Lewis Structure Handout
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Valence Bond Theory
UMass Boston, Chem 103, Spring 2006 CHEM 103 Molecular Geometry and Valence Bond Theory Lecture Notes May 2, 2006 Prof. Sevian Announcements z The final exam is scheduled for Monday, May 15, 8:00- 11:00am It will NOT be in our regularly scheduled lecture hall (S- 1-006). The final exam location has been changed to Snowden Auditorium (W-1-088). © 2006 H. Sevian 1 UMass Boston, Chem 103, Spring 2006 More announcements Information you need for registering for the second semester of general chemistry z If you will take it in the summer: z Look for chem 104 in the summer schedule (includes lecture and lab) z If you will take it in the fall: z Look for chem 116 (lecture) and chem 118 (lab). These courses are co-requisites. z If you plan to re-take chem 103, in the summer it will be listed as chem 103 (lecture + lab). In the fall it will be listed as chem 115 (lecture) + chem 117 (lab), which are co-requisites. z Note: you are only eligible for a lab exemption if you previously passed the course. Agenda z Results of Exam 3 z Molecular geometries observed z How Lewis structure theory predicts them z Valence shell electron pair repulsion (VSEPR) theory z Valence bond theory z Bonds are formed by overlap of atomic orbitals z Before atoms bond, their atomic orbitals can hybridize to prepare for bonding z Molecular geometry arises from hybridization of atomic orbitals z σ and π bonding orbitals © 2006 H. Sevian 2 UMass Boston, Chem 103, Spring 2006 Molecular Geometries Observed Tetrahedral See-saw Square planar Square pyramid Lewis Structure Theory -

Introduction to Molecular Orbital Theory
Chapter 2: Molecular Structure and Bonding Bonding Theories 1. VSEPR Theory 2. Valence Bond theory (with hybridization) 3. Molecular Orbital Theory ( with molecualr orbitals) To date, we have looked at three different theories of molecular boning. They are the VSEPR Theory (with Lewis Dot Structures), the Valence Bond theory (with hybridization) and Molecular Orbital Theory. A good theory should predict physical and chemical properties of the molecule such as shape, bond energy, bond length, and bond angles.Because arguments based on atomic orbitals focus on the bonds formed between valence electrons on an atom, they are often said to involve a valence-bond theory. The valence-bond model can't adequately explain the fact that some molecules contains two equivalent bonds with a bond order between that of a single bond and a double bond. The best it can do is suggest that these molecules are mixtures, or hybrids, of the two Lewis structures that can be written for these molecules. This problem, and many others, can be overcome by using a more sophisticated model of bonding based on molecular orbitals. Molecular orbital theory is more powerful than valence-bond theory because the orbitals reflect the geometry of the molecule to which they are applied. But this power carries a significant cost in terms of the ease with which the model can be visualized. One model does not describe all the properties of molecular bonds. Each model desribes a set of properties better than the others. The final test for any theory is experimental data. Introduction to Molecular Orbital Theory The Molecular Orbital Theory does a good job of predicting elctronic spectra and paramagnetism, when VSEPR and the V-B Theories don't. -
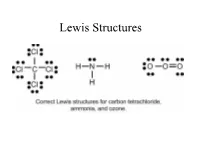
Lewis Structures
Lewis Structures Valence electrons for Elements Recall that the valence electrons for the elements can be determined based on the elements position on the periodic table. Lewis Dot Symbol Valence electrons and number of bonds Number of bonds elements prefers depending on the number of valence electrons. In general - F a m i l y → # C o v a l e n t B o n d s* H a l o g e n s X F , B r , C l , I → 1 bond often C a l c o g e n s O 2 bond often O , S → N i t r o g e n N 3 bond often N , P → C a r b o n C → 4 bond always C , S i The above chart is a guide on the number of bonds formed by these atoms. Lewis Structure, Octet Rule Guidelines When compounds are formed they tend to follow the Octet Rule. Octet Rule: Atoms will share electrons (e-) until it is surrounded by eight valence electrons. 4 unpaired 3unpaired 2unpaired 1unpaired up = unpaired e- 4 bonds 3 bonds 2 bonds 1 bond O=C=O N≡ N O = O F - F Atomic Connectivity The atomic arrangement for a molecule is usually given. CH2ClF HNO3 CH3COOH H2SO4 Cl N H O O O O H C F O H C C H O S O H H H H O H O In general when there is a single central atom in the 3- molecule, CH2ClF, SeCl2, O3 (CO2, NH3, PO4 ), the central atom is the first atom in the chemical formula. -

