The Feasibility of Patient-Specific Circulating Tumor DNA Monitoring
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Directors Arup Medical Directors and Consulting Faculty | 2015
MEDICAL DIRECTORS ARUP MEDICAL DIRECTORS AND CONSULTING FACULTY | 2015 MAY 2015 www.aruplab.com Information in this brochure is current as of May 2015. All content is subject to change. Please contact ARUP Client Services at (800) 522-2787 with any questions or concerns. ARUP LABORATORIES ARUP Laboratories is a national clinical and anatomic pathology reference laboratory and a nonprofit enterprise of the University of Utah and its Department of Pathology. Located in Salt Lake City, Utah, ARUP offers in excess of 3,000 tests and test combinations, ranging from routine screening tests to esoteric molecular and genetic assays. Rather than competing with its clients for physician office business, ARUP chooses instead to support clients’ existing test menus by offering complex and unique tests, with accompanying consultative support, to enhance their abilities to provide local and regional laboratory services. ARUP’s clients include many of the nation’s university teaching hospitals and children’s hospitals, as well as multihospital groups, major commercial laboratories, group purchasing organizations, military and other government facilities, and major clinics. In addition, ARUP is a worldwide leader in innovative laboratory research and development, led by the efforts of the ARUP Institute for Clinical and Experimental Pathology®. Since its formation in 1984 by the Department of Pathology at the University of Utah, ARUP has founded its reputation on reliable and consistent laboratory testing and service. This simple strategy contributes significantly to client satisfaction. When ARUP conducts surveys, clients regularly rate ARUP highly and respond that they would recommend ARUP to others. As the most responsive source of quality information and knowledge, ARUP strives to be the reference laboratory of choice for community healthcare systems. -

Circulating Tumor DNA As Biomarkers for Cancer Detection
Genomics Proteomics Bioinformatics 15 (2017) 59–72 HOSTED BY Genomics Proteomics Bioinformatics www.elsevier.com/locate/gpb www.sciencedirect.com REVIEW Circulating Tumor DNA as Biomarkers for Cancer Detection Xiao Han 1,2,a, Junyun Wang 1,b, Yingli Sun 1,*,c 1 CAS Key Laboratory of Genomic and Precision Medicine, China Gastrointestinal Cancer Research Center, Beijing Institute of Genomics, Chinese Academy of Sciences, Beijing 100101, China 2 University of Chinese Academy of Sciences, Beijing 100049, China Received 1 July 2016; revised 13 December 2016; accepted 20 December 2016 Available online 7 April 2017 Handled by Cesar Wong KEYWORDS Abstract Detection of circulating tumor DNAs (ctDNAs) in cancer patients is an important com- Precision medicine; ponent of cancer precision medicine ctDNAs. Compared to the traditional physical and biochemical Liquid biopsy; methods, blood-based ctDNA detection offers a non-invasive and easily accessible way for cancer Circulating tumor DNA; diagnosis, prognostic determination, and guidance for treatment. While studies on this topic are Biomarker; currently underway, clinical translation of ctDNA detection in various types of cancers has been Clinical diagnosis; attracting much attention, due to the great potential of ctDNA as blood-based biomarkers for early Cell-free nucleic acids diagnosis and treatment of cancers. ctDNAs are detected and tracked primarily based on tumor- related genetic and epigenetic alterations. In this article, we reviewed the available studies on ctDNA detection and described the representative methods. We also discussed the current understanding of ctDNAs in cancer patients and their availability as potential biomarkers for clin- ical purposes. Considering the progress made and challenges involved in accurate detection of speci- fic cell-free nucleic acids, ctDNAs hold promise to serve as biomarkers for cancer patients, and further validation is needed prior to their broad clinical use. -

