Predicting Protein Targets for Drug-Like Compounds Using Transcriptomics
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Snps) Distant from Xenobiotic Response Elements Can Modulate Aryl Hydrocarbon Receptor Function: SNP-Dependent CYP1A1 Induction S
Supplemental material to this article can be found at: http://dmd.aspetjournals.org/content/suppl/2018/07/06/dmd.118.082164.DC1 1521-009X/46/9/1372–1381$35.00 https://doi.org/10.1124/dmd.118.082164 DRUG METABOLISM AND DISPOSITION Drug Metab Dispos 46:1372–1381, September 2018 Copyright ª 2018 by The American Society for Pharmacology and Experimental Therapeutics Single Nucleotide Polymorphisms (SNPs) Distant from Xenobiotic Response Elements Can Modulate Aryl Hydrocarbon Receptor Function: SNP-Dependent CYP1A1 Induction s Duan Liu, Sisi Qin, Balmiki Ray,1 Krishna R. Kalari, Liewei Wang, and Richard M. Weinshilboum Division of Clinical Pharmacology, Department of Molecular Pharmacology and Experimental Therapeutics (D.L., S.Q., B.R., L.W., R.M.W.) and Division of Biomedical Statistics and Informatics, Department of Health Sciences Research (K.R.K.), Mayo Clinic, Rochester, Minnesota Received April 22, 2018; accepted June 28, 2018 ABSTRACT Downloaded from CYP1A1 expression can be upregulated by the ligand-activated aryl fashion. LCLs with the AA genotype displayed significantly higher hydrocarbon receptor (AHR). Based on prior observations with AHR-XRE binding and CYP1A1 mRNA expression after 3MC estrogen receptors and estrogen response elements, we tested treatment than did those with the GG genotype. Electrophoretic the hypothesis that single-nucleotide polymorphisms (SNPs) map- mobility shift assay (EMSA) showed that oligonucleotides with the ping hundreds of base pairs (bp) from xenobiotic response elements AA genotype displayed higher LCL nuclear extract binding after (XREs) might influence AHR binding and subsequent gene expres- 3MC treatment than did those with the GG genotype, and mass dmd.aspetjournals.org sion. -

Table 1. Swine Proteins Identified As Differentially Expressed at 24Dpi in OURT 88/3 Infected Animals
Table 1. swine proteins identified as differentially expressed at 24dpi in OURT 88/3 infected animals. Gene name Protein ID Protein Name -Log p-value control vs A_24DPI Difference control Vs A_24DPI F8 K7GL28 Coagulation factor VIII 2.123919902 5.42533493 PPBP F1RUL6 C-X-C motif chemokine 3.219079808 4.493174871 SDPR I3LDR9 Caveolae associated protein 2 2.191007299 4.085711161 IGHG L8B0X5 IgG heavy chain 2.084611488 -4.282530149 LOC100517145 F1S3H9 Complement C3 (LOC100517145) 3.885740476 -4.364484406 GOLM1 F1S4I1 Golgi membrane protein 1 1.746130664 -4.767168681 FCN2 I3L5W3 Ficolin-2 2.937884686 -6.029483795 Table 2. swine proteins identified as differentially expressed at 7dpi in Benin ΔMGF infected animals. Gene name Protein ID Protein Name -Log p-value control vs B_7DPI Difference control Vs B_7DPI A0A075B7I5 Ig-like domain-containing protein 1.765578164 -3.480728149 ATP5A1 F1RPS8_PIG ATP synthase subunit alpha 2.270386995 3.270935059 LOC100627396 F1RX35_PIG Fibrinogen C-terminal domain-containing protein 2.211242648 3.967363358 LOC100514666;LOC102158263 F1RX36_PIG Fibrinogen alpha chain 2.337934993 3.758180618 FGB F1RX37_PIG Fibrinogen beta chain 2.411948004 4.03753376 PSMA8 F1SBA5_PIG Proteasome subunit alpha type 1.473601007 -3.815182686 ACAN F1SKR0_PIG Aggrecan core protein 1.974489764 -3.726634026 TFG F1SL01_PIG PB1 domain-containing protein 1.809215274 -3.131304741 LOC100154408 F1SSL6_PIG Proteasome subunit alpha type 1.701949053 -3.944885254 PSMA4 F2Z528_PIG Proteasome subunit alpha type 2.045768185 -4.502977371 PSMA5 F2Z5K2_PIG -

Gene Set Size Count Z-Score P-Value Q-Value List of Genes
Supplementary Data 2-1 Results from competitive pathway enrichment analysis based on genome-wide summary-level data of hsGWAS. Gene Set Size Count z-score p-value q-value List of Genes KEGG_ALLOGRAFT_ 38 36 12.6476 0.00E+00 0.00E+00 HLA-DRB5(4.11028); IL2(3.62922); HLA-E(3.27708); HLA-G(3.2749); HLA- REJECTION DQA2(3.16347); HLA-DRA(3.14994); HLA-DOB(3.11438); HLA-DMB(3.09917); HLA-DMA(3.09615); TNF(3.09414); HLA-DPB1(3.03922); HLA-DPA1(3.0355); HLA-A(3.02312); HLA-C(2.98835); HLA-DQB1(2.97025); HLA-B(2.95626); HLA- DQA1(2.94591); HLA-F(2.83254); HLA-DRB1(2.76738); HLA-DOA(2.72508); IFNG(1.74523); PRF1(1.46483); FASLG(1.10083); FAS(0.923795); HLA- DRB3(0.760622); CD80(0.436654); IL4(0.324564); CD40(0.271072); HLA- DRB4(0.0708609); IL12A(0.0685942); IL5(0.0659333); CD86(0.049911); CD28(- 0.16103); IL10(-0.165903); IL12B(-0.245281); GZMB(-0.268581); KEGG_GRAFT_VERSUS_HOS 42 37 12.6129 0.00E+00 0.00E+00 HLA-DRB5(4.11028); IL2(3.62922); HLA-E(3.27708); HLA-G(3.2749); HLA- T_DISEASE DQA2(3.16347); HLA-DRA(3.14994); HLA-DOB(3.11438); HLA-DMB(3.09917); HLA-DMA(3.09615); TNF(3.09414); HLA-DPB1(3.03922); HLA-DPA1(3.0355); HLA-A(3.02312); HLA-C(2.98835); HLA-DQB1(2.97025); HLA-B(2.95626); HLA- DQA1(2.94591); HLA-F(2.83254); HLA-DRB1(2.76738); HLA-DOA(2.72508); IL6(1.94139); IFNG(1.74523); PRF1(1.46483); FASLG(1.10083); FAS(0.923795); HLA-DRB3(0.760622); CD80(0.436654); IL1A(0.402186); KLRD1(0.29064); KIR3DL1(0.157683); HLA-DRB4(0.0708609); CD86(0.049911); CD28(-0.16103); GZMB(-0.268581); IL1B(-0.388308); KLRC1(-0.466394); KIR3DL2(-0.786806); KEGG_TYPE_I_DIABETES_ME -

Polyubiquitin Gene Ubb Is Required for Upregulation of Piwi Protein Level During Mouse Testis Development
www.nature.com/cddiscovery ARTICLE OPEN Polyubiquitin gene Ubb is required for upregulation of Piwi protein level during mouse testis development 1,4 2,4 2 1 1 2 ✉ Bitnara Han , Byung-Kwon✉ Jung , So-Hyun Park , Kyu Jin Song , Muhammad Ayaz Anwar , Kwon-Yul Ryu and Kwang Pyo Kim 1,3 © The Author(s) 2021 Testis development, including early embryonic gonad formation and late postnatal spermatogenesis, is essential for the reproduction of higher metazoans to generate fertile gametes, called sperm. We have previously reported that the polyubiquitin gene Ubb is required for fertility in both male and female mice. In particular, the Ubb-null male mice showed an azoospermia phenotype due to arrest of spermatogenesis at the pachytene stage. Here, we analyzed the whole testis proteome at postnatal day 20 to define the molecular mediators of the male-infertility phenotype caused by Ubb knockout. From the identified proteome, 564 proteins were significantly and differentially expressed in Ubb-knockout testes and, among these, 36 downregulated proteins were involved at different stages of spermatogenesis. We also found that levels of piRNA metabolic process-related proteins, including Piwil2 and Tdrd1, were downregulated in Ubb-null testes through functional gene ontology analysis. Further, protein–protein interaction mapping revealed that 24 testis development-related proteins, including Hsp90aa1, Eef1a1, and Pabpc1, were directly influenced by the depletion of ubiquitin. In addition, the reduced mRNA levels of these proteins were observed in Ubb-knockout testes, which closely resembled the global downregulation of piRNA-metabolic gene expression at the transcriptional and post- transcriptional levels. Together with proteomic and transcriptional analyses, our data suggest that Ubb expression is essential for the maintenance of testicular RNA-binding regulators and piRNA-metabolic proteins to complete spermatogenesis in mice. -

A Genomic Analysis of Rat Proteases and Protease Inhibitors
A genomic analysis of rat proteases and protease inhibitors Xose S. Puente and Carlos López-Otín Departamento de Bioquímica y Biología Molecular, Facultad de Medicina, Instituto Universitario de Oncología, Universidad de Oviedo, 33006-Oviedo, Spain Send correspondence to: Carlos López-Otín Departamento de Bioquímica y Biología Molecular Facultad de Medicina, Universidad de Oviedo 33006 Oviedo-SPAIN Tel. 34-985-104201; Fax: 34-985-103564 E-mail: [email protected] Proteases perform fundamental roles in multiple biological processes and are associated with a growing number of pathological conditions that involve abnormal or deficient functions of these enzymes. The availability of the rat genome sequence has opened the possibility to perform a global analysis of the complete protease repertoire or degradome of this model organism. The rat degradome consists of at least 626 proteases and homologs, which are distributed into five catalytic classes: 24 aspartic, 160 cysteine, 192 metallo, 221 serine, and 29 threonine proteases. Overall, this distribution is similar to that of the mouse degradome, but significatively more complex than that corresponding to the human degradome composed of 561 proteases and homologs. This increased complexity of the rat protease complement mainly derives from the expansion of several gene families including placental cathepsins, testases, kallikreins and hematopoietic serine proteases, involved in reproductive or immunological functions. These protease families have also evolved differently in the rat and mouse genomes and may contribute to explain some functional differences between these two closely related species. Likewise, genomic analysis of rat protease inhibitors has shown some differences with the mouse protease inhibitor complement and the marked expansion of families of cysteine and serine protease inhibitors in rat and mouse with respect to human. -
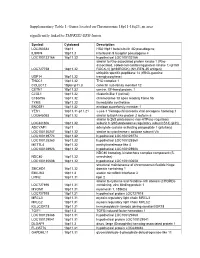
Supplementary Table 1: Genes Located on Chromosome 18P11-18Q23, an Area Significantly Linked to TMPRSS2-ERG Fusion
Supplementary Table 1: Genes located on Chromosome 18p11-18q23, an area significantly linked to TMPRSS2-ERG fusion Symbol Cytoband Description LOC260334 18p11 HSA18p11 beta-tubulin 4Q pseudogene IL9RP4 18p11.3 interleukin 9 receptor pseudogene 4 LOC100132166 18p11.32 hypothetical LOC100132166 similar to Rho-associated protein kinase 1 (Rho- associated, coiled-coil-containing protein kinase 1) (p160 LOC727758 18p11.32 ROCK-1) (p160ROCK) (NY-REN-35 antigen) ubiquitin specific peptidase 14 (tRNA-guanine USP14 18p11.32 transglycosylase) THOC1 18p11.32 THO complex 1 COLEC12 18pter-p11.3 collectin sub-family member 12 CETN1 18p11.32 centrin, EF-hand protein, 1 CLUL1 18p11.32 clusterin-like 1 (retinal) C18orf56 18p11.32 chromosome 18 open reading frame 56 TYMS 18p11.32 thymidylate synthetase ENOSF1 18p11.32 enolase superfamily member 1 YES1 18p11.31-p11.21 v-yes-1 Yamaguchi sarcoma viral oncogene homolog 1 LOC645053 18p11.32 similar to BolA-like protein 2 isoform a similar to 26S proteasome non-ATPase regulatory LOC441806 18p11.32 subunit 8 (26S proteasome regulatory subunit S14) (p31) ADCYAP1 18p11 adenylate cyclase activating polypeptide 1 (pituitary) LOC100130247 18p11.32 similar to cytochrome c oxidase subunit VIc LOC100129774 18p11.32 hypothetical LOC100129774 LOC100128360 18p11.32 hypothetical LOC100128360 METTL4 18p11.32 methyltransferase like 4 LOC100128926 18p11.32 hypothetical LOC100128926 NDC80 homolog, kinetochore complex component (S. NDC80 18p11.32 cerevisiae) LOC100130608 18p11.32 hypothetical LOC100130608 structural maintenance -

Isolation and Identification of Proteins from Swine Sperm Chromatin and Nuclear Matrix
DOI: 10.21451/1984-3143-AR816 Anim. Reprod., v.14, n.2, p.418-428, Apr./Jun. 2017 Isolation and identification of proteins from swine sperm chromatin and nuclear matrix Guilherme Arantes Mendonça1,3, Romualdo Morandi Filho2, Elisson Terêncio Souza2, Thais Schwarz Gaggini1, Marina Cruvinel Assunção Silva-Mendonça1, Robson Carlos Antunes1, Marcelo Emílio Beletti1,2 1Post-graduation Program in Veterinary Science, Federal University of Uberlandia, Uberlandia, MG, Brazil. 2Post-graduation Program in Cellular and Molecular Biology, Federal University of Uberlandia, Uberlandia, MG, Brazil. Abstract (Yamauchi et al., 2011). According to the same authors, these active sperm chromatin sites in protamine toroids The aim of this study was to perform a may contain important epigenetic information for the proteomic analysis to isolate and identify proteins from developing embryo. the swine sperm nuclear matrix to contribute to a The isolated use of genomic and transcriptomic database of swine sperm nuclear proteins. We used pre- information may be insufficient to fully understand a chilled diluted semen from seven boars (19 to 24 week- complex organism because proteomics and old) from the commercial line Landrace x Large White transcriptomics can be discordant and DNA-RNA x Pietran. The semen was processed to separate the relationships cannot be fully correlated. Thus, sperm heads and extract the chromatin and nuclear measurements of other metabolic levels should also be matrix for protein quantification and analysis by mass obtained, such as the study of proteins (Wright et al., spectrometry, by LTQ Orbitrap ELITE mass 2012). According to these same authors, large-scale spectrometer (Thermo-Finnigan) coupled to a nanoflow protein research in organisms (i.e., the proteome-protein chromatography system (LC-MS/MS). -

Integrated Network Analysis Identifying Potential Novel Drug Candidates
www.nature.com/scientificreports OPEN Integrated network analysis identifying potential novel drug candidates and targets for Parkinson’s disease Pusheng Quan1, Kai Wang2, Shi Yan1, Shirong Wen1, Chengqun Wei3, Xinyu Zhang1, Jingwei Cao1 & Lifen Yao1* This study aimed to identify potential novel drug candidates and targets for Parkinson’s disease. First, 970 genes that have been reported to be related to PD were collected from fve databases, and functional enrichment analysis of these genes was conducted to investigate their potential mechanisms. Then, we collected drugs and related targets from DrugBank, narrowed the list by proximity scores and Inverted Gene Set Enrichment analysis of drug targets, and identifed potential drug candidates for PD treatment. Finally, we compared the expression distribution of the candidate drug-target genes between the PD group and the control group in the public dataset with the largest sample size (GSE99039) in Gene Expression Omnibus. Ten drugs with an FDR < 0.1 and their corresponding targets were identifed. Some target genes of the ten drugs signifcantly overlapped with PD-related genes or already known therapeutic targets for PD. Nine diferentially expressed drug-target genes with p < 0.05 were screened. This work will facilitate further research into the possible efcacy of new drugs for PD and will provide valuable clues for drug design. Parkinson’s disease (PD) is a pervasive, progressive, disabling neurodegenerative disorder with motor and non- motor features1. PD places a signifcant burden on society and the afected individuals, and approximately 6.1 million people worldwide had been diagnosed with PD in 20162. Dopamine depletion leading to hyperactivity of the corticostriatal glutamatergic pathway is thought to be primarily responsible for parkinsonian symptoms such as resting tremors, rigidity, dyskinesia and postural instability 3,4. -

Proteomic Analyses of Human Sperm Cells: Understanding the Role of Proteins and Molecular Pathways Affecting Male Reproductive Health
International Journal of Molecular Sciences Review Proteomic Analyses of Human Sperm Cells: Understanding the Role of Proteins and Molecular Pathways Affecting Male Reproductive Health Ashok Agarwal * , Manesh Kumar Panner Selvam and Saradha Baskaran American Center for Reproductive Medicine, Cleveland Clinic, Cleveland, OH 44195, USA; [email protected] (M.K.P.S.); [email protected] (S.B.) * Correspondence: [email protected]; Tel.: +1-216-444-9485 Received: 19 January 2020; Accepted: 24 February 2020; Published: 27 February 2020 Abstract: Human sperm proteomics research has gained increasing attention lately, which provides complete information about the functional state of the spermatozoa. Changes in the sperm proteome are evident in several male infertility associated conditions. Global proteomic tools, such as liquid chromatography tandem mass spectrometry and matrix-assisted laser desorption/ionization time-of-flight, are used to profile the sperm proteins to identify the molecular pathways that are defective in infertile men. This review discusses the use of proteomic techniques to analyze the spermatozoa proteome. It also highlights the general steps involved in global proteomic approaches including bioinformatic analysis of the sperm proteomic data. Also, we have presented the findings of major proteomic studies and possible biomarkers in the diagnosis and therapeutics of male infertility. Extensive research on sperm proteome will help in understanding the role of fertility associated sperm proteins. Validation of the sperm proteins as biomarkers in different male infertility conditions may aid the physician in better clinical management. Keywords: sperm; proteomics; male infertility; bioinformatics; molecular pathways 1. Introduction Spermatozoa are matured motile cells that are products of spermatogenesis. A healthy man produces between 20 to 240 million sperms per day [1]. -

Sensitive and Frequent Identification of High Avidity Neo-Epitope Specific CD8+ T Cells in Immunotherapy-Naive Ovarian Cancer
CORE Metadata, citation and similar papers at core.ac.uk Provided by Serveur académique lausannois ARTICLE DOI: 10.1038/s41467-018-03301-0 OPEN Sensitive and frequent identification of high avidity neo-epitope specific CD8+ T cells in immunotherapy-naive ovarian cancer Sara Bobisse1, Raphael Genolet1, Annalisa Roberti2, Janos L. Tanyi2, Julien Racle 1,3, Brian J. Stevenson3, Christian Iseli3, Alexandra Michel1, Marie-Aude Le Bitoux1, Philippe Guillaume1, Julien Schmidt1, Valentina Bianchi1, Denarda Dangaj1, Craig Fenwick4, Laurent Derré 5, Ioannis Xenarios3, Olivier Michielin1,3, Pedro Romero1, Dimitri S. Monos6, Vincent Zoete1,3, David Gfeller1,3, Lana E. Kandalaft1,2, George Coukos1 & Alexandre Harari 1 1234567890():,; Immunotherapy directed against private tumor neo-antigens derived from non-synonymous somatic mutations is a promising strategy of personalized cancer immunotherapy. However, feasibility in low mutational load tumor types remains unknown. Comprehensive and deep analysis of circulating and tumor-infiltrating lymphocytes (TILs) for neo-epitope specific CD8+ T cells has allowed prompt identification of oligoclonal and polyfunctional such cells from most immunotherapy-naive patients with advanced epithelial ovarian cancer studied. Neo-epitope recognition is discordant between circulating T cells and TILs, and is more likely to be found among TILs, which display higher functional avidity and unique TCRs with higher predicted affinity than their blood counterparts. Our results imply that identification of neo- epitope specific CD8+ T cells is achievable even in tumors with relatively low number of somatic mutations, and neo-epitope validation in TILs extends opportunities for mutanome- based personalized immunotherapies to such tumors. 1 Department of Oncology, Lausanne University Hospital, Ludwig Institute for Cancer Research, University of Lausanne, Lausanne CH-1066, Switzerland. -

The Unique Functions of Tissue-Specific Proteasomes
Erschienen in: Trends in Biochemical Sciences ; 39 (2014), 1. - S. 17-24 https://dx.doi.org/10.1016/j.tibs.2013.10.004 The unique functions of tissue-specific proteasomes 1 1,2 Andrea Kniepert and Marcus Groettrup 1 Division of Immunology, Department of Biology, University of Konstanz, D-78457 Konstanz, Germany 2 Biotechnology Institute Thurgau at the University of Konstanz, CH-8280 Kreuzlingen, Switzerland The 26S proteasome is the main protease in eukaryotes. specialization of substrate specificity of these three b sub- Proteolysis occurs within the cylindrical 20S proteasome units, which is measured via the hydrolysis of small fluoro- that is constitutively expressed in most tissues. Howev- genic peptides with certain amino acids in the P1 position. er, three tissue-specific versions of the 20S proteasome These define the chymotrypsin-like activity (cleavage after have been discovered to date. The immunoproteasome hydrophobic residues), the trypsin-like activity (cleavage is optimized to process antigens and it directs the dif- after basic residues), and the caspase-like activity of the ferentiation of T helper (Th) cells. The thymoproteasome proteasome (cleavage after acidic residues). Although the is selectively expressed in cortical epithelial cells of the archaeal and bacterial proteasomes only show chymotryp- thymus where it plays an essential role in the positive sin-like activity, the eukaryotic proteasomes show all three selection of T lymphocytes. Finally, the spermatoprotea- activities that are associated with the b1 (caspase-like some is found in the testes where it is required during activity), b2 (trypsin-like activity), and b5 (chymotryp- spermatogenesis. Here, we outline how tissue-specific sin-like activity) subunits. -

Neonatal-Onset Autoinflammation and Immunodeficiency Caused by Heterozygous Missense Mutation of the Proteasome Subunit Β-Type 9
medRxiv preprint doi: https://doi.org/10.1101/2021.02.01.21250077; this version posted February 2, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. Neonatal-onset autoinflammation and immunodeficiency caused by heterozygous missense mutation of the proteasome subunit β-type 9 Nobuo Kanazawa1,2*, Hiroaki Hemmi3,4*, Noriko Kinjo5, Hidenori Ohnishi6, Jun Hamazaki7, Hiroyuki Mishima8, Akira Kinoshita8, Tsunehiro Mizushima9, Satoru Hamada5, Kazuya Hamada5, Norio Kawamoto6, Saori Kadowaki6, Yoshitaka Honda10, Kazushi Izawa10, Ryuta Nishikomori11, Miyuki Tsumura12, Yusuke Yamashita13, Shinobu Tamura13, Takashi Orimo3,14, Toshiya Ozasa3, Takashi Kato3, Izumi Sasaki3, Yuri Fukuda-Ohta3, Naoko Wakaki-Nishiyama3, Yutaka Inaba1, Kayo Kunimoto1, Satoshi Okada12, Takeshi Taketani15, Koichi Nakanishi5, Shigeo Murata7, Koh-ichiro Yoshiura8, Tsuneyasu Kaisho3 1 Department of Dermatology, Wakayama Medical University, Wakayama, Japan. 2Department of Dermatology, Hyogo College of Medicine, Hyogo, Japan. 3Department of Immunology, Institute of Advanced Medicine, Wakayama Medical University, Wakayama, Japan. 4Laboratory of Immunology, Faculty of Veterinary Medicine, Okayama University of Science, Ehime, Japan. 5Department of Child Health and Welfare (Pediatrics), Graduate School of Medicine, University of the Ryukyus, Okinawa, Japan 6Department of Pediatrics, Graduate School