Peracetic Acid
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

UC San Diego Electronic Theses and Dissertations
UC San Diego UC San Diego Electronic Theses and Dissertations Title Vanadium-dependent bromoperoxidase in a marine Synechococcus / Permalink https://escholarship.org/uc/item/34x4t8rp Author Johnson, Todd Laurel Publication Date 2013 Peer reviewed|Thesis/dissertation eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA, SAN DIEGO Vanadium-dependent bromoperoxidase in a marine Synechococcus A dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Marine Biology by Todd L. Johnson Committee in charge: Brian Palenik, Chair Bianca Brahamsha, Co-Chair Lihini Aluwihare James Golden Jens Mühle Bradley Moore 2013 Copyright Todd L. Johnson, 2013 All rights reserved. The dissertation of Todd L. Johnson is approved, and it is acceptable in quality and form for publication on microfilm and electronically: ________________________________________________________ ________________________________________________________ ________________________________________________________ ________________________________________________________ ________________________________________________________ Co-Chair ________________________________________________________ Chair University of California, San Diego 2013 iii DEDICATION To Janet, Tim, and Andrew Johnson, for unconditional love and support. iv TABLE OF CONTENTS Signature Page……………………………………………………………………………iii Dedication ………………………………………………………………………………..iv Table of Contents………………………………………………………………………….v List -

Sodium Percarbonate: a Versatile Oxidizing Reagent
SPOTLIGHT 2969 SYNLETT Sodium Percarbonate: A Versatile Spotlight 335 Oxidizing Reagent Compiled by Nadiya Koukabi This feature focuses on a re- Nadiya Koukabi was born in Shiraz, Iran. She received her B.Sc. agent chosen by a postgradu- (2000) in Chemistry from the Islamic Azad University Firouzabad ate, highlighting the uses and Branch and her M.Sc. in Organic Chemistry from the Bu-Ali Sina preparation of the reagent in University under the guidance of Prof. M. A. Zolfigol. She is pres- current research ently working toward her Ph.D. degree under the supervision of Prof. A. Khazaei and Prof. M. A. Zolfigol. Her research interests fo- cuse on the development of new reagents and catalysts in organic reactions. Faculty of Chemistry, Bu-Ali Sina University, Hamedan 6517838683, Iran E-mail: [email protected] Introduction available and furthermore, it is more risky to handle. Con- sequently, the ability of SPC to release oxidative species Sodium percarbonate (SPC, Na2CO3·1.5H2O) is an inor- in an organic medium has made it an useful reagent in or- ganic, inexpensive, environmentally friendly, stable, and ganic synthesis.8–11 easily handled reagent that has an excellent shelf life. The + Na H H H H – O O O O O name ‘sodium percarbonate’ does not reflect the structure O + O Na H O O or true nature of the material. Its erroneous name is due to + O Na – H H O 1,2 H successive confusions in its structure. The structure of – O O + H Na + Na – SPC (Figure 1) has been determined, the cohesion of the – O O O O O H – + O Na adduct being due to hydrogen bonding between carbonate O – – – O O 3,4 O O + ions and hydrogen peroxide molecules. -

The Decomposition Kinetics of Peracetic Acid and Hydrogen Peroxide in Municipal Wastewaters
Disinfection Forum No 10, October 2015 The Decomposition Kinetics of Peracetic Acid and Hydrogen Peroxide in Municipal Wastewaters INTRODUCTION Efficient control of microbial populations in municipal wastewater using peracetic acid (PAA) requires an understanding of the PAA decomposition kinetics. This knowledge is critical to ensure the proper dosing of PAA needed to achieve an adequate concentration within the contact time of the disinfection chamber. In addition, the impact of PAA on the environment, post-discharge into the receiving water body, also is dependent upon the longevity of the PAA in the environment, before decomposing to acetic acid, oxygen and water. As a result, the decomposition kinetics of PAA may have a significant impact on aquatic and environmental toxicity. PAA is not manufactured as a pure compound. The solution exists as an equilibrium mixture of PAA, hydrogen peroxide, acetic acid, and water: ↔ + + Acetic Acid Hydrogen Peroxide Peracetic Acid Water PeroxyChem’s VigorOx® WWT II Wastewater Disinfection Technology contains 15% peracetic acid by weight and 23% hydrogen peroxide as delivered. Although hydrogen peroxide is present in the formulation, peracetic acid is considered to be the active component for disinfection1 in wastewater. There have been several published studies investigating the decomposition kinetics of PAA in different water matrices, including municipal wastewater2-7. Yuan7 states that PAA may be consumed in the following three competitive reactions: 1. Spontaneous decomposition 2 CH3CO3H à 2 CH3CO2H + O2 Eq (1) 2. Hydrolysis CH3CO3H + H2O à CH3CO2H + H2O2 Eq (2) 3. Transition metal catalyzed decomposition + CH3CO3H + M à CH3CO2H + O2 + other products Eq (3) At neutral pH’s, both peracetic acid and hydrogen peroxide can be rapidly consumed by these reactions7 (hydrogen peroxide will decompose to water and oxygen via 2H2O2 à 2H2O + O2). -