Understanding Your Pathology Report: Benign Breast Conditions
cancer.org | 1.800.227.2345 Understanding Your Pathology Report: Benign Breast Conditions When your breast was biopsied, the samples taken were studied under the microscope by a specialized doctor with many years of training called a pathologist. The pathologist sends your doctor a report that gives a diagnosis for each sample taken. Information in this report will be used to help manage your care. The questions and answers that follow are meant to help you understand medical language you might find in the pathology report from a breast biopsy1, such as a needle biopsy or an excision biopsy. In a needle biopsy, a hollow needle is used to remove a sample of an abnormal area. An excision biopsy removes the entire abnormal area, often with some of the surrounding normal tissue. An excision biopsy is much like a type of breast-conserving surgery2 called a lumpectomy. What does it mean if my report uses any of the following terms: adenosis, sclerosing adenosis, apocrine metaplasia, cysts, columnar cell change, columnar cell hyperplasia, collagenous spherulosis, duct ectasia, columnar alteration with prominent apical snouts and secretions (CAPSS), papillomatosis, or fibrocystic changes? All of these are terms that describe benign (non-cancerous) changes that the pathologist might see under the microscope. They do not need to be treated. They are of no concern when found along with cancer. More information about many of these can be found in Non-Cancerous Breast Conditions3. What does it mean if my report says fat necrosis? Fat necrosis is a benign condition that is not linked to cancer risk. -

797 Circulating Tumor DNA and Circulating Tumor Cells for Cancer
Medical Policy Circulating Tumor DNA and Circulating Tumor Cells for Cancer Management (Liquid Biopsy) Table of Contents • Policy: Commercial • Coding Information • Information Pertaining to All Policies • Policy: Medicare • Description • References • Authorization Information • Policy History • Endnotes Policy Number: 797 BCBSA Reference Number: 2.04.141 Related Policies Biomarkers for the Diagnosis and Cancer Risk Assessment of Prostate Cancer, #336 Policy1 Commercial Members: Managed Care (HMO and POS), PPO, and Indemnity Plasma-based comprehensive somatic genomic profiling testing (CGP) using Guardant360® for patients with Stage IIIB/IV non-small cell lung cancer (NSCLC) is considered MEDICALLY NECESSARY when the following criteria have been met: Diagnosis: • When tissue-based CGP is infeasible (i.e., quantity not sufficient for tissue-based CGP or invasive biopsy is medically contraindicated), AND • When prior results for ALL of the following tests are not available: o EGFR single nucleotide variants (SNVs) and insertions and deletions (indels) o ALK and ROS1 rearrangements o PDL1 expression. Progression: • Patients progressing on or after chemotherapy or immunotherapy who have never been tested for EGFR SNVs and indels, and ALK and ROS1 rearrangements, and for whom tissue-based CGP is infeasible (i.e., quantity not sufficient for tissue-based CGP), OR • For patients progressing on EGFR tyrosine kinase inhibitors (TKIs). If no genetic alteration is detected by Guardant360®, or if circulating tumor DNA (ctDNA) is insufficient/not detected, tissue-based genotyping should be considered. Other plasma-based CGP tests are considered INVESTIGATIONAL. CGP and the use of circulating tumor DNA is considered INVESTIGATIONAL for all other indications. 1 The use of circulating tumor cells is considered INVESTIGATIONAL for all indications. -

