Alkali Metal Actinide Complex Halides : Thermochemical and Structural Considerations J
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

IA Metals: Alkali Metals
IA Metals: Alkali Metals INTRODUCTION: The alkali metals are a group in the periodic table consisting of the chemical elements lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs). You should remember that there is a separate group called the alkaline earth metals in Group Two. They are a very different family, even though they have a similar name. The seventh member of alkali metals group – francium, is radioactive and so rare that only 20 atoms of francium may exist on Earth at any given moment. The term alkali is derived from an Arabic word meaning “ashes.” PHYSICAL PROPERTIES: Shiny Soft (They can all be cut easily with a knife ) Highly reactive at standard temperature and pressure Because of their high reactivity, they must be stored under oil to prevent reaction with air Their density increases as we move from Li to F White/metal coloured Very good conductors of heat and electricity Have the ability to impart colour to the flame. This property of alkali metals is used in their identification. CHEMICAL PROPERTIES: The atom of any given alkali metal has only one valence electron. The chemical reactivity of alkali metals increase as we move from the top to the bottom of the group. Like any other metals, ionization potential is very low. In fact, alkali metals have the lowest ionization potential among the elements of any given period of the periodic table. Any alkali metal when comes in contact with air or oxygen, starts burning and oxides are formed in the process. At the end of the chemical reaction, lithium gives lithium monoxide (LiO), sodium gives sodium peroxide (Na2O2) and other alkali metals give superoxides. -

An Alternate Graphical Representation of Periodic Table of Chemical Elements Mohd Abubakr1, Microsoft India (R&D) Pvt
An Alternate Graphical Representation of Periodic table of Chemical Elements Mohd Abubakr1, Microsoft India (R&D) Pvt. Ltd, Hyderabad, India. [email protected] Abstract Periodic table of chemical elements symbolizes an elegant graphical representation of symmetry at atomic level and provides an overview on arrangement of electrons. It started merely as tabular representation of chemical elements, later got strengthened with quantum mechanical description of atomic structure and recent studies have revealed that periodic table can be formulated using SO(4,2) SU(2) group. IUPAC, the governing body in Chemistry, doesn‟t approve any periodic table as a standard periodic table. The only specific recommendation provided by IUPAC is that the periodic table should follow the 1 to 18 group numbering. In this technical paper, we describe a new graphical representation of periodic table, referred as „Circular form of Periodic table‟. The advantages of circular form of periodic table over other representations are discussed along with a brief discussion on history of periodic tables. 1. Introduction The profoundness of inherent symmetry in nature can be seen at different depths of atomic scales. Periodic table symbolizes one such elegant symmetry existing within the atomic structure of chemical elements. This so called „symmetry‟ within the atomic structures has been widely studied from different prospects and over the last hundreds years more than 700 different graphical representations of Periodic tables have emerged [1]. Each graphical representation of chemical elements attempted to portray certain symmetries in form of columns, rows, spirals, dimensions etc. Out of all the graphical representations, the rectangular form of periodic table (also referred as Long form of periodic table or Modern periodic table) has gained wide acceptance. -

Suppression Mechanisms of Alkali Metal Compounds
SUPPRESSION MECHANISMS OF ALKALI METAL COMPOUNDS Bradley A. Williams and James W. Fleming Chemistry Division, Code 61x5 US Naval Research Lnhoratory Washington, DC 20375-5342, USA INTRODUCTION Alkali metal compounds, particularly those of sodium and potassium, are widely used as fire suppressants. Of particular note is that small NuHCOi particles have been found to be 2-4 times more effective by mass than Halon 1301 in extinguishing both eountertlow flames [ I] and cup- burner flames [?]. Furthermore, studies in our laboratory have found that potassium bicarbonate is some 2.5 times more efficient by weight at suppression than sodium bicarhonatc. The primary limitation associated with the use of alkali metal compounds is dispersal. since all known compounds have very low volatility and must he delivered to the fire either as powders or in (usually aqueous) solution. Although powders based on alkali metals have been used for many years, their mode of effective- ness has not generally been agreed upon. Thermal effects [3],namely, the vaporization of the particles as well as radiative energy transfer out of the flame. and both homogeneous (gas phase) and heterogeneous (surface) chemistry have been postulated as mechanisms by which alkali metals suppress fires [4]. Complicating these issues is the fact that for powders, particle size and morphology have been found to affect the suppression properties significantly [I]. In addition to sodium and potassium, other alkali metals have been studied, albeit to a consider- ably lesser extent. The general finding is that the suppression effectiveness increases with atomic weight: potassium is more effective than sodium, which is in turn more effective than lithium [4]. -