Peracetic Acid Processing
Peracetic Acid Processing Identification Chemical Name(s): CAS Number: peroxyacetic acid, ethaneperoxic acid 79-21-0 Other Names: Other Codes: per acid, periacetic acid, PAA NIOSH Registry Number: SD8750000 TRI Chemical ID: 000079210 UN/ID Number: UN3105 Summary Recommendation Synthetic / Allowed or Suggested Non-Synthetic: Prohibited: Annotation: Synthetic Allowed (consensus) Allowed only for direct food contact for use in wash water. Allowed as a (consensus) sanitizer on surfaces in contact with organic food. (consensus) From hydrogen peroxide and fermented acetic acid sources only. (Not discussed by processing reviewers--see discussion of source under Crops PAA TAP review.) Characterization Composition: C2H4O3. Peracetic acid is a mixture of acetic acid (CH3COOH) and hydrogen peroxide (H2O2) in an aqueous solution. Acetic acid is the principle component of vinegar. Hydrogen peroxide has been previously recommended by the NOSB for the National List in processing (synthetic, allowed at Austin, 1995). Properties: It is a very strong oxidizing agent and has stronger oxidation potential than chlorine or chlorine dioxide. Liquid, clear, and colorless with no foaming capability. It has a strong pungent acetic acid odor, and the pH is acid (2.8). Specific gravity is 1.114 and weighs 9.28 pounds per gallon. Stable upon transport. How Made: Peracetic acid (PAA) is produced by reacting acetic acid and hydrogen peroxide. The reaction is allowed to continue for up to ten days in order to achieve high yields of product according to the following equation. O O || || CH3-C-OH + H2O2 CH3C-O-OH + H2O acetic acid hydrogen peroxyacetic peroxide acid Due to reaction limitations, PAA generation can be up to 15% with residual levels of hydrogen peroxide (up to 25%) and acetic acid (up to 35%) with water up to 25%. -

Fatty Acids: Essential…Therapeutic
Volume 3, No.2 May/June 2000 A CONCISE UPDATE OF IMPORTANT ISSUES CONCERNING NATURAL HEALTH INGREDIENTS Written and Edited By: Thomas G. Guilliams Ph.D. FATTY ACIDS: Essential...Therapeutic Few things have been as confusing to both patient and health care provider as the issue of fats and oils. Of all the essential nutrients required for optimal health, fatty acids have not only been forgotten they have been considered hazardous. Health has somehow been equated with “low-fat” or “fat-free” for so long, to suggest that fats could be essential or even therapeutic is to risk credibility. We hope to give a view of fats that is both balanced and scientific. This review will cover the basics of most fats that will be encountered in dietary or supplemental protocols. Recommendations to view essential fatty acids in a similar fashion as essential vitamins and minerals will be combined with therapeutic protocols for conditions ranging from cardiovascular disease, skin conditions, diabetes, nerve related disorders, retinal disorders and more. A complete restoration of health cannot be accomplished until there is a restoration of fatty acid nutritional information among health care professionals and their patients. Fats- What are they? Dietary fats come to us from a variety of sources, but primarily in the form of triglycerides. That is, three fatty acid molecules connected by a glycerol backbone (see fatty acid primer page 3 for diagram). These fatty acids are then used as energy by our cells or modified into phospholipids to be used as cell or organelle membranes. Some fatty acids are used in lipoprotein molecules to shuttle cholesterol and fats to and from cells, and fats may also be stored for later use. -