Simple Technique to Identify Haemosiderin in Immunoperoxidase Stained Sections
J Clin Pathol: first published as 10.1136/jcp.37.10.1190 on 1 October 1984. Downloaded from 1190 Technical methods Phosphate buffer at pH 8*0 gave the sharpest 2 Rozenszajn L, Leibovich M, Shoham D, Epstein J. The esterase staining reactions, although there was little differ- activity in megaloblasts, leukaemic and normal haemopoietic cells. Br J Haematol 1968; 14:605-19. ence at pH 7-0 or pH 7-5. As the buffer pH was 3Hayhoe FGJ, Quaglino D. Haematological cytochemistry. Edin- increased above pH 8-0 staining with both substrates burgh: Churchill Livingstone, 1980. became progressively weaker, especially above pH 4Li CY, Lam KW, Yam LT. Esterases in human leucocytes. J 9.0. Below pH 7-0 staining with a-naphthyl butyrate Histochem Cytochem 1973;21:1-12. Yam LT, Li CY, Crosby WH. Cytochemical identification of became weaker, and below pH 5*0 staining with monocytes and granulocytes. Am J Clin Pathol 1971;55:283- naphthol AS-D chloroacetate began to disappear. 90. 6 Armitage RJ, Linch DC, Worman CP, Cawley JC. The morphol- This work was supported by a Medical Research ogy and cytochemistry of human T-cell subpopulations defined by monoclonal antibodies and Fc receptors. Br J Haematol Council project grant. I thank Professor FGJ 1983;51:605-13. Hayhoe for valuable advice. References Requests for reprints to: Dr DM Swirsky, Department of Gomori G. Chloroacyl esters as histochemical substrates. J His- Haematological Medicine, University Clinical School, Hills tochem Cytochem 1953;1:469-70. Road, Cambridge CB2 2QL, England. Simple technique to identify identification of the two compounds on the same haemosiderin in slide. -

The Interplay of Circulating Tumor DNA and Chromatin Modification
Zhang et al. Molecular Cancer (2019) 18:36 https://doi.org/10.1186/s12943-019-0989-z REVIEW Open Access The interplay of circulating tumor DNA and chromatin modification, therapeutic resistance, and metastasis Lei Zhang1,2,3†, Yiyi Liang1,2,3†, Shifu Li1,2,3†, Fanyuan Zeng1,2,3†, Yongan Meng1,2,3†, Ziwei Chen1,2,3, Shuang Liu3, Yongguang Tao1,2,3,4* and Fenglei Yu4* Abstract Peripheral circulating free DNA (cfDNA) is DNA that is detected in plasma or serum fluid with a cell-free status. For cancer patients, cfDNA not only originates from apoptotic cells but also from necrotic tumor cells and disseminated tumor cells that have escaped into the blood during epithelial-mesenchymal transition. Additionally, cfDNA derived from tumors, also known as circulating tumor DNA (ctDNA), carries tumor-associated genetic and epigenetic changes in cancer patients, which makes ctDNA a potential biomarker for the early diagnosis of tumors, monitory and therapeutic evaluations, and prognostic assessments, among others, for various kinds of cancer. Moreover, analyses of cfDNA chromatin modifications can reflect the heterogeneity of tumors and have potential for predicting tumor drug resistance. Keywords: ctDNA, Chromatin modification, Therapeutic resistance, Metastasis, Tumor heterogeneity Biological features of ctDNA enter the blood in single, double or triple forms, and Peripheral circulating free DNA is DNA that is detected thus most ctDNA shows significant fragmentary charac- in plasma or serum fluid with a cell-free status. It may teristics. Moreover, the half-life of ctDNA in the blood originate from apoptosis or necrosis. The amount of cir- circulation is less than 2 hours [3]. -

Circulating Cell-Free DNA in Hepatocellular Carcinoma: Current Insights and Outlook
