Common and Disorders-Specific Cortical Thickness Alterations in Internalizing, Externalizing and Thought Disorders in the Preado
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genetic Variations Influence Brain Changes in Patients with Attention
Yadav et al. Translational Psychiatry (2021) 11:349 https://doi.org/10.1038/s41398-021-01473-w Translational Psychiatry REVIEW ARTICLE Open Access Genetic variations influence brain changes in patients with attention-deficit hyperactivity disorder Santosh K. Yadav1,AjazA.Bhat 1, Sheema Hashem1,SabahNisar1, Madeeha Kamal2,NajeebSyed3, Mohamed-Ramzi Temanni3, Rakesh K. Gupta4, Saddat Kamran 5, Muhammad Waqar Azeem6, Amit K. Srivastava7, Puneet Bagga8, Sanjeev Chawla9, Ravinder Reddy10, Michael P. Frenneaux11, Khalid Fakhro 12,13 and Mohammad Haris 1,14 Abstract Attention-deficit hyperactivity disorder (ADHD) is a neurological and neurodevelopmental childhood-onset disorder characterized by a persistent pattern of inattentiveness, impulsiveness, restlessness, and hyperactivity. These symptoms may continue in 55–66% of cases from childhood into adulthood. Even though the precise etiology of ADHD is not fully understood, it is considered as a multifactorial and heterogeneous disorder with several contributing factors such as heritability, auxiliary to neurodevelopmental issues, severe brain injuries, neuroinflammation, consanguineous marriages, premature birth, and exposure to environmental toxins. Neuroimaging and neurodevelopmental assessments may help to explore the possible role of genetic variations on ADHD neuropsychobiology. Multiple genetic studies have observed a strong genetic association with various aspects of neuropsychobiological functions, including neural abnormalities and delayed neurodevelopment in ADHD. The advancement in neuroimaging and 1234567890():,; 1234567890():,; 1234567890():,; 1234567890():,; molecular genomics offers the opportunity to analyze the impact of genetic variations alongside its dysregulated pathways on structural and functional derived brain imaging phenotypes in various neurological and psychiatric disorders, including ADHD. Recently, neuroimaging genomic studies observed a significant association of brain imaging phenotypes with genetic susceptibility in ADHD. -

Supplementary Table 4
Li et al. mir-30d in human cancer Table S4. The probe list down-regulated in MDA-MB-231 cells by mir-30d mimic transfection Gene Probe Gene symbol Description Row set 27758 8119801 ABCC10 ATP-binding cassette, sub-family C (CFTR/MRP), member 10 15497 8101675 ABCG2 ATP-binding cassette, sub-family G (WHITE), member 2 18536 8158725 ABL1 c-abl oncogene 1, receptor tyrosine kinase 21232 8058591 ACADL acyl-Coenzyme A dehydrogenase, long chain 12466 7936028 ACTR1A ARP1 actin-related protein 1 homolog A, centractin alpha (yeast) 18102 8056005 ACVR1 activin A receptor, type I 20790 8115490 ADAM19 ADAM metallopeptidase domain 19 (meltrin beta) 15688 7979904 ADAM21 ADAM metallopeptidase domain 21 14937 8054254 AFF3 AF4/FMR2 family, member 3 23560 8121277 AIM1 absent in melanoma 1 20209 7921434 AIM2 absent in melanoma 2 19272 8136336 AKR1B10 aldo-keto reductase family 1, member B10 (aldose reductase) 18013 7954777 ALG10 asparagine-linked glycosylation 10, alpha-1,2-glucosyltransferase homolog (S. pombe) 30049 7954789 ALG10B asparagine-linked glycosylation 10, alpha-1,2-glucosyltransferase homolog B (yeast) 28807 7962579 AMIGO2 adhesion molecule with Ig-like domain 2 5576 8112596 ANKRA2 ankyrin repeat, family A (RFXANK-like), 2 23414 7922121 ANKRD36BL1 ankyrin repeat domain 36B-like 1 (pseudogene) 29782 8098246 ANXA10 annexin A10 22609 8030470 AP2A1 adaptor-related protein complex 2, alpha 1 subunit 14426 8107421 AP3S1 adaptor-related protein complex 3, sigma 1 subunit 12042 8099760 ARAP2 ArfGAP with RhoGAP domain, ankyrin repeat and PH domain 2 30227 8059854 ARL4C ADP-ribosylation factor-like 4C 32785 8143766 ARP11 actin-related Arp11 6497 8052125 ASB3 ankyrin repeat and SOCS box-containing 3 24269 8128592 ATG5 ATG5 autophagy related 5 homolog (S. -