Relationship Between Dietary Intake of Fatty Acids and Disease Activity in Pediatric Inflammatory Bowel Disease Patients
Relationship between Dietary Intake of Fatty Acids and Disease Activity in Pediatric Inflammatory Bowel Disease Patients A thesis submitted to the Graduate School of the University of Cincinnati in partial fulfillment of the requirements for the degree of Master of Science in the Department of Nutrition of the College of Allied Health Sciences by Michael R. Ciresi B.S. The Ohio State University June 2008 Committee Chair: Grace Falciglia, Ph.D. Abstract Background. Crohn’s disease (CD) and ulcerative colitis (UC), collectively known as inflammatory bowel disease (IBD), are chronic illnesses that affect predominately the gastrointestinal tract. The pathogenesis and etiology remain unclear but the importance of environmental factors, in particular diet, is evidenced by the increased incidence rates of the recent decades that genetic inheritance cannot account for. In particular, the quantity of fatty acid consumption has been consistently linked with IBD risk. While several studies have investigated the connections between diet, etiology, signs and symptoms associated with IBD, very few have explored the relationship between disease state and specific fatty acid intake in the pediatric IBD population. Methods. In this cross-sectional study, 100 pediatric patients from Cincinnati Children’s Hospital and the Hospital for Sick Children in Toronto with diagnosed IBD (73 with Crohn’s disease (CD) and 27 with ulcerative colitis (UC)) were included. Three-day diet records were collected from the patients for the assessment of their dietary intake. The abbreviated Pediatric Crohn’s Disease Activity Index (PCDAI), the abbreviated Ulcerative Colitis Activity Index (PUCAI), and markers of inflammation (lipopolysaccharide binding protein (LBP) and S100A12) were used to assess disease severity. -

(12) United States Patent (10) Patent No.: US 8,187,615 B2 Friedman (45) Date of Patent: May 29, 2012
US008187615B2 (12) United States Patent (10) Patent No.: US 8,187,615 B2 Friedman (45) Date of Patent: May 29, 2012 (54) NON-AQUEOUS COMPOSITIONS FOR ORAL 6,054,136 A 4/2000 Farah et al. DELIVERY OF INSOLUBLE BOACTIVE 6,140,375 A 10/2000 Nagahama et al. AGENTS 2003/O149061 A1* 8/2003 Nishihara et al. .......... 514,266.3 FOREIGN PATENT DOCUMENTS (76) Inventor: Doron Friedman, Karme-Yosef (IL) GB 2222770 A 3, 1990 JP 2002-121929 5, 1990 (*) Notice: Subject to any disclaimer, the term of this WO 96,13273 * 5/1996 patent is extended or adjusted under 35 WO 200056346 A1 9, 2000 U.S.C. 154(b) by 1443 days. OTHER PUBLICATIONS (21) Appl. No.: 10/585,298 Pouton, “Formulation of Self-Emulsifying Drug Delivery Systems' Advanced Drug Delivery Reviews, 25:47-58 (1997). (22) PCT Filed: Dec. 19, 2004 Lawrence and Rees, “Microemulsion-based media as novel drug delivery systems' Advanced Drug Delivery Reviews, 45:89-121 (86). PCT No.: PCT/L2004/OO1144. (2000). He et al., “Microemulsions as drug delivery systems to improve the S371 (c)(1), solubility and the bioavailability of poorly water-soluble drugs' (2), (4) Date: Jul. 6, 2006 Expert Opin. Drug Deliv. 7:445-460 (2010). Prajpati et al. “Effect of differences in Fatty Acid Chain Lengths of (87) PCT Pub. No.: WO2005/065652 Medium-Chain Lipids on Lipid/Surfactant/Water Phase Diagrams PCT Pub. Date: Jul. 21, 2005 and Drug Solubility” J. Excipients and Food Chem, 2:73-88 (2011). (65) Prior Publication Data * cited by examiner US 2007/O190O80 A1 Aug. -