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by edoc REVIEW published: 26 March 2018 doi: 10.3389/fmed.2018.00078 Circulating Cell-Free DNA in Hepatocellular Carcinoma: Current Insights and Outlook Charlotte K. Y. Ng1,2*, Giovan Giuseppe Di Costanzo3, Luigi M. Terracciano1 and Salvatore Piscuoglio1* 1 Institute of Pathology, University Hospital Basel, Basel, Switzerland, 2 Department of Biomedicine, Hepatology Laboratory, University of Basel, Basel, Switzerland, 3 Department of Transplantation – Liver Unit, Cardarelli Hospital, Naples, Italy Over the past decade, the advancements in massively parallel sequencing have pro- vided a new paradigm in biomedical research to uncover the genetic basis of human diseases. Integration of ‘omics information has begun transforming clinical management of cancer patients in terms of diagnostics and treatment options, giving rise to the era of precision medicine. Currently, nucleic acids for molecular profiling for patients diagnosed with hepatocellular carcinoma (HCC) are typically obtained from resected tumor mate- rials or transplanted neoplastic liver and occasionally from biopsies. Given the intrinsic risks associated with such invasive procedures, circulating cell-free DNA (cfDNA) has been proposed as an alternative source for tumor DNA. Circulating cfDNA is a type of Edited by: cell-free nucleic acid that derives from apoptotic, necrotic, as well as living eukaryotic Venancio Avancini Alves, University of São Paulo, Brazil cells. Importantly, the detection of abnormal forms of circulating cfDNA that originate Reviewed by: from cancer cells provides a new tool for cancer detection, disease monitoring, and Fernando Schmitt, molecular profiling. Currently, cfDNA is beginning to be adopted into clinical practice Universidade do Porto, Portugal Marco Volante, as a non-invasive tool to monitor disease by tracking the evolution of disease-specific Università degli Studi di Torino, Italy genetic alterations in several major cancer types. -

Highly Sensitive Detection of Sentinel Lymph Node Metastasis of Breast Cancer by Digital PCR for RASSF1A Methylation
2382 ONCOLOGY REPORTS 42: 2382-2389, 2019 Highly sensitive detection of sentinel lymph node metastasis of breast cancer by digital PCR for RASSF1A methylation MIZUHO ABE, NAOFUMI KAGARA, TOMOHIRO MIYAKE, TOMONORI TANEI, YASUTO NAOI, MASAFUMI SHIMODA, KENZO SHIMAZU, SEUNG JIN KIM and SHINZABURO NOGUCHI Department of Breast and Endocrine Surgery, Osaka University Graduate School of Medicine, Suita, Osaka 565‑0871, Japan Received June 4, 2019; Accepted September 19, 2019 DOI: 10.3892/or.2019.7363 Abstract. One-step nucleic acid amplification (OSNA) and is confirmed by postoperative pathological examination of targeting cytokeratin 19 (CK19) mRNA expression and permanent sections (3,4). One‑step nucleic acid amplification pathological examination are widely used for the intraop- (OSNA) can be used to detect SN metastasis through the erative diagnosis of sentinel node (SN) metastasis. The aim amplification of cytokeratin 19 (CK19) mRNA (which is of the present study was to develop a novel assay for detecting expressed in tumor cells, but not normal cells of LNs) with the SN metastasis by targeting Ras association domain-containing same accuracy as routine pathological examination (5). OSNA protein 1 (RASSF1A) methylation in tumor cells, and to is also used to determine total tumor load (TTL) in SNs as the compare its performance with OSNA. Using digital PCR with sum of CK19 mRNA copies, which is reportedly useful for methylation-specific restriction enzymes (RE-dMSP), our predicting non-SN metastatic status (6,7), as well as patient assay was able to detect ≥3 copies of methylated DNA per prognosis (8). well, and was ≥10 times more sensitive than real‑time PCR However, TTL determination by OSNA does not always with bisulfite modification. -

Circulating Tumor DNA Testing Opens New Perspectives in Melanoma Management