Novel Approach to Evaluate Central Autonomic Regulation in Attention Deficit/Hyperactivity Disorder (ADHD)
Physiol. Res. 68: 531-545, 2019 https://doi.org/10.33549/physiolres.934160 REVIEW Novel Approach to Evaluate Central Autonomic Regulation in Attention Deficit/Hyperactivity Disorder (ADHD) N. SEKANINOVA1,2, M. MESTANIK2,1, A. MESTANIKOVA1,2, A. HAMRAKOVA1,3, I. TONHAJZEROVA1,2 1Department of Physiology, Jessenius Faculty of Medicine in Martin, Comenius University in Bratislava, Martin, Slovak Republic, 2Biomedical Center Martin, Jessenius Faculty of Medicine in Martin, Comenius University in Bratislava, Martin, Slovak Republic, 3Psychiatric Clinic, Jessenius Faculty of Medicine in Martin, Comenius University in Bratislava, University Hospital Martin, Martin, Slovak Republic Received March 7, 2019 Accepted April 15, 2019 Epub Ahead of Print June 6, 2019 Summary Key words Attention deficit/hyperactivity disorder (ADHD) is one of the most ADHD • Neurobiology • Autonomic regulation • Pupillometry • commonly diagnosed developmental disorders in childhood Eye-tracking characterized by hyperactivity, impulsivity and inattention. ADHD manifests in the child’s development by deficits in cognitive, Corresponding author executive and perceptor-motor functions, emotional regulation I. Tonhajzerova, Department of Physiology and Biomedical and social adaptation. Although the exact cause has not yet been Center Martin, Jessenius Faculty of Medicine in Martin, Comenius known, the crucial role in the development of this disease plays University in Bratislava, Mala Hora 4C, 036 01 Martin, Slovak the interaction of genetic, neurobiological and epigenetic factors. Republic. E-mail: [email protected] According to current knowledge, ADHD is defined as a biological dysfunction of central nervous system with genetically or Introduction organically defined deficits in noradrenergic and dopaminergic neurotransmission associated with structural abnormalities, ADHD (Attention Deficit Hyperactivity especially in prefronto-striatal regions. -

Knockout of Latrophilin-3 in Sprague-Dawley Rats Causes Hyperactivity, Hyper-Reactivity, Under-Response to Amphetamine, and Disrupted Dopamine Markers
ACCEPTED MANUSCRIPT Knockout of latrophilin-3 in Sprague-Dawley rats causes hyperactivity, hyper- reactivity, under-response to amphetamine, and disrupted dopamine markers Samantha L. Regana, Jillian R. Hufgardb, Emily M. Pitzera, Chiho Sugimotoc, Yueh- Chiang Hud, Michael T. Williamsc, and Charles V. Vorheesc* aNeuroscience Graduate Program, University of Cincinnati, bDepartment of Obstetrics and Gynecology, Indiana University School of Medicine, cDepartment of Pediatrics, University of Cincinnati College of Medicine and Division of Neurology, Cincinnati Children’s Hospital Medical Center, dDepartment of Pediatrics, University of Cincinnati College of Medicine and Division of Developmental Biology, Cincinnati Children’s Hospital Medical Center, Cincinnati, OH 45229, USA ORCID: S.L.R. 0000-0001-9198-1411, J.R.H. 0000-0002-6248-1087, M.T.W. 0000- 0001-9841-9683, C.V.V. 0000-0003-3558-8812. *Corresponding author: Charles V. Vorhees, Ph.D., Div. of Neurology, Cincinnati Children’s Research Foundation, 3333 Burnet Ave., Cincinnati, OH 45229, USA: email: [email protected] Abbreviated title: Lphn3 KO rat, hyperactivity and dopaminergic effects ACCEPTED MANUSCRIPT ___________________________________________________________________ This is the author's manuscript of the article published in final edited form as: Regan, S. L., Hufgard, J. R., Pitzer, E. M., Sugimoto, C., Hu, Y.