Bull. Hist. Chem. 4
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

James Chadwick: Ahead of His Time
July 15, 2020 James Chadwick: ahead of his time Gerhard Ecker University of Vienna, Faculty of Physics Boltzmanngasse 5, A-1090 Wien, Austria Abstract James Chadwick is known for his discovery of the neutron. Many of his earlier findings and ideas in the context of weak and strong nuclear forces are much less known. This biographical sketch attempts to highlight the achievements of a scientist who paved the way for contemporary subatomic physics. arXiv:2007.06926v1 [physics.hist-ph] 14 Jul 2020 1 Early years James Chadwick was born on Oct. 20, 1891 in Bollington, Cheshire in the northwest of England, as the eldest son of John Joseph Chadwick and his wife Anne Mary. His father was a cotton spinner while his mother worked as a domestic servant. In 1895 the parents left Bollington to seek a better life in Manchester. James was left behind in the care of his grandparents, a parallel with his famous predecessor Isaac Newton who also grew up with his grandmother. It might be an interesting topic for sociologists of science to find out whether there is a correlation between children educated by their grandmothers and future scientific geniuses. James attended Bollington Cross School. He was very attached to his grandmother, much less to his parents. Nevertheless, he joined his parents in Manchester around 1902 but found it difficult to adjust to the new environment. The family felt they could not afford to send James to Manchester Grammar School although he had been offered a scholarship. Instead, he attended the less prestigious Central Grammar School where the teaching was actually very good, as Chadwick later emphasised. -

Marie Curie and Her Time
Marie Curie and Her Time by Hélène Langevin-Joliot to pass our lives near each other hypnotized by our dreams, your patriotic dream, our humanitarian dream, arie Curie (1867–1934) belongs to that exclu- and our scientific dream.” sive group of women whose worldwide rec- Frederick Soddy wrote about Marie that she was Mognition and fame have endured for a century “the most beautiful discovery of Pierre Curie.” Of or more. She was indeed one of the major agents of course, it might also be said that Pierre Curie was the scientific revolution which allowed experimen- “the most beautiful discovery of Marie Skłodowska.” tal investigation to extend beyond the macroscopic It is difficult to imagine more contrasting personali- world. Her work placed the first stone in the founda- ties than those of Pierre and of Marie. In spite of that, tion of a new discipline: radiochemistry. And Curie’s or because of that, they complemented each other achievements are even more remarkable since they astonishingly well. Pierre was as dreamy as Marie was occurred in the field of science, an intellectual activ- organized. At the same time, they shared similar ideas ity traditionally forbidden to women. However, these about family and society. accomplishments alone don’t seem to fully explain the near mythic status of Marie Curie today. One hundred years ago, she was often considered to be just an assistant to her husband. Perhaps the reason her name still resonates is because of the compelling story of her life and her intriguing personality. The Most Beautiful Discovery of Pierre Curie The story of the young Maria Skłodowska leaving In this iconic photograph of participants at the Fifth her native Poland to pursue upper-level studies in Solvay Conference in 1927, Marie Curie is third from Paris sounds like something out of a novel. -

Goverdhan Mehta Chemistry - a 21St Century Science for Global Sustainability: Is It Future Ready?
Goverdhan Mehta Chemistry - A 21st Century Science for Global Sustainability: Is it future ready? Goverdhan Mehta A ‘selfie’ with theSchool chemical of Chemistry world…… University of Hyderabad National Geophysical Research Institute, CSIR Foundation Day, Sept. 27, 2019 Introducing Chemistry through the Lens of Earth's Systems: What Role Can Systems Thinking Play in Developing Chemically and Environmentally Literate Citizens? J. Kornfeld, S. Stokoe. J. Chemical Education 2019, 96, 2910-2917 A bouquet of ‘matters’ that matter New symbols Passion Sustainability Legacies Ethics & values Responsible Connections Systems Directions Humility Ideas & icons Inspirations Un mélange de beaucoup de choses “Chemistry ought not to be for chemists alone” - Miguel de Unamuno ‘…Life, Universe and Everything’ Chemistry – a source of happiness…. Chem -Connectome ‘...I feel sorry for people who don’t know anything about chemistry. They are missing an important source of happiness....’ - Linus Pauling 1901-1994 S.A. Matlin, G. Mehta, H. Hopf. Chemistry Embraced by All. Science 2015, 347, 1179 Chemistry is in everything. and everything is in it, it is the basis of life, without it we wouldn't exist. Green tea has ~ 200 chemicals Coffee has ~ 1000 chemicals Wine has >1000 chemicals Light cigarette ~ 4000 chemicals Chemistry is ubiquitous/omnipresent Chemistry – Tracing the roots and to the present BCE Art & craft of mixing substances A giant knowledge leap Alchemy to modern science Evidence based science Discipline in a Table - systematization Mendeleev’s Periodic Law ‘Molecularization’ of chemical matter 20th Century A century of evolutionary march of chemistry 20h Century Value added products from almost anything “Utility science” and everything Molecular understanding of life processes and “Core Science” chemical matter Interdisciplinarity in forefront “Integrative Science” Resource stressed planet “Sustainability Science” 21st Century S. -

