of early leaves of one of the most important evolutionary events affecting the basal euphyllophytes remain enigmatic. -

A New Microsporangiate Organ from the Lower Carboniferous of the Novgorod Region, Russia O
ISSN 0031-0301, Paleontological Journal, 2009, Vol. 43, No. 10, pp. 1316–1329. © Pleiades Publishing, Ltd., 2009. A New Microsporangiate Organ from the Lower Carboniferous of the Novgorod Region, Russia O. A. Orlovaa, N. R. Meyer-Melikian†,a, and N. E. Zavialovab a Moscow State University (MGU), Leninskie gory 1, Moscow, 119991 Russia b Borissiak Paleontological Institute of the Russian Academy of Sciences, ul. Profsoyuznaya 123, Moscow, 117997 Russia e-mail: [email protected] Received February 5, 2008 Abstract—A new species of the genus Telangiopsis, T. nonnae O. Orlova et Zavialova, was described on the basis of a microsporangiate organ from the Lower Carboniferous deposits of the Novgorod Region. The mor- phology of branching fertile axes, synangia, and sporangia was thoroughly studied. The three-dimensional sys- tem of fertile axes branches monopodially; ultimate axes bear numerous connivent bunches of synangia, which consist of three to six basally fused elongated ovate sporangia. The morphology and ultrastructure of prepollen grains were studied, which were extracted from the rock matrix surrounding the sporangia. The two-layered exine includes a well-developed endexine and an alveolate ectexine, with one–three rows of large thin-walled alveolae. The new species was compared with other Early Carboniferous microsporangiate organs. Key words: Early Carboniferous, Novgorod Region, fertile axis, synangia, Lyginopteridales, trilete prepollen, exine ultrastructure. DOI: 10.1134/S003103010910013X INTRODUCTION synangia and numerous casts and imprints of detached synangia were found in yellow ferruginous sandstone. During the three last decades, the interest of bota- In addition to the fertile axes, sterile remains of Lygi- nists dealing with fossil and modern plants to Carbon- nopteridales, Medullosales, and Calamopytiales were iferous synangiate pollen organs has considerably found (Orlova and Snigirevskii, 2003, 2004). -

JUDD W.S. Et. Al. (2002) Plant Systematics: a Phylogenetic Approach. Chapter 7. an Overview of Green
UNCORRECTED PAGE PROOFS An Overview of Green Plant Phylogeny he word plant is commonly used to refer to any auto- trophic eukaryotic organism capable of converting light energy into chemical energy via the process of photosynthe- sis. More specifically, these organisms produce carbohydrates from carbon dioxide and water in the presence of chlorophyll inside of organelles called chloroplasts. Sometimes the term plant is extended to include autotrophic prokaryotic forms, especially the (eu)bacterial lineage known as the cyanobacteria (or blue- green algae). Many traditional botany textbooks even include the fungi, which differ dramatically in being heterotrophic eukaryotic organisms that enzymatically break down living or dead organic material and then absorb the simpler products. Fungi appear to be more closely related to animals, another lineage of heterotrophs characterized by eating other organisms and digesting them inter- nally. In this chapter we first briefly discuss the origin and evolution of several separately evolved plant lineages, both to acquaint you with these important branches of the tree of life and to help put the green plant lineage in broad phylogenetic perspective. We then focus attention on the evolution of green plants, emphasizing sev- eral critical transitions. Specifically, we concentrate on the origins of land plants (embryophytes), of vascular plants (tracheophytes), of 1 UNCORRECTED PAGE PROOFS 2 CHAPTER SEVEN seed plants (spermatophytes), and of flowering plants dons.” In some cases it is possible to abandon such (angiosperms). names entirely, but in others it is tempting to retain Although knowledge of fossil plants is critical to a them, either as common names for certain forms of orga- deep understanding of each of these shifts and some key nization (e.g., the “bryophytic” life cycle), or to refer to a fossils are mentioned, much of our discussion focuses on clade (e.g., applying “gymnosperms” to a hypothesized extant groups.