-C., Williams, M. T., & Vorhees, C. V. (2019). Knockout of latrophilin-3 in Sprague-Dawley rats causes hyperactivity, hyper-reactivity, under-response to amphetamine, and disrupted dopamine markers. Neurobiology of Disease, 130, 104494. https://doi.org/10.1016/j.nbd.2019.104494 ACCEPTED MANUSCRIPT Abstract Attention deficit hyperactivity disorder is a pervasive developmental disorder characterized by inattention, impulsivity, and hyperactivity and is 75-90% heritable. -

Zebrafish Models for Attention Deficit Hyperactivity Disorder (ADHD)
Accepted Manuscript Title: Zebrafish models for attention deficit hyperactivity disorder (ADHD) Authors: Barbara D. Fontana, Francini Franscescon, Denis B. Rosemberg, William H.J. Norton, Allan V. Kalueff, Matthew O. Parker PII: S0149-7634(18)30880-7 DOI: https://doi.org/10.1016/j.neubiorev.2019.02.009 Reference: NBR 3349 To appear in: Received date: 15 November 2018 Revised date: 15 February 2019 Accepted date: 15 February 2019 Please cite this article as: Fontana BD, Franscescon F, Rosemberg DB, Norton WHJ, Kalueff AV, Parker MO, Zebrafish models for attention deficit hyperactivity disorder (ADHD), Neuroscience and Biobehavioral Reviews (2019), https://doi.org/10.1016/j.neubiorev.2019.02.009 This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain. 1 Zebrafish models for attention deficit hyperactivity disorder (ADHD) Barbara D. Fontanaa*, Francini Franscesconb, Denis B. Rosembergb,k, William H.J. Nortonc,k, Allan V. Kalueffd,e,f,g,h,i,j,k and Matthew O. Parkera,k* aBrain and Behaviour Laboratory, School of Pharmacy and Biomedical Sciences, University of Portsmouth, UK. bGraduate Program in Biological Sciences: Toxicological Biochemistry, Federal University of Santa Maria, 1000 Roraima Avenue, Santa Maria, RS 97105-900, Brazil cDepartment of Neuroscience, Psychology and Behaviour, College of Medicine, Biological Sciences and Psychology, University of Leicester, Leicester, LE1 7RH, UK. -

Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells Amy Sue Bogard University of Tennessee Health Science Center
University of Tennessee Health Science Center UTHSC Digital Commons Theses and Dissertations (ETD) College of Graduate Health Sciences 12-2013 Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells Amy Sue Bogard University of Tennessee Health Science Center Follow this and additional works at: https://dc.uthsc.edu/dissertations Part of the Medical Cell Biology Commons, and the Medical Molecular Biology Commons Recommended Citation Bogard, Amy Sue , "Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells" (2013). Theses and Dissertations (ETD). Paper 330. http://dx.doi.org/10.21007/etd.cghs.2013.0029. This Dissertation is brought to you for free and open access by the College of Graduate Health Sciences at UTHSC Digital Commons. It has been accepted for inclusion in Theses and Dissertations (ETD) by an authorized administrator of UTHSC Digital Commons. For more information, please contact [email protected]. Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells Document Type Dissertation Degree Name Doctor of Philosophy (PhD) Program Biomedical Sciences Track Molecular Therapeutics and Cell Signaling Research Advisor Rennolds Ostrom, Ph.D. Committee Elizabeth Fitzpatrick, Ph.D. Edwards Park, Ph.D. Steven Tavalin, Ph.D. Christopher Waters, Ph.D. DOI 10.21007/etd.cghs.2013.0029 Comments Six month embargo expired June 2014 This dissertation is available at UTHSC Digital Commons: https://dc.uthsc.edu/dissertations/330 Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells A Dissertation Presented for The Graduate Studies Council The University of Tennessee Health Science Center In Partial Fulfillment Of the Requirements for the Degree Doctor of Philosophy From The University of Tennessee By Amy Sue Bogard December 2013 Copyright © 2013 by Amy Sue Bogard. -

Mutations in Sphingolipid Metabolism Genes Are Associated with ADHD Marcela Henriquez-Henriquez1,2,Mariat.Acosta3, Ariel F
Henriquez-Henriquez et al. Translational Psychiatry (2020) 10:231 https://doi.org/10.1038/s41398-020-00881-8 Translational Psychiatry ARTICLE Open Access Mutations in sphingolipid metabolism genes are associated with ADHD Marcela Henriquez-Henriquez1,2,MariaT.Acosta3, Ariel F. Martinez 3, Jorge I. Vélez 4, Francisco Lopera5, David Pineda 5, Juan D. Palacio 5, Teresa Quiroga1, Tilla S. Worgall6, Richard J. Deckelbaum7, Claudio Mastronardi8, Brooke S. G. Molina 9, the MTA Cooperative Group, Mauricio Arcos-Burgos 10 and Maximilian Muenke 3 Abstract Attention deficit hyperactivity disorder (ADHD) is the most prevalent neurodevelopmental disorder in children, with genetic factors accounting for 75–80% of the phenotypic variance. Recent studies have suggested that ADHD patients might present with atypical central myelination that can persist into adulthood. Given the essential role of sphingolipids in myelin formation and maintenance, we explored genetic variation in sphingolipid metabolism genes for association with ADHD risk. Whole-exome genotyping was performed in three independent cohorts from disparate regions of the world, for a total of 1520 genotyped subjects. Cohort 1 (MTA (Multimodal Treatment study of children with ADHD) sample, 371 subjects) was analyzed as the discovery cohort, while cohorts 2 (Paisa sample, 298 subjects) and 3 (US sample, 851 subjects) were used for replication. A set of 58 genes was manually curated based on their roles in sphingolipid metabolism. A targeted exploration for association between ADHD and 137 markers encoding for common and rare potentially functional allelic variants in this set of genes was performed in the screening cohort. Single- and multi-locus additive, dominant and recessive linear mixed-effect models were used. -
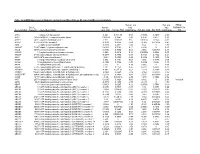
Mrna Expression in Human Leiomyoma and Eker Rats As Measured by Microarray Analysis
Table 3S: mRNA Expression in Human Leiomyoma and Eker Rats as Measured by Microarray Analysis Human_avg Rat_avg_ PENG_ Entrez. Human_ log2_ log2_ RAPAMYCIN Gene.Symbol Gene.ID Gene Description avg_tstat Human_FDR foldChange Rat_avg_tstat Rat_FDR foldChange _DN A1BG 1 alpha-1-B glycoprotein 4.982 9.52E-05 0.68 -0.8346 0.4639 -0.38 A1CF 29974 APOBEC1 complementation factor -0.08024 0.9541 -0.02 0.9141 0.421 0.10 A2BP1 54715 ataxin 2-binding protein 1 2.811 0.01093 0.65 0.07114 0.954 -0.01 A2LD1 87769 AIG2-like domain 1 -0.3033 0.8056 -0.09 -3.365 0.005704 -0.42 A2M 2 alpha-2-macroglobulin -0.8113 0.4691 -0.03 6.02 0 1.75 A4GALT 53947 alpha 1,4-galactosyltransferase 0.4383 0.7128 0.11 6.304 0 2.30 AACS 65985 acetoacetyl-CoA synthetase 0.3595 0.7664 0.03 3.534 0.00388 0.38 AADAC 13 arylacetamide deacetylase (esterase) 0.569 0.6216 0.16 0.005588 0.9968 0.00 AADAT 51166 aminoadipate aminotransferase -0.9577 0.3876 -0.11 0.8123 0.4752 0.24 AAK1 22848 AP2 associated kinase 1 -1.261 0.2505 -0.25 0.8232 0.4689 0.12 AAMP 14 angio-associated, migratory cell protein 0.873 0.4351 0.07 1.656 0.1476 0.06 AANAT 15 arylalkylamine N-acetyltransferase -0.3998 0.7394 -0.08 0.8486 0.456 0.18 AARS 16 alanyl-tRNA synthetase 5.517 0 0.34 8.616 0 0.69 AARS2 57505 alanyl-tRNA synthetase 2, mitochondrial (putative) 1.701 0.1158 0.35 0.5011 0.6622 0.07 AARSD1 80755 alanyl-tRNA synthetase domain containing 1 4.403 9.52E-05 0.52 1.279 0.2609 0.13 AASDH 132949 aminoadipate-semialdehyde dehydrogenase -0.8921 0.4247 -0.12 -2.564 0.02993 -0.32 AASDHPPT 60496 aminoadipate-semialdehyde -