Draw Three Resonance Structures for the Chlorate Ion, Clo3
University Chemistry Quiz 3 2014/11/6 + 1. (10%) Explain why the bond order of N2 is greater than that of N2 , but the bond + order of O2 is less than that of O2 . Sol. + In forming the N2 from N2, an electron is removed from the sigma bonding molecular orbital. Consequently, the bond order decreases to 2.5 from 3.0. In + forming the O2 ion from O2, an electron is removed from the pi antibonding molecular orbital. Consequently, the bond order increases to 2.5 from 2.0. - 2. (10%) Draw three resonance structures for the chlorate ion, ClO3 . Show formal charges. Sol. Strategy: We follow the procedure for drawing Lewis structures outlined in Section 3.4 of the text. After we complete the Lewis structure, we draw the resonance structures. Solution: Following the procedure in Section 3.4 of the text, we come up with − the following Lewis structure for ClO3 . O − + − O Cl O We can draw two more equivalent Lewis structures with the double bond between Cl and a different oxygen atom. The resonance structures with formal charges are as follows: − − O O O + − − + − − + O Cl O O Cl O O Cl O 3. (5%) Use the molecular orbital energy-level diagram for O2 to show that the following Lewis structure corresponds to an excited state: Sol. The Lewis structure shows 4 pairs of electrons on the two oxygen atoms. From Table 3.4 of the text, we see that these 8 valence electrons are placed in the σ p p p p 2p , 2p , 2p , 2p , and 2p orbitals. -

Molecular Orbital Theory
Molecular Orbital Theory The Lewis Structure approach provides an extremely simple method for determining the electronic structure of many molecules. It is a bit simplistic, however, and does have trouble predicting structures for a few molecules. Nevertheless, it gives a reasonable structure for many molecules and its simplicity to use makes it a very useful tool for chemists. A more general, but slightly more complicated approach is the Molecular Orbital Theory. This theory builds on the electron wave functions of Quantum Mechanics to describe chemical bonding. To understand MO Theory let's first review constructive and destructive interference of standing waves starting with the full constructive and destructive interference that occurs when standing waves overlap completely. When standing waves only partially overlap we get partial constructive and destructive interference. To see how we use these concepts in Molecular Orbital Theory, let's start with H2, the simplest of all molecules. The 1s orbitals of the H-atom are standing waves of the electron wavefunction. In Molecular Orbital Theory we view the bonding of the two H-atoms as partial constructive interference between standing wavefunctions of the 1s orbitals. The energy of the H2 molecule with the two electrons in the bonding orbital is lower by 435 kJ/mole than the combined energy of the two separate H-atoms. On the other hand, the energy of the H2 molecule with two electrons in the antibonding orbital is higher than two separate H-atoms. To show the relative energies we use diagrams like this: In the H2 molecule, the bonding and anti-bonding orbitals are called sigma orbitals (σ). -