Valorization of Glycerol Through the Enzymatic Synthesis of Acylglycerides with High Nutritional Value
catalysts Article Valorization of Glycerol through the Enzymatic Synthesis of Acylglycerides with High Nutritional Value Daniel Alberto Sánchez 1,3,* , Gabriela Marta Tonetto 1,3 and María Luján Ferreira 2,3 1 Departamento de Ingeniería Química, Universidad Nacional del Sur (UNS), Bahía Blanca 8000, Argentina; [email protected] 2 Departamento de Química, Universidad Nacional del Sur (UNS), Bahía Blanca 8000, Argentina; [email protected] 3 Planta Piloto de Ingeniería Química–PLAPIQUI (UNS-CONICET), Bahía Blanca 8000, Argentina * Correspondence: [email protected]; Tel.: +54-291-4861700 Received: 8 December 2019; Accepted: 1 January 2020; Published: 14 January 2020 Abstract: The production of specific acylglycerides from the selective esterification of glycerol is an attractive alternative for the valorization of this by-product of the biodiesel industry. In this way, products with high added value are generated, increasing the profitability of the overall process and reducing an associated environmental threat. In this work, nutritional and medically interesting glycerides were obtained by enzymatic esterification through a two-stage process. In the first stage, 1,3-dicaprin was obtained by the regioselective esterification of glycerol and capric acid mediated by the commercial biocatalyst Lipozyme RM IM. Under optimal reaction conditions, 73% conversion of fatty acids and 76% selectivity to 1,3-dicaprin was achieved. A new model to explain the participation of lipase in the acyl migration reaction is presented. It evaluates the conditions in the microenvironment of the active site of the enzyme during the formation of the tetrahedral intermediate. In the second stage, the esterification of the sn-2 position of 1,3-dicaprin with palmitic acid was performed using the lipase from Burkholderia cepacia immobilized on chitosan as the biocatalyst. -

Great People Great Products Perfect Chemistry Oci
OCI CHEMICAL CORPORATION GREAT PEOPLE GREAT PRODUCTS PERFECT CHEMISTRY OCI Chemical is a subsidiary of OCI Company Ltd., a leading chemical manufacturer and publicly traded company on the Korean Exchange. OCI Company has five primary business divisions: renewable energy, inorganic chemicals, petro and coal chemicals, fine chemicals and insulation. OCI Chemical is an integral part of the inorganic chemicals division and a major contributor to this division’s sales and profits. Since 1962, OCI Chemical’s plant in Green River, Wyoming has been providing high quality soda ash to companies across North America and around the globe. While we are still among the world’s largest producers of soda ash, OCI Chemical has also become a leading producer of sodium percarbonate (PC) and hydrogen peroxide. A GLOBAL LEADER A GLOBAL OCI Chemical is committed to excellence and safety in all aspects of business. Every OCI employee, from the plants in Green River, Decatur and Columbus to our corporate headquarters in Atlanta, is empowered to modify any operation that poses a potential threat to themselves, their fellow workers, the surrounding community or the environment. Our customers know they can count on us for more than just quality chemicals. They know OCI was built on strong values, strong character and a passion for excellence. We know that great people and great products equal perfect chemistry. OCI Wyoming, L.P. Green River, Wyoming Soda Ash Mining and Production Stauffer Chemical opens Big THROUGH THE YEARS THROUGH THE Island Mine and Refinery in Green River, WY and produces 1962 soda ash from the mined trona. -