ADHD Stimulant Medication and the Risk of Sudden Cardiac Death
ADHD Stimulant Medication and the Risk of Sudden Cardiac Death Marc Lerner, M.D. CHOC Childrens Hospital University of California, Irvine Attention Deficit Hyperactivity Disorder Neurobehavioral disorder marked by one or more of the following: Inattention (poor focus / distractibility) Hyperactivity (excessive motor activity) Impulsivity (no “brakes”) Prevalence rates 33--8%8% of the school-school-ageage population Clinically presents more often in boys than in girls (3:1) Three quarters of children retain ADHD symptoms in adolescence, and up to one half as adults http://www.cdc.gov/ncbddd/adhd/ Froehlich TE, Lanphear BP, et al. Arch Pediatr Adolesc Med. 2007 Sep;161(9):857-64. January 14-15, 2011 SCA Conference Molecular Genetics of ADHD Specific genes associated with ADHD Dopamine receptor D4 gene (DRD4) on chromosome 11 Dopamine transporter gene (DAT1) on chromosome 5 D2 dopamine receptor gene DopamineDopamine--betabeta--hydroxylasehydroxylase gene Possible association of noradrenergic genes Most recently identidentified:ified: Latrophilin 3 gene (LPHN3), may contribute sign ifican tltly Association suggested between ADHD, parenting characteristics and serotonergic genotypes Swanson et al, 1998, NikolasSunohara M et al, G, Beh et al. and J Am Brain Acad Func Adolesc 2010 Psychiatry. (6) 23 2000;39:1537-1592. Giros B, et al. Nature. 1996;379:606-612. ArcosArcos--BurgosBurgos M, Jain M , et al Mol Psychiatry 2/16/10 ADHD and Copy Number Variants Comparison of genomegenome--widewide analysis in children with ADHD (366) -

Proton-Gated Coincidence Detection Is a Common Feature of GPCR Signaling
bioRxiv preprint doi: https://doi.org/10.1101/2021.01.08.425945; this version posted January 9, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. Proton-gated coincidence detection is a common feature of GPCR signaling Nicholas J. Kapolkaa, Jacob B. Rowea, Geoffrey J. Taghona, William M. Morgana, Corin R. O’Sheaa, and Daniel G. Isoma,b,c* From the aDepartment of Molecular and Cellular Pharmacology, University of Miami Miller School of Medicine, Miami, FL 33136; bUniversity of Miami Sylvester Comprehensive Cancer Center, Miami, FL 33136; and cUniversity of Miami Institute for Data Science and Computing, Miami, FL 33146 *Corresponding author: Daniel G. Isom: [email protected] Key Words: GPCR, coincidence detection, agonism, antagonism, protons, proton sensors, logic gating, Boolean logic, acidosis 1 bioRxiv preprint doi: https://doi.org/10.1101/2021.01.08.425945; this version posted January 9, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. Abstract The evolutionary expansion of G protein-coupled receptors (GPCRs) has produced a rich diversity of transmembrane sensors for many physical and chemical signals. In humans alone, over 800 GPCRs detect stimuli such as light, hormones, and metabolites to guide cellular decision making primarily using intracellular G protein signaling networks. -

Latrophilin 3, a Gene Involved in ADHD
National Human Genome Research Institute (NHGRI) Patented & Patent-pending Technologies Available for Licensing Latrophilin 3, a Gene Summary Involved in ADHD Attention Deficit Hyperactivity Disorder (ADHD) is the most commonly diagnosed childhood behavioral disorder, affecting about ten percent of children and NHGRI invention number: adolescents in the U.S. At this time there is no accurate way for doctors to determine E-312-2006/0 in advance which ADHD therapeutic drug is likely to be of most benefit to a particular E-187-2011/0 individual. Patent Status Scientists from the National Human Genome Research Institute (NHGRI) at the U.S. patent 8,003,406 National Institutes of Health (NIH) have identified specific latrophilin 3 (LPHN3) issued August 23, 2011 mutations, groups of LPHN3 variants (e.g., LPHN3 alleles), and other loci that are U.S. provisional application associated with an increased susceptibility for and severity of ADHD. LPHN3 is a G- 61/505,864 filed protein coupled receptor specifically expressed in the brain’s mesolimbic system, July 8, 2011 which is part of a dopaminergic pathway. This LPHN3 protein expression pattern fits with the dopamine theory of ADHD, according to which patients affected with the Lead Inventor disorder have lower levels of dopamine receptors and transporters. Maximilian Muenke, M.D. Potential Commercial Applications Chief, Medical Genetics Branch, NHGRI, NIH Identification of mutations in this ADHD susceptibility gene could be used as the basis for both a DNA-based ADHD diagnostic test as well as used to aid in the Licensing Contact development and clinical validation of more efficient and individualized therapies. -

RT² Profiler PCR Array (384-Well Format) Mouse G Protein Coupled Receptors 384HT
RT² Profiler PCR Array (384-Well Format) Mouse G Protein Coupled Receptors 384HT Cat. no. 330231 PAMM-3009ZE For pathway expression analysis Format For use with the following real-time cyclers RT² Profiler PCR Array, Applied Biosystems® models 7900HT (384-well block), Format E ViiA™ 7 (384-well block); Bio-Rad CFX384™ RT² Profiler PCR Array, Roche® LightCycler® 480 (384-well block) Format G Description The Mouse G Protein Coupled Receptors 384HT RT² Profiler™ PCR Array profiles the expression of a comprehensive panel of 370 genes encoding the most important G Protein Coupled Receptors (GPCR). GPCR regulate a number of normal biological processes and play roles in the pathophysiology of many diseases upon dysregulation of their downstream signal transduction activities. As a result, they represent 30 percent of the targets for all current drug development. Developing drug screening assays requires a survey of which GPCR the chosen cell-based model system expresses, to determine not only the expression of the target GPCR, but also related GPCR to assess off-target side effects. Expression of other unrelated GPCR (even orphan receptors whose ligand are unknown) may also correlate with off-target side effects. The ligands that bind and activate the receptors on this array include neurotransmitters and neuropeptides, hormones, chemokines and cytokines, lipid signaling molecules, light-sensitive compounds, and odorants and pheromones. The normal biological processes regulated by GPCR include, but are not limited to, behavioral and mood regulation (serotonin, dopamine, GABA, glutamate, and other neurotransmitter receptors), autonomic (sympathetic and parasympathetic) nervous system transmission (blood pressure, heart rate, and digestive processes via hormone receptors), inflammation and immune system regulation (chemokine receptors, histamine receptors), vision (opsins like rhodopsin), and smell (olfactory receptors for odorants and vomeronasal receptors for pheromones).