Physics Connections
Charles Tracy L P S / y a a w s n e v a R n a v v e l t e D Physics connections Last year we celebrated the International Year of Physics — time and light. In particular, it described how space also known as Einstein Year. In the century since Einstein’s becomes distorted near a massive object such as a star, so that light follows a curved path rather than annus mirabilis (see CATALYST Vol. 16, No. 1) there has travelling in a straight line (Figure 1). been a revolution in the study of physics. This article explores Its publication in wartime and the fact that it was the links between some of the architects of this revolution. written in German meant that few people read it. However, one person who did read it was the British astronomer Arthur Eddington. The story goes that it was Eddington who brought Einstein to the world’s attention — though Einstein might have disagreed. instein did not become instantly famous in Eddington was a conscientious objector. As such, GCSE key words 1905; it took several years before he became a he should have spent the war in jail. Instead, he got Gravity Eglobal icon. In 1915, while Europe was caught official permission to prepare for a trip to observe a Alpha particle scattering up in the First World War, Einstein broadened his total eclipse of the Sun — a rare event. His intention Nuclear fission special theory of relativity. His general theory of was to verify Einstein’s general theory of relativity. -

Pauling-Linus.Pdf
NATIONAL ACADEMY OF SCIENCES L I N U S C A R L P A U L I N G 1901—1994 A Biographical Memoir by J A C K D. D UNITZ Any opinions expressed in this memoir are those of the author(s) and do not necessarily reflect the views of the National Academy of Sciences. Biographical Memoir COPYRIGHT 1997 NATIONAL ACADEMIES PRESS WASHINGTON D.C. LINUS CARL PAULING February 28, 1901–August 19, 1994 BY JACK D. DUNITZ INUS CARL PAULING was born in Portland, Oregon, on LFebruary 28, 1901, and died at his ranch at Big Sur, California, on August 19, 1994. In 1922 he married Ava Helen Miller (died 1981), who bore him four children: Linus Carl, Peter Jeffress, Linda Helen (Kamb), and Edward Crellin. Pauling is widely considered the greatest chemist of this century. Most scientists create a niche for themselves, an area where they feel secure, but Pauling had an enormously wide range of scientific interests: quantum mechanics, crys- tallography, mineralogy, structural chemistry, anesthesia, immunology, medicine, evolution. In all these fields and especially in the border regions between them, he saw where the problems lay, and, backed by his speedy assimilation of the essential facts and by his prodigious memory, he made distinctive and decisive contributions. He is best known, perhaps, for his insights into chemical bonding, for the discovery of the principal elements of protein secondary structure, the alpha-helix and the beta-sheet, and for the first identification of a molecular disease (sickle-cell ane- mia), but there are a multitude of other important contri- This biographical memoir was prepared for publication by both The Royal Society of London and the National Academy of Sciences of the United States of America. -

John D. Roberts
John D. (Jack) Roberts 1918 – 2016 John D. Roberts, the Institute Professor of Chemistry, Emeritus, and one of the most influential chemists of the 20th century, passed away on October 29, 2016 at the age of 98 following a stroke. John Dombrowski “Jack” Roberts was born on June 8, 1918 in Los Angeles, California. He spent most of his 98 years in Southern California and witnessed first hand its transformation from a reasonably under- populated region into one of the world’s busiest metropolitan areas. In fact, Jack (or “JDR” as he was oft referred in the labs at Caltech) was born essentially right underneath what is now the famous four level interchange connecting the 101 and 110 freeways in modern day downtown LA. JDR also witnessed the growth and explosion of science and in particular chemistry over that century span. As summarized in his J. Org. Chem. 2009, 74, 4897-4917 article and numerous talks over the later part of his life, the explosion of instrumentation capabilities available to the organic chemist progressed in the course of his scientific career from no less than the melting point apparatus to some of the most advanced instruments on the planet. Without doubt, the advances most influential to JDR’s monumental career in chemistry were the advent of nuclear magnetic resonance (NMR) spectroscopy and the accompanying explosion in computing. Combined, these tools greatly facilitated the insightfully designed experimentation and careful analyses that became the hallmark of JDR’s career. It is clear that Jack’s thoroughgoing nature combined with his deep understanding of instrumentation and fundamental chemistry served as an inspiration to nearly four generations of scientists. -