cancers Review Circulating Tumor DNA Testing Opens New Perspectives in Melanoma Management 1, 1, 1 1 Alessandra Sacco y , Laura Forgione y, Marianeve Carotenuto , Antonella De Luca , Paolo A. Ascierto 2 , Gerardo Botti 3 and Nicola Normanno 1,* 1 Cell Biology and Biotherapy Unit, Istituto Nazionale Tumori-IRCCS-Fondazione G. Pascale, 80131 Naples, Italy; [email protected] (A.S.); [email protected] (L.F.); [email protected] (M.C.); [email protected] (A.D.L.) 2 Department of Melanoma, Cancer Immunotherapy and Development Therapeutics, Istituto Nazionale Tumori IRCCS Fondazione Pascale, 80131 Napoli, Italy; [email protected] 3 Scientific Direction, Istituto Nazionale Tumori IRCCS Fondazione Pascale, 80131 Napoli, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-081-5903-826 These authors contributed equally to this work. y Received: 15 September 2020; Accepted: 8 October 2020; Published: 10 October 2020 Simple Summary: Melanoma, like other solid tumors, releases DNA molecules that are referred to as circulating tumor DNA (ctDNA), into the blood and other biological fluids. ctDNA analysis performed with molecular biology techniques can provide important information on the aggressiveness of the disease and its genetic characteristics. This review aims to highlight all the possible clinical applications of ctDNA analysis that can contribute to an improvement in the diagnosis and therapy of melanoma. Abstract: Malignant melanoma accounts for about 1% of all skin cancers, but it causes most of the skin cancer-related deaths. Circulating tumor DNA (ctDNA) testing is emerging as a relevant tool for the diagnosis and monitoring of cancer. -

Consensus Guideline on Concordance Assessment of Image-Guided Breast Biopsies and Management of Borderline Or High-Risk Lesions
- Official Statement - Consensus Guideline on Concordance Assessment of Image-Guided Breast Biopsies and Management of Borderline or High-Risk Lesions Purpose To outline the management approach for borderline and high risk lesions identified on image-guided breast biopsy. Associated ASBrS Guidelines or Quality Measures 1. Image-Guided Percutaneous Biopsy of Palpable and Nonpalpable Breast Lesions 2. Performance and Practice Guidelines for Stereotactic Breast Procedures 3. Concordance Assessment Following Image-Guided Breast Biopsy Methods Literature review inclusive of recent randomized controlled trials evaluating the management of various borderline and high-risk lesions (including atypical hyperplasia, lobular neoplasia, papillary lesions, radial scars and complex sclerosing lesions, fibroepithelial lesions, mucocele-like lesions, spindle cell lesions, and pseudoangiomatous stromal hyperplasia [PASH]) identified on image-guided breast biopsies. This is not a complete systematic review but a comprehensive review of the modern literature on this subject. The ASBS Research Committee developed a consensus document which the ASBS Board of Directors reviewed and approved. Summary of Data Reviewed Percutaneous core needle biopsy (CNB) is the preferred, initial, minimally invasive diagnostic procedure for nonpalpable breast lesions or palpable breast masses.1 Concordance assessment of the histologic, imaging, and clinical findings determines further management. Discordance refers to the situation in which a breast CNB demonstrates benign histology, while the clinical or imaging findings are suspicious for malignancy. If there is discordance between imaging and pathology, histological evaluation is still needed. This can be accomplished either by repeat CNB, perhaps with consideration of larger gauge or vacuum- assisted device, or surgical excision.2-5 Some nonmalignant CNB findings are considered “borderline” because of their potential association with malignancy. -
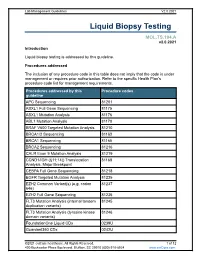
Liquid Biopsy Testing