25 WORDS CHLORINE Chlorine, Cl, Is a Very Poisonous Green Gas That's
25 WORDS CHLORINE Chlorine, Cl, is a very poisonous green gas that's extremely reactive. It's used for sanitizing, purifying, and was used as a weapon during World War I by the Germans. But in chemistry, it is an oxidizer. Chlorine, Cl, is a green gaseous element with an atomic number of 17. This halogen is a powerful oxidant and used to produce many things, such as cleaning products. Chlorine; it's chemical symbol is Cl. Chloride is abundant in nature and necessary for life but a large amount can cause choking and and poisoning. It's mainly used for water purification but has other uses. Chlorine is a halogen and to test if it has a halogen, we use the Beilstein Copper Wire Test. It is also used to produce safe drinking water. Chlorine, atomic number seventeen, is a halogen that is found in table salt, NaCl, making it essential to life. However, pure chlorine, Cl2, is a poisonous gas, detectable at even 1 ppm. Chlorine, (Symbol Cl), belongs to the halogen family of elements, found in group 17 on the periodic table. Chlorine has an atomic number of 17 and atomic weight of 35.453. Chlorine is the 17th element on the periodic table, and is in the "Halogens" group, which has a tendency to gain one electron to form anions. Its anion can be found commonly in table salt Chlorine (symbolized Cl) is the chemical element with atomic number 17. Clorine is a powerful oxidant and is used in bleaching and disinfectants. It is a pale yellow-green gas that has a specific strong smell. -

Actinide Ground-State Properties-Theoretical Predictions
Actinide Ground-State Properties Theoretical predictions John M. Wills and Olle Eriksson electron-electron correlations—the electronic energy of the ground state of or nearly fifty years, the actinides interactions among the 5f electrons and solids, molecules, and atoms as a func- defied the efforts of solid-state between them and other electrons—are tional of electron density. The DFT Ftheorists to understand their expected to affect the bonding. prescription has had such a profound properties. These metals are among Low-symmetry crystal structures, impact on basic research in both the most complex of the long-lived relativistic effects, and electron- chemistry and solid-state physics that elements, and in the solid state, they electron correlations are very difficult Walter Kohn, its main inventor, was display some of the most unusual to treat in traditional electronic- one of the recipients of the 1998 behaviors of any series in the periodic structure calculations of metals and, Nobel Prize in Chemistry. table. Very low melting temperatures, until the last decade, were outside the In general, it is not possible to apply large anisotropic thermal-expansion realm of computational ability. And DFT without some approximation. coefficients, very low symmetry crystal yet, it is essential to treat these effects But many man-years of intense research structures, many solid-to-solid phase properly in order to understand the have yielded reliable approximate transitions—the list is daunting. Where physics of the actinides. Electron- expressions for the total energy in does one begin to put together an electron correlations are important in which all terms, except for a single- understanding of these elements? determining the degree to which 5f particle kinetic-energy term, can be In the last 10 years, together with electrons are localized at lattice sites. -

Driving Halogen Lamps Application Note
Application Note 1604 Driving Halogen Lamps Abstract: This application note looks at the suitability of the Ultimod wide trim powerMods for applications driving Halogen lamps. An incandescent lamp generates light by heating is that the powerMod will go into a protective a tungsten wire or filament until it glows (at current limit. The characteristics of this current around 2,500 ºC) by passing an electric current limit are shown in Figure 1 below. through it. A halogen lamp is basically a modified version of an incandescent lamp. The difference is that the bulb of a halogen lamp has a small amount of a halogen gas added. The presence of this halogen in the bulb produces a chemical reaction (known as the halogen cycle) that redeposists tungsten evaporated by heating back onto the filament. In a standard incandescent the constant evaporation leads to the eventual failure of the lamp as the filament progressively thins and breaks or “burns out”. Since in a halogen lamp the tungsten is redeposited back on the filament Figure 1 Current Limit Characteristics its lifetime is extended, and it also be heated to a higher temperature (in the region of 3,000 ºC), which increases its efficiency. You can see from Figure 1 that when we increase the load (by reducing the loads The high operational temperature of the filament resistance) and the current increases above the results in a challenge for a constant voltage set current limit of the modules we enter a mode power supplies like the Ultimod due to the of operation known as straight line current different resistance of the tungsten filament limiting where the current is held constant and at room temperature and its resistance at 3,000 the voltage is reduced. -