Walter Loveland Oral History Interview, “Of Glenn Seaborg and Super Heavy Elements: a Nuclear Chemist Looks Back”, July Page 3 of 24 22, 2015
Walter Loveland Oral History Interview, July 22, 2015 Title “Of Glenn Seaborg and Super Heavy Elements: A Nuclear Chemist Looks Back” Date July 22, 2015 Location Valley Library, Oregon State University. Summary In the interview, Loveland discusses his colorful family background and upbringing in blue-collar suburban Chicago. He also describes his earliest interests in science, his path through undergraduate and graduate studies, and those who influenced him as he made his way through his higher education, including his contacts with luminaries like Charles Coryell and John Huizenga. From there, Loveland begins to reflect on his long association with both Oregon State University and the University of California, Berkeley. In so doing, he shares his memories of his initial impressions of OSU and Corvallis, his first contacts with Glenn Seaborg, a few initial research experiences in research, and his impressions of Seaborg as a personality. He likewise recounts his interactions with Linus Pauling as well as major figures in nuclear science at OSU, Chih Wang, John Ringle and Dale Trout among them. Loveland next recounts his memories of the Radiation Biology program at OSU; discusses the life and career of a former student, Sister Mary Joseph Bouchard; and comments on the climate for women and people of color in the sciences at OSU and in the community at large. Loveland's research is the next focus of the interview. In this he provides an overview of his work with super-heavy ions while also describing his research collaborations and the frequent trips to Berkeley that these collaborations demanded. He also recounts his interactions with OSU's Campus Radiation Safety Committee, his disinterest in working at the Hanford Nuclear Site, his experience of co-authoring two books with Glenn Seaborg, and hindrances to scientific advancement that he has noted as a result of denials of security clearance. -

Named Organic Reactions (U – Z)
Dr. John Andraos, http://www.careerchem.com/NAMED/Named-Rxns(U-Z).pdf 1 NAMED ORGANIC REACTIONS (U – Z) © Dr. John Andraos, 2000 - 2014 Department of Chemistry, York University 4700 Keele Street, Toronto, ONTARIO M3J 1P3, CANADA For suggestions, corrections, additional information, and comments please send e-mails to [email protected] http://www.chem.yorku.ca/NAMED/ Sakae Uemura Japanese, b. Japan Uemura oxidation Nishimura, T.; Onoue, T.; Ohe, K.; Uemura S. Tetrahedron Lett. 1998 , 39 , 6011 Nishimura, T.; Onoue, T.; Ohe, K.; Uemura S. J. Org. Chem. 1999 , 64 , 6750 Biographical references: Ivar Karl Ugi 5 September 1930 – 29 September 2005 German, b. Kuressaare, Estland, Germany Ugi condensation Ugi, I.; Meyr, R.; Fetzer, U.; Steinbrueckner, C. Angew. Chem. 1959 , 71 , 386 Urban, R.; Ugi, I., Angew. Chem. Int. Engl. Ed . 1975 , 14 , 61 Biographical references: Chemical Research Faculties: An International Directory 1996 , American Chemical Society: Washington, D.C., 1996 Wer ist Wer? Das Deutsche Who’s Who 1999/2000 , Schmidt-Römhild: Lübeck, 1999 Lemmen, P.; Fontain, E.; Bauer, J. Angew. Chem. Int. Ed. 2006 , 45 , 193 Fritz Ullmann 2 July 1875 - 17 March 1939 German, b. Fürth, Germany Ullmann reaction Ullmann, F. Ann. Chem . 1904 , 332 , 38 Dr. John Andraos, http://www.careerchem.com/NAMED/Named-Rxns(U-Z).pdf 2 Biographical references: Meyer, K.H., Helv. Chim. Acta 1940 , 23 , 93 Upjohn reaction Van Rheenen, V.; Kelly, R.C.; Cha, D.Y. Tetrahedron Lett. 1976 , 1973 A. Verley French, b. ? Meerwein-Pondorff-Verley reduction see Hans Leberecht Meerwein Biographical references: Victor Villiger 1 September 1868 - 1934 Swiss, b. -