Lab Management Guidelines V2.0.2021 Liquid Biopsy Testing MOL.TS.194.A v2.0.2021 Introduction Liquid biopsy testing is addressed by this guideline. Procedures addressed The inclusion of any procedure code in this table does not imply that the code is under management or requires prior authorization. Refer to the specific Health Plan's procedure code list for management requirements. Procedures addressed by this Procedure codes guideline APC Sequencing 81201 ASXL1 Full Gene Sequencing 81175 ASXL1 Mutation Analysis 81176 ABL1 Mutation Analysis 81170 BRAF V600 Targeted Mutation Analysis 81210 BRCA1/2 Sequencing 81163 BRCA1 Sequencing 81165 BRCA2 Sequencing 81216 CALR Exon 9 Mutation Analysis 81219 CCND1/IGH (t(11;14)) Translocation 81168 Analysis, Major Breakpoint CEBPA Full Gene Sequencing 81218 EGFR Targeted Mutation Analysis 81235 EZH2 Common Variant(s) (e.g. codon 81237 646) EZH2 Full Gene Sequencing 81236 FLT3 Mutation Analysis (internal tandem 81245 duplication variants) FLT3 Mutation Analysis (tyrosine kinase 81246 domain variants) FoundationOne Liquid CDx 0239U Guardant360 CDx 0242U ©2021 eviCore healthcare. All Rights Reserved. 1 of 12 400 Buckwalter Place Boulevard, Bluffton, SC 29910 (800) 918-8924 www.eviCore.com Lab Management Guidelines V2.0.2021 Procedures addressed by this Procedure codes guideline Hematolymphoid Neoplasm Molecular 81450 Profiling; 5-50 genes IDH1 Mutation Analysis 81120 IDH2 Mutation Analysis 81121 IGH@/BCL2 (t(14;18)) Translocation 81278 Analysis, Major Breakpoint Region (MBR) and Minor Cluster Region (mcr) Breakpoints JAK2 Targeted Mutation Analysis (e.g 81279 exons 12 and 13) JAK2 V617F Targeted Mutation Analysis 81270 KIT Targeted Sequence Analysis 81272 KIT D816 Targeted Mutation Analysis 81273 KRAS Exon 2 Targeted Mutation Analysis 81275 KRAS Targeted Mutation Analysis, 81276 Additional Variants MGMT Promoter Methylation Analysis 81287 MLH1 Sequencing 81292 Molecular Tumor Marker Test 81400 81401 81402 g 81403 n i 81405 t 81406 s e T 81407 81408 y s 81479 p o Molecular Tumor Marker Test 88271 i B MPL Common Variants (e.g. -

Neratinib Efficacy and Circulating Tumor DNA Detection of HER2 Mutations in HER2 Nonamplified Metastatic Breast Cancer
Published OnlineFirst July 5, 2017; DOI: 10.1158/1078-0432.CCR-17-0900 Cancer Therapy: Clinical Clinical Cancer Research Neratinib Efficacy and Circulating Tumor DNA Detection of HER2 Mutations in HER2 Nonamplified Metastatic Breast Cancer Cynthia X. Ma1, Ron Bose1, Feng Gao2, Rachel A. Freedman3, Melinda L. Telli4, Gretchen Kimmick5, Eric Winer3, Michael Naughton1, Matthew P. Goetz6, Christy Russell7, Debu Tripathy7, Melody Cobleigh8, Andres Forero9, Timothy J. Pluard10, Carey Anders11, Polly Ann Niravath12, Shana Thomas1, Jill Anderson1, Caroline Bumb1, Kimberly C. Banks13, Richard B. Lanman13, Richard Bryce14, Alshad S. Lalani14, John Pfeifer15, Daniel F. Hayes16, Mark Pegram17, Kimberly Blackwell5, Philippe L. Bedard18, Hussam Al-Kateb15, and Matthew J.C. Ellis12 Abstract Purpose: Based on promising preclinical data, we conducted a Sixteen patients [median age 58 (31–74) years and three (2–10) single-arm phase II trial to assess the clinical benefit rate (CBR) of prior metastatic regimens] received neratinib. The CBR was 31% neratinib, defined as complete/partial response (CR/PR) or stable [90% confidence interval (CI), 13%–55%], including one CR, one disease (SD) 24 weeks, in HER2mut nonamplified metastatic PR, and three SD 24 weeks. Median PFS was 16 (90% CI, 8–31) breast cancer (MBC). Secondary endpoints included progression- weeks. Diarrhea (grade 2, 44%; grade 3, 25%) was the most free survival (PFS), toxicity, and circulating tumor DNA (ctDNA) common adverse event. Baseline ctDNA sequencing identified HER2mut detection. the same HER2mut in 11 of 14 tumor-positive cases (sensitivity, Experimental Design: Tumor tissue positive for HER2mut was 79%; 90% CI, 53%–94%) and correctly assigned 32 of 32 infor- required for eligibility.