Largest Mixed Transition Metal/Actinide Cluster: a Bimetallic Mn/Th Complex with A
Inorg. Chem. 2006, 45, 2364−2366 Largest Mixed Transition Metal/Actinide Cluster: A Bimetallic Mn/Th 18+ Complex with a [Mn10Th6O22(OH)2] Core Abhudaya Mishra, Khalil A. Abboud, and George Christou* Department of Chemistry, UniVersity of Florida, GainesVille, Florida 32611-7200 Received December 6, 2005 A high-nuclearity mixed transition metal/actinide complex has been well-characterized transition metal/actinide complexes, among III - - prepared from the reaction of a Mn 4 complex with Th(NO3)4 in which are the dinuclear metal metal bonded M An orga- ) ) 6a MeCN/MeOH. The complex [Th6Mn10O22(OH)2(O2CPh)16(NO3)2- nometallic complexes (M Fe, Ru and An Th, U) and the family of linear trimetallic M IIUIV (M ) Co, Ni, Cu, (H2O)8] is the largest such complex to date and the first Th/Mn 2 6b species. It is rich in oxide groups, which stabilize all of the metals Zn) complexes containing a hexadentate Schiff base. in the high ThIV and MnIV oxidation levels. Magnetic characterization However, only one of these contains Mn, trinuclear [MnU O L (py) ](L- ) 1,7-diphenyl-1,3,5,7-heptanetetro- establishes that the complex has an S ) 3 ground-state spin value. 2 2 2 4 nato).7 Although Th is used in a wide array of products and processes, the cluster chemistry of Th is poorly developed compared to transition metals: Currently, there - 8a We have had a longstanding interest in the development are metal organic frameworks and organically templated 8b of manganese carboxylate cluster chemistry, mainly because Th complexes known, and the largest molecular Th 9 of its relevance to a variety of areas, including bioinorganic complex is Th6. -

Of the Periodic Table
of the Periodic Table teacher notes Give your students a visual introduction to the families of the periodic table! This product includes eight mini- posters, one for each of the element families on the main group of the periodic table: Alkali Metals, Alkaline Earth Metals, Boron/Aluminum Group (Icosagens), Carbon Group (Crystallogens), Nitrogen Group (Pnictogens), Oxygen Group (Chalcogens), Halogens, and Noble Gases. The mini-posters give overview information about the family as well as a visual of where on the periodic table the family is located and a diagram of an atom of that family highlighting the number of valence electrons. Also included is the student packet, which is broken into the eight families and asks for specific information that students will find on the mini-posters. The students are also directed to color each family with a specific color on the blank graphic organizer at the end of their packet and they go to the fantastic interactive table at www.periodictable.com to learn even more about the elements in each family. Furthermore, there is a section for students to conduct their own research on the element of hydrogen, which does not belong to a family. When I use this activity, I print two of each mini-poster in color (pages 8 through 15 of this file), laminate them, and lay them on a big table. I have students work in partners to read about each family, one at a time, and complete that section of the student packet (pages 16 through 21 of this file). When they finish, they bring the mini-poster back to the table for another group to use. -

Adverse Health Effects of Heavy Metals in Children
TRAINING FOR HEALTH CARE PROVIDERS [Date …Place …Event …Sponsor …Organizer] ADVERSE HEALTH EFFECTS OF HEAVY METALS IN CHILDREN Children's Health and the Environment WHO Training Package for the Health Sector World Health Organization www.who.int/ceh October 2011 1 <<NOTE TO USER: Please add details of the date, time, place and sponsorship of the meeting for which you are using this presentation in the space indicated.>> <<NOTE TO USER: This is a large set of slides from which the presenter should select the most relevant ones to use in a specific presentation. These slides cover many facets of the problem. Present only those slides that apply most directly to the local situation in the region. Please replace the examples, data, pictures and case studies with ones that are relevant to your situation.>> <<NOTE TO USER: This slide set discusses routes of exposure, adverse health effects and case studies from environmental exposure to heavy metals, other than lead and mercury, please go to the modules on lead and mercury for more information on those. Please refer to other modules (e.g. water, neurodevelopment, biomonitoring, environmental and developmental origins of disease) for complementary information>> Children and heavy metals LEARNING OBJECTIVES To define the spectrum of heavy metals (others than lead and mercury) with adverse effects on human health To describe the epidemiology of adverse effects of heavy metals (Arsenic, Cadmium, Copper and Thallium) in children To describe sources and routes of exposure of children to those heavy metals To understand the mechanism and illustrate the clinical effects of heavy metals’ toxicity To discuss the strategy of prevention of heavy metals’ adverse effects 2 The scope of this module is to provide an overview of the public health impact, adverse health effects, epidemiology, mechanism of action and prevention of heavy metals (other than lead and mercury) toxicity in children. -
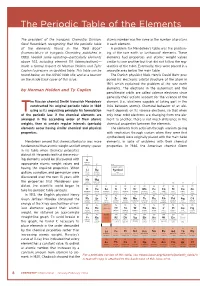
The Periodic Table of the Elements
The Periodic Table of the Elements The president of the Inorganic Chemistry Division, atomic number was the same as the number of protons Gerd Rosenblatt, recognizing that the periodic table in each element. of the elements found in the “Red Book” A problem for Mendeleev’s table was the position- (Nomenclature of Inorganic Chemistry, published in ing of the rare earth or lanthanoid* elements. These 1985) needed some updating—particularly elements elements had properties and atomic weight values above 103, including element 110 (darmstadtium)— similar to one another but that did not follow the reg- made a formal request to Norman Holden and Tyler ularities of the table. Eventually, they were placed in a Coplen to prepare an updated table. This table can be separate area below the main table. found below, on the IUPAC Web site, and as a tear-off The Danish physicist Niels Henrik David Bohr pro- on the inside back cover of this issue. posed his electronic orbital structure of the atom in 1921, which explained the problem of the rare earth by Norman Holden and Ty Coplen elements. The electrons in the outermost and the penultimate orbits are called valence electrons since generally their actions account for the valence of the he Russian chemist Dmitri Ivanovich Mendeleev element (i.e., electrons capable of taking part in the constructed his original periodic table in 1869 links between atoms). Chemical behavior of an ele- Tusing as its organizing principle his formulation ment depends on its valence electrons, so that when of the periodic law: if the chemical elements are only inner orbit electrons are changing from one ele- arranged in the ascending order of their atomic ment to another, there is not much difference in the weights, then at certain regular intervals (periods) chemical properties between the elements. -