Chemical Bonding II: Molecular Shapes, Valence Bond Theory, and Molecular Orbital Theory Review Questions
Chemical Bonding II: Molecular Shapes, Valence Bond Theory, and Molecular Orbital Theory Review Questions 10.1 J The properties of molecules are directly related to their shape. The sensation of taste, immune response, the sense of smell, and many types of drug action all depend on shape-specific interactions between molecules and proteins. According to VSEPR theory, the repulsion between electron groups on interior atoms of a molecule determines the geometry of the molecule. The five basic electron geometries are (1) Linear, which has two electron groups. (2) Trigonal planar, which has three electron groups. (3) Tetrahedral, which has four electron groups. (4) Trigonal bipyramid, which has five electron groups. (5) Octahedral, which has six electron groups. An electron group is defined as a lone pair of electrons, a single bond, a multiple bond, or even a single electron. H—C—H 109.5= ijj^^jl (a) Linear geometry \ \ (b) Trigonal planar geometry I Tetrahedral geometry I Equatorial chlorine Axial chlorine "P—Cl: \ Trigonal bipyramidal geometry 1 I Octahedral geometry I 369 370 Chapter 10 Chemical Bonding II The electron geometry is the geometrical arrangement of the electron groups around the central atom. The molecular geometry is the geometrical arrangement of the atoms around the central atom. The electron geometry and the molecular geometry are the same when every electron group bonds two atoms together. The presence of unbonded lone-pair electrons gives a different molecular geometry and electron geometry. (a) Four electron groups give tetrahedral electron geometry, while three bonding groups and one lone pair give a trigonal pyramidal molecular geometry. -
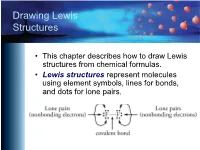
Drawing Lewis Structures
Drawing Lewis Structures • This chapter describes how to draw Lewis structures from chemical formulas. • Lewis structures represent molecules using element symbols, lines for bonds, and dots for lone pairs. Short Procedue for Drawing Lewis Structures • The first procedure involves drawing Lewis structures by attempting to give each atom in a molecule its most common bonding pattern. Most Common Bonding Patterns for Nonmetals Element # Bonds # lone pairs H 1 0 C 4 0 N, P 3 1 O, S, Se 2 2 F, Cl, Br, I 1 3 Most Common Bonding Patterns for Nonmetals https://preparatorychemistry.com/Bishop_periodic_table.pdf Example 1 Short Technique Methane, CH4 • Hydrogen atoms have 1 bond and no lone pairs. • Carbon atoms usually have 4 bonds and no lone pairs. Example 2 Short Technique Ammonia, NH3 • Hydrogen atoms have 1 bond and no lone pairs. • Nitrogen atoms usually have 3 bonds and 1 lone pair. Example 3 Short Technique Water, H2O • Hydrogen atoms have 1 bond and no lone pairs. • Oxygen atoms usually have 2 bonds and 2 lone pairs. Example 4 Short Technique Hypochlorous acid, HOCl • Hydrogen atoms have 1 bond and no lone pairs. • Oxygen atoms usually have 2 bonds and 2 lone pairs. • Chlorine atoms usually have one bond and three lone pairs. Example 5 Short Technique CFC-11, CCl3F • Carbon atoms usually have 4 bonds and no lone pairs. • Both fluorine and chlorine atoms usually have one bond and three lone pairs. Example 6 Short Technique Ethyne (acetylene), C2H2 (burned in oxyacetylene torches) • Carbon atoms usually have 4 bonds and no lone pairs. -

How Do We Know That There Are Atoms
Chemistry 11 Fall 2010 Discussion – 12/13/10 MOs and Hybridization 1. a. [:C N:]- Bond order = (8 – 2) / 2 = 3 *2p * 2p C 2p N 2p 2p 2p *2s C 2s N 2s 2s CN- b. Comparison of Lewis and MO models of CN- CN- Consistent: Both have triple bond (B.O. = 3) Inconsistent: - Lewis structure shows lone pairs on each atom rather than all e- being shared (MO) - Lewis structure indicates a negative formal charge on C, whereas MO suggests more electron density on N, which is consistent with EN considerations c. Both molecules exhibit sp mixing, and the MOs should reflect this. In N2, the molecular orbitals would be distributed equally between the two N atoms, whereas in CN-, the bonding orbitals have greater electron density on the more electronegative atom (N). The antibonding orbitals have more electron density on the less electronegative atom (C). - 1 - Chemistry 11 Fall 2010 2. a. Completed structure of guanine (carbons at each intersection are implied): * * * * b. 17 sigma bonds; 4 pi bonds. c. All carbons are sp2 hybridized; trigonal planar; with 120˚ bond angles. d. We predict that the three N’s marked with * are sp3 hybridized, with 109.5˚ bond angles. The remaining N atoms are sp2 hybridized, with 120˚ bond angles. e. i. Benzene: ii. For the structure drawn in (a), the bond angles in the six membered ring would be expected to be 120˚ for each atom except for the N§, which is predicted to have bond angles of 109.5˚. However, it is not possible to form a planar six-membered ring unless ALL the angles are 120˚. -

Localized Electron Model for Bonding What Does Hybridization Mean?
190 Localized electron model for bonding 1. Determine the Lewis Structure for the molecule 2. Determine resonance structures 3. Determine the shape of the molecule 4. Determine the hybridization of the atoms What does hybridization mean? Why hybridize? Look at the shape of many molecules, and compare this to the shape of the orbitals of each of the atoms. Shape of orbitals Orbitals on atoms are s, p, d, and f, and there are certain shapes associated with each orbital. an s orbital is a big sphere the f orbitals look like two four leaf the p orbitals looks like “8” ’s clovers each one points in along an axis three f orbitals look like weird “8”’s with two doughnuts around each four of the d orbitals look like four one leaf clovers one d orbital looks like an “8” with a doughnut around the middle Shape of molecules Remember we determine the shape of molecules by determining the number of sets of electrons around the atom. count double and triple bonds as one set, count single bonds as one set of electrons and count lone pairs as a set 2 sets—each pair of electrons points 180° away from the other 5 sets—each pair points toward the corner of a trigonal bipyramid 3 sets—each pair points toward the corner of a triangle 6 sets—each pair points toward the corners of an octahedron 4 sets—each pair points toward the corner of a tetrahedron 191 To make bonds we need orbitals—the electrons need to go some where. -

Understanding Hypervalency
Created by Gerard Rowe ([email protected]) and posted on VIPEr (www.ionicviper.org) on Nov 5, 2013. Copyright Gerard Rowe 2013. This work is licensed under the Creative Commons Attribution-NonCommerical-ShareAlike 3.0 Unported License. To view a copy of this license visit http://creativecommons.org/about/license/. Understanding Hypervalency In the world of quantum chemistry, there are two prevailing theories that explain chemical bonding: valence bond (VB) theory and molecular orbital (MO) theory. By this point in time, you have had exposure to both. The concepts of equivalent hybrid orbitals and resonance structures are part of VB theory, and VB orbitals generally only sit between two atoms. MO theory, on the other hand, uses orbitals that are delocalized; that is, they can span multiple atoms, or even the entire molecule. The interesting thing about these two theories is that they are both equally valid, and make the exact same predictions. Which one you use depends on the situation. The utility of VB hybrid orbitals lies in their simplicity in explaining structural features such as the four equivalent C-H bonds of methane (as does MO theory in a more complex manner). However, when one looks at the electronic spectrum of methane, two major peaks are observed. Hybrid orbital theory and molecular orbital theory predict very different orbital energy levels for methane (Figure 1). Using MO theory, it is very easy to see why there would be two peaks; electrons jump from the HOMO to each of the two higher energy levels (Figure 1b). With VB theory, it takes a lot of extra math to explain how there could be two peaks. -

Chem 115 Quiz #4
Name CHM 115 QUIZ #3 Practice Answer the following. Provide as much information as required to answer the question clearly. (5 points) 1. (a) Complete the LEWIS SYMBOLS of the elements listed below by adding the correct number of electrons to each: H C N O Cl (15 points) + 2. For the following molecules and ion CO, N2, NO, N2 , do the following: (a) Draw the Lewis structure (use resonance ONLY if it is needed). Also, calculate and SHOW the formal charge on each atom in each structure, (find the average formal charge if you used resonance structures) CO N2 :C≡O: :N≡N: -1 +1 0 0 + NO N2 :N=O + + ⋅N≡N: ÅÆ :N≡N⋅ 0 0 +1 0 0 +1 1 AveFC (N) = + /2 Which of the structures do not obey the octet rule? Explain why not? + The NO and the N2 do not obey the octet rule. In both of these cases, the species (molecule and ion) are odd electron species and MUST have an atom that does not have a full octet. In the case of NO, the oxygen is the atom that gets the full octet (it is more electronegative than is N) while N is left with seven electrons. However, both the O and the N both have formal charges of zero. In the case of N2+, resonance structures must be drawn to show that the ionic charge is uniformly shared by the two N in the ion. Each of the ions carries an average formal charge of 1 + /2. (continued) (8 points) 3. -

Chapter 12 Arenes and Aromaticity
CH. 12 Chapter 12 Arenes and Aromaticity Arenes Arenes are hydrocarbon derivatives of benzene. They are called aromatic systems due to their special stability (not due to their aroma!). The special stability results from the highly conjugated system with the electrons delocalized all the way around the ring. The name benzene comes from the Arabic luban jawi or “incense from Java” since it is isolated as a degradation product of gum benzoin. This is a balsam obtained from a tree that grows in Java and Sumatra. This degradation product is benzoic acid, which can be decarboxylated by heating with calcium oxide, CaO. O C OH CaO + CO2 heat benzoic acid benzene And tolu balsam from the South American tolu tree, when distilled, produces toluene or methylbenzene. The term aliphatic hydrocarbons, given to alkanes, alkenes, alkynes and benzene derivatives, comes from the Greek word aleiphar which means oil or unguent. Benzene Benzene has three double bonds that are conjugated and in a circular arrangement. Due to this conjugation and circular arrangement, the double bonds are much less reactive than normal, isolated alkenes. For example, benzene will not react with bromine or hydrogen gas in the presence of a metal catalyst, reactions that alkenes undergo readily. Br H Br2 CH3 CH CH CH3 CH3 C C CH3 rt H Br Br2 No Reaction rt 1 CH. 12 H2, Pd CH3 CH CH CH3 CH3CH2CH2CH3 rt H , Pd 2 No Reaction rt If we examine the structure of benzene we do not see a series of alternating double and single bonds as we would expect from the Lewis structure.