With ~ Ovules; Ovary (Disk) Flat
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
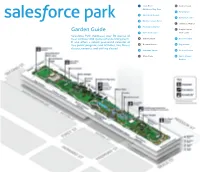
Salesforce Park Garden Guide
Start Here! D Central Lawn Children’s Play Area Garden Guide6 Palm Garden 1 Australian Garden Start Here! D Central Lawn Salesforce Park showcases7 California over Garden 50 species of Children’s Play Area 2 Mediterraneantrees and Basin over 230 species of understory plants. 6 Palm Garden -ã ¼ÜÊ ÊăØÜ ØÊèÜãE úØƀØÊèÃJapanese Maples ¼ÃØ Ê¢ 1 Australian Garden 3 Prehistoric¢ØÕ輫ÕØÊ£ØÂÜÃã«ó«ã«Üŧ¼«¹ĆãÃÜÜ Garden 7 California Garden ¼ÜÜÜŧÊÃØãÜŧÃØ¢ã«Ã£¼ÜÜÜũF Amphitheater Garden Guide 2 Mediterranean Basin 4 Wetland Garden Main Lawn E Japanese Maples Salesforce Park showcases over 50 species of 3 Prehistoric Garden trees and over 230 species of understory plants. A Oak Meadow 8 Desert Garden F Amphitheater It also offers a robust year-round calendar of 4 Wetland Garden Main Lawn free public programs and activities, like fitness B Bamboo Grove 9 Fog Garden Desert Garden classes, concerts, and crafting classes! A Oak Meadow 8 5 Redwood Forest 10 Chilean Garden B Bamboo Grove 9 Fog Garden C Main Plaza 11 South African 10 Chilean Garden Garden 5 Redwood Forest C Main Plaza 11 South African Garden 1 Children’s Australian Play Area Garden ABOUT THE GARDENS The botanist aboard the Endeavor, Sir Joseph Banks, is credited with introducing many plants from Australia to the western world, and many This 5.4 acre park has a layered soil system that plants today bear his name. balances seismic shifting, collects and filters storm- water, and irrigates the gardens. Additionally, the soil Native to eastern Australia, Grass Trees may grow build-up and dense planting help offset the urban only 3 feet in 100 years, and mature plants can be heat island effect by lowering the air temperature. -

Phylogenetic Models Linking Speciation and Extinction to Chromosome and Mating System Evolution
Phylogenetic Models Linking Speciation and Extinction to Chromosome and Mating System Evolution by William Allen Freyman A dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Integrative Biology and the Designated Emphasis in Computational and Genomic Biology in the Graduate Division of the University of California, Berkeley Committee in charge: Dr. Bruce G. Baldwin, Chair Dr. John P. Huelsenbeck Dr. Brent D. Mishler Dr. Kipling W. Will Fall 2017 Phylogenetic Models Linking Speciation and Extinction to Chromosome and Mating System Evolution Copyright 2017 by William Allen Freyman Abstract Phylogenetic Models Linking Speciation and Extinction to Chromosome and Mating System Evolution by William Allen Freyman Doctor of Philosophy in Integrative Biology and the Designated Emphasis in Computational and Genomic Biology University of California, Berkeley Dr. Bruce G. Baldwin, Chair Key evolutionary transitions have shaped the tree of life by driving the processes of spe- ciation and extinction. This dissertation aims to advance statistical and computational ap- proaches that model the timing and nature of these transitions over evolutionary trees. These methodological developments in phylogenetic comparative biology enable formal, model- based, statistical examinations of the macroevolutionary consequences of trait evolution. Chapter 1 presents computational tools for data mining the large-scale molecular sequence datasets needed for comparative phylogenetic analyses. I describe a novel metric, the miss- ing sequence decisiveness score (MSDS), which assesses the phylogenetic decisiveness of a matrix given the pattern of missing sequence data. In Chapter 2, I introduce a class of phylogenetic models of chromosome number evolution that accommodate both anagenetic and cladogenetic change. -