Otto Hahn Otto Hahn
R.N. 70269/98 Postal Registration No.: DL-SW-1/4082/15-17 ISSN : 0972-169X Date of posting: 26-27 of advance month Date of publication: 24 of advance month May 2017 Vol. 19 No. 8 Rs. 5.00 Otto Hahn Discoverer of Nuclear Fission Editorial: Consolidating 35 science communication activities in our country Otto Hahn: Discoverer of 34 Nuclear Fission Keep Your Eyes Healthy 31 Phenol: A Serious 30 Environmental Threat Accidental Discoveries in 28 Medical Science Cures for haemorrhoids— 24 Simple treatments and Surgeries Recent developments 21 in science and technology 36 Editorial Consolidating science communication activities in our country Dr. R. Gopichandran It is well known that the National Council of Science Museums of India’s leadership in science technology and innovation (STI) across the Ministry of Culture, Government of India, the National Institute the bilateral and multilateral framework also. The news feature service of Science Communication and Information Resources (NISCAIR) and the portal activity have well defined action plans to reach out to of CSIR, the National Council for Science and Technology fellow institutions and citizens with suitably embellished platform Communication (NCSTC) of the Department of Science and and opportunities for all to deliver together. Technology (DST), Government of India and Vigyan Prasar, also While these are interesting and extremely important, especially of DST, have been carrying out excellent science communication because they respond to the call to upscale and value add science activities over the years. It cannot be denied that the reach has been and technology communication, it is equally important to document quite significant collectively. -
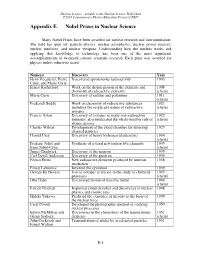
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

ARIE SKLODOWSKA CURIE Opened up the Science of Radioactivity
ARIE SKLODOWSKA CURIE opened up the science of radioactivity. She is best known as the discoverer of the radioactive elements polonium and radium and as the first person to win two Nobel prizes. For scientists and the public, her radium was a key to a basic change in our understanding of matter and energy. Her work not only influenced the development of fundamental science but also ushered in a new era in medical research and treatment. This file contains most of the text of the Web exhibit “Marie Curie and the Science of Radioactivity” at http://www.aip.org/history/curie/contents.htm. You must visit the Web exhibit to explore hyperlinks within the exhibit and to other exhibits. Material in this document is copyright © American Institute of Physics and Naomi Pasachoff and is based on the book Marie Curie and the Science of Radioactivity by Naomi Pasachoff, Oxford University Press, copyright © 1996 by Naomi Pasachoff. Site created 2000, revised May 2005 http://www.aip.org/history/curie/contents.htm Page 1 of 79 Table of Contents Polish Girlhood (1867-1891) 3 Nation and Family 3 The Floating University 6 The Governess 6 The Periodic Table of Elements 10 Dmitri Ivanovich Mendeleev (1834-1907) 10 Elements and Their Properties 10 Classifying the Elements 12 A Student in Paris (1891-1897) 13 Years of Study 13 Love and Marriage 15 Working Wife and Mother 18 Work and Family 20 Pierre Curie (1859-1906) 21 Radioactivity: The Unstable Nucleus and its Uses 23 Uses of Radioactivity 25 Radium and Radioactivity 26 On a New, Strongly Radio-active Substance -

Wilhelm Ostwald – the Scientist
ARTICLE-IN-A-BOX Wilhelm Ostwald – The Scientist Friedrich Wilhelm Ostwald was born on September 2, 1853 at Riga, Latvia, Russia to Gottfried Ostwald, a master cooper and Elisabeth Leuckel. He was the second son to his parents who both were descended from German immigrants. He had his early education at Riga. His subsequent education was at the University of Dorpat (now Tartu, Estonia) where he enrolled in 1872. At the university he studied chemistry under the tutelage of Carl ErnstHeinrich Schmidt (1822– 1894) who again was a pupil of Justus von Liebig. Besides Schmidt, Johannes Lemberg (1842– 1902) and Arthur von Oettingen (1836–1920) who were his teachers in physical chemistry were also principal influences. It was in 1877 that he defended his thesis ‘Volumchemische Studien über Affinität’. Subsequently he taught as Privatdozent for a couple of years. During this period, his personal life saw some changes as well. He wedded Helene von Reyher (1854–1946) in 1880. With Helene, he had three sons, and two daughters. Amongst his children, Wolfgang went on to become a famous colloid chemist. On the strength of recommendation from Dorpat, Ostwald was appointed as a Professor at Riga Polytechnicum in 1882. He worked on multiple applications of the law of mass action. He also conducted measurements in chemical reaction kinetics as well as conductivity of solutions. To this end, the pyknometer was developed which was used to determine the density of liquids. He also had a thermostat built and both of these were named after him. He was prolific in his teaching and research which helped establish a school of science at the university.