Periodic Trends and the S-Block Elements”, Chapter 21 from the Book Principles of General Chemistry (Index.Html) (V
This is “Periodic Trends and the s-Block Elements”, chapter 21 from the book Principles of General Chemistry (index.html) (v. 1.0M). This book is licensed under a Creative Commons by-nc-sa 3.0 (http://creativecommons.org/licenses/by-nc-sa/ 3.0/) license. See the license for more details, but that basically means you can share this book as long as you credit the author (but see below), don't make money from it, and do make it available to everyone else under the same terms. This content was accessible as of December 29, 2012, and it was downloaded then by Andy Schmitz (http://lardbucket.org) in an effort to preserve the availability of this book. Normally, the author and publisher would be credited here. However, the publisher has asked for the customary Creative Commons attribution to the original publisher, authors, title, and book URI to be removed. Additionally, per the publisher's request, their name has been removed in some passages. More information is available on this project's attribution page (http://2012books.lardbucket.org/attribution.html?utm_source=header). For more information on the source of this book, or why it is available for free, please see the project's home page (http://2012books.lardbucket.org/). You can browse or download additional books there. i Chapter 21 Periodic Trends and the s-Block Elements In previous chapters, we used the principles of chemical bonding, thermodynamics, and kinetics to provide a conceptual framework for understanding the chemistry of the elements. Beginning in Chapter 21 "Periodic Trends and the ", we use the periodic table to guide our discussion of the properties and reactions of the elements and the synthesis and uses of some of their commercially important compounds. -

ELECTRONEGATIVITY D Qkq F=
ELECTRONEGATIVITY The electronegativity of an atom is the attracting power that the nucleus has for it’s own outer electrons and those of it’s neighbours i.e. how badly it wants electrons. An atom’s electronegativity is determined by Coulomb’s Law, which states, “ the size of the force is proportional to the size of the charges and inversely proportional to the square of the distance between them”. In symbols it is represented as: kq q F = 1 2 d 2 where: F = force (N) k = constant (dependent on the medium through which the force is acting) e.g. air q1 = charge on an electron (C) q2 = core charge (C) = no. of protons an outer electron sees = no. of protons – no. of inner shell electrons = main Group Number d = distance of electron from the nucleus (m) Examples of how to calculate the core charge: Sodium – Atomic number 11 Electron configuration 2.8.1 Core charge = 11 (no. of p+s) – 10 (no. of inner e-s) +1 (Group i) Chlorine – Atomic number 17 Electron configuration 2.8.7 Core charge = 17 (no. of p+s) – 10 (no. of inner e-s) +7 (Group vii) J:\Sciclunm\Resources\Year 11 Chemistry\Semester1\Notes\Atoms & The Periodic Table\Electronegativity.doc Neon holds onto its own electrons with a core charge of +8, but it can’t hold any more electrons in that shell. If it was to form bonds, the electron must go into the next shell where the core charge is zero. Group viii elements do not form any compounds under normal conditions and are therefore given no electronegativity values.