Phytochemical and Antimicrobial Studies of Ludwigia Hyssopifolia (G.Don) Exell Thet Su Hlaing1 Abstract Ludwigia Hyssopifolia (G.Don) Exell
2nd Myanmar Korea Conference Research Journal 433 Phytochemical and Antimicrobial Studies of Ludwigia hyssopifolia (G.Don) Exell Thet Su Hlaing1 Abstract Ludwigia hyssopifolia (G.Don) Exell. is annual herb belongs to the family Onagraceae in the order Myrtales. It is locally known as Taw-lay-hnyin. This plant was collected from Hinthada University Campus, Hinthada Township, Ayeyarwaddy Region. The morphological characters of vegetative and reproductive parts of the fresh specimens were investigated with the help of available literatures. The preliminary phytochemical tests were conducted from the powdered sample of the whole plant. The presence of alkaloid, phenolic compound, flavonoid, terpenoid, starch, reducing sugar, glycoside, saponin, tannin, α-amino acid and carbohydrate were found in the examination. Elemental analysis of powdered sample was examined by using EDXRF spectrometer. Percentage of calcium was higher than the other elements.Antimicrobial activities of various crude extracts were carried out by using paper disc diffusion assay with four test organisms. The methanolic extracts exhibited the highest against on different pathogenic microorganisms. These results showed that Ludwigia hyssopifolia (G.Don) Exell is endowed with many active constituents, minerals and antimicrobial properties that can be found useful in medicinal activities as well as possible application in nutrition and pharmacology. Key words : Ludwigia hyssopifolia (G.Don) Exell, phytochemical tests, antimicrobial activity Introduction Ludwigia hyssopifolia (G.Don) Exell. is commonly known as Taw-lay-hnyin in Myanmar and water primrose in English name. It is one of the traditionally used medicinal plants in Myanmar. It is distributed all over the world, particularly in both temperate and tropical regions of the world (Valkenburg, 2002). -

FULL ACCOUNT FOR: Fuchsia Boliviana Global Invasive Species
FULL ACCOUNT FOR: Fuchsia boliviana Fuchsia boliviana System: Terrestrial Kingdom Phylum Class Order Family Plantae Magnoliophyta Magnoliopsida Myrtales Onagraceae Common name Synonym Fuchsia boliviana , Carri?re var. luxurians Johnst.? Similar species Fuchsia magellanica, Fuchsia x exoniensis Summary Fuchsia boliviana is a small tree that develops rapidly. In tropical regions where it has been introduced (such as La Réunion or Hawaii), this plant tends to dominate native plant species. The dense foliage of large stands of Fuchsia boliviana intercept the light limiting the development of native understory species. view this species on IUCN Red List Principal source: Compiler: Comité français de l'UICN (IUCN French Committee) & IUCN SSC Invasive Species Specialist Group (ISSG) Review: Pubblication date: 2008-03-14 ALIEN RANGE [1] REUNION [1] SAINT HELENA BIBLIOGRAPHY 7 references found for Fuchsia boliviana Managment information Varnham, K. 2006. Non-native species in UK Overseas Territories: a review. JNCC Report 372. Peterborough: United Kingdom. Summary: This database compiles information on alien species from British Overseas Territories. Available from: http://www.jncc.gov.uk/page-3660 [Accessed 10 November 2009] General information Baret, S., Rouget, M., Richardson, D. M., Lavergne, C., Egoh, B., Dupont, J., & Strasberg, D. 2006. Current distribution and potential extent of the most invasive alien plant species on La R?union (Indian Ocean, Mascarene islands). Austral Ecology, 31, 747-758. Summary: L objectif de ce papier est d identifier les zones prioritaires en mati?re de gestion des invasions biologiques ? La R?union en mod?lisant la distribution actuelle et potentiellle d une s?lection de plantes parmi les plus envahissantes. -

Universidade Federal Do Recôncavo Da Bahia Centro De Ciências Agrárias, Ambientais E Biológicas
UNIVERSIDADE FEDERAL DO RECÔNCAVO DA BAHIA CENTRO DE CIÊNCIAS AGRÁRIAS, AMBIENTAIS E BIOLÓGICAS CARACTERES IMPORTANTES NA IDENTIFICAÇÃO DE ESPÉCIES DE LUDWIGIA L. (ONAGRACEAE) OCORRENTES NO RECÔNCAVO DA BAHIA, BRASIL NELMA XAVIER MARQUES DE SOUSA Bacharel em Biologia CRUZ DAS ALMAS BAHIA – BRASIL 2017 NELMA XAVIER MARQUES DE SOUSA CARACTERES IMPORTANTES NA IDENTIFICAÇÃO DE ESPÉCIES DE LUDWIGIA L. (ONAGRACEAE) OCORRENTES NO RECÔNCAVO DA BAHIA, BRASIL Trabalho de Conclusão de Curso apresentado à Universidade Federal do Recôncavo da Bahia, como parte das exigências do Curso de Graduação de Bacharelado em Biologia, para obtenção do título de Bacharel em Biologia. CRUZ DAS ALMAS BAHIA - BRASIL 2017 NELMA XAVIER MARQUES DE SOUSA CARACTERES IMPORTANTES NA IDENTIFICAÇÃO DE ESPÉCIES DE LUDWIGIA L. (ONAGRACEAE) OCORRENTES NO RECÔNCAVO DA BAHIA, BRASIL Trabalho de Conclusão de Curso apresentado à Universidade Federal do Recôncavo da Bahia, como parte das exigências do Curso de Graduação de Bacharelado em Biologia, para obtenção do título de Bacharel em Biologia. APROVADA: 03 de Fevereiro de 2017 I Dedicatória Aos meus pais (in memoriam), pela sabedoria de ter colocado a educação em primeiro lugar. “O mundo está nas mãos daqueles que têm a coragem de sonhar e de correr o risco de viver seus sonhos.” Paulo Coelho. II AGRADECIMENTOS Minha gratidão a professora Dra. Lidyanne Aona, por ser uma excelente professora e orientadora. Obrigada pelo suporte, por sempre estar presente, contribuindo muito com minha aprendizagem e ajudando-me em todos os trabalhos, pelo carinho que sempre teve comigo e, principalmente, pela paciência. A professora Dra. Ana Odete Vieira pelos ensinamentos, cooperação e importante contribuição. -

Vestured Pits in Wood of Onagraceae: Correlations with Ecology, Habit, and Phylogeny1
VESTURED PITS IN WOOD OF Sherwin Carlquist2 and Peter H. Raven3 ONAGRACEAE: CORRELATIONS WITH ECOLOGY, HABIT, AND PHYLOGENY1 ABSTRACT All Onagraceae for which data are available have vestured pits on vessel-to-vessel pit pairs. Vestures may also be present in some species on the vessel side of vessel-to-ray pit pairs. Herbaceous Onagraceae do not have fewer vestures, although woods with lower density (Circaea L. and Oenothera L.) have fewer vestures. Some Onagraceae from drier areas tend to have smaller vessel pits, and on that account may have fewer vestures (Epilobium L. and Megacorax S. Gonz´alez & W. L. Wagner). Pit apertures as seen on the lumen side of vessel walls are elliptical, occasionally oval, throughout the family. Vestures are predominantly attached to pit aperture margins. As seen from the outer surfaces of vessels, vestures may extend across the pit cavities. Vestures are usually absent or smaller on the distal portions of pit borders (except for Ludwigia L., which grows consistently in wet areas). Distinctive vesture patterns were observed in the several species of Lopezia Cav. and in Xylonagra Donn. Sm. & Rose. Vestures spread onto the lumen-facing vessel walls of Ludwigia octovalvis (Jacq.) P. H. Raven. Although the genera are presented here in the sequence of a recent molecular phylogeny of Onagraceae, ecology and growth forms are more important than evolutionary relationships with respect to abundance, degree of grouping, and morphology of vestured pits. Designation of vesture types is not warranted based on the distribution of named types in Onagraceae and descriptive adjectives seem more useful, although more data on vesturing in the family are needed before patterns of diversity and their extent can be fully ascertained. -

Rare Plants of Louisiana
Rare Plants of Louisiana Agalinis filicaulis - purple false-foxglove Figwort Family (Scrophulariaceae) Rarity Rank: S2/G3G4 Range: AL, FL, LA, MS Recognition: Photo by John Hays • Short annual, 10 to 50 cm tall, with stems finely wiry, spindly • Stems simple to few-branched • Leaves opposite, scale-like, about 1mm long, barely perceptible to the unaided eye • Flowers few in number, mostly born singly or in pairs from the highest node of a branchlet • Pedicels filiform, 5 to 10 mm long, subtending bracts minute • Calyx 2 mm long, lobes short-deltoid, with broad shallow sinuses between lobes • Corolla lavender-pink, without lines or spots within, 10 to 13 mm long, exterior glabrous • Capsule globe