Peracetic Acid
Regulation (EU) No 528/2012 concerning the making available on the market and use of biocidal products Evaluation of active substances Assessment Report Peracetic acid Product-types 11 and 12 (Preservatives for liquid cooling and processing systems) (Slimicides) August 2016 Finland Peracetic acid Product-types 11 and 12 August 2016 CONTENTS 1. STATEMENT OF SUBJECT MATTER AND PURPOSE ........................................................ 2 1.1. Procedure followed ............................................................................................. 2 1.2. Purpose of the assessment report ....................................................................... 2 2. OVERALL SUMMARY AND CONCLUSIONS ..................................................................... 4 2.1. Presentation of the Active Substance .................................................................. 4 2.1.1. Identity, Physico-Chemical Properties & Methods of Analysis ................. 4 2.1.2. Intended Uses and Efficacy ..................................................................... 7 2.1.3. Classification and Labelling ..................................................................... 8 2.2. Summary of the Risk Assessment ....................................................................... 9 2.2.1. Human Health Risk Assessment .............................................................. 9 2.2.1.1. Hazard identification ................................................................................... 9 2.2.1.2. Effects assessment ................................................................................... -
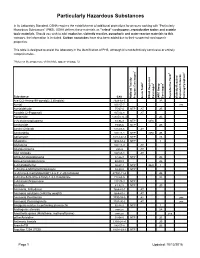
Particularly Hazardous Substances
Particularly Hazardous Substances In its Laboratory Standard, OSHA requires the establishment of additional protections for persons working with "Particularly Hazardous Substances" (PHS). OSHA defines these materials as "select" carcinogens, reproductive toxins and acutely toxic materials. Should you wish to add: explosive, violently reactive, pyrophoric and water-reactve materials to this category, the information is included. Carbon nanotubes have also been added due to their suspected carcinogenic properties. This table is designed to assist the laboratory in the identification of PHS, although it is not definitively conclusive or entirely comprehensive. *Notes on the proper use of this table appear on page 12. 1 6 5 2 3 4 Substance CAS National Toxicity National Program Carcinogen Toxin Acute Regulated OSHA Carcinogen Group IARC Carcinogen Toxin Reproductive Violently Reactive/ Explosive/Peroxide Forming/Pyrophoric A-a-C(2-Amino-9H-pyrido[2,3,b]indole) 2648-68-5 2B Acetal 105-57-7 yes Acetaldehyde 75-07-0 NTP AT 2B Acrolein (2-Propenal) 107-02-8 AT Acetamide 126850-14-4 2B 2-Acetylaminofluorene 53-96-3 NTP ORC Acrylamide 79-06-6 NTP 2B Acrylyl Chloride 814-68-6 AT Acrylonitrile 107-13-1 NTP ORC 2B Adriamycin 23214-92-8 NTP 2A Aflatoxins 1402-68-2 NTP 1 Allylamine 107-11-9 AT Alkylaluminums varies AT Allyl Chloride 107-05-1 AT ortho-Aminoazotoluene 97-56-3 NTP 2B para-aminoazobenzene 60-09-3 2B 4-Aminobiphenyl 92-67-1 NTP ORC 1 1-Amino-2-Methylanthraquinone 82-28-0 NTP (2-Amino-6-methyldipyrido[1,2-a:3’,2’-d]imidazole) 67730-11-4 2B -

Methods and Compositions for the Treatment of Infection and Control of Flora Using Haloperoxidase
Europaisches Patentamt 19 European Patent Office Office europeen des brevets © Publication number : 0 500 387 A2 12 EUROPEAN PATENT APPLICATION (2j) Application number : 92301448.4 6i) int. ci.5: A61K 37/50 (22) Date of filing : 21.02.92 (30) Priority: 21.02.91 US 660994 (72) Inventor : Allen, Robert Charles 3215 Woodcrest San Antonio, Texas 78209 (US) (43) Date of publication of application 26.08.92 Bulletin 92/35 @) Representative : Sheard, Andrew Gregory et al Kilburn & Strode 30, John Street @ Designated Contracting States : London WC1N 2DD (GB) AT BE CH DE DK ES FR GB GR IT LI LU MC NL PT SE © Applicant : EXOXEMIS, INC. 18585 Sigma Road, Suite 100 San Antonio, Texas 78209 (US) (54) Methods and compositions for the treatment of infection and control of flora using haloperoxidase. (57) Haloperoxidases are used to selectively bind to and, in the presence of peroxide and halide, inhibit the growth of target microbes without eliminating desirable microbes or significantly damaging other components, such as host cells, in the environment of the target microbe. When a target microbe, e.g., a pathogenic microbe, has a binding capacity for haloperoxidase greater than that of a desired microbe, e.g., members of the normal flora, the target microbe selectively binds the haloperoxidase with little or no binding of the haloperoxidase by the desired microbe. In the presence of peroxide and halide, the target bound haloperoxidase catalyzes halide oxidation and facilitates the disproportionation of peroxide to singlet molecular oxygen at the surface of the target microbe. The lifetime of singlet molecular oxygen restricts damage to the surface resulting in selective killing of the target microbe with a minimum of collateral damage to the desired microbe or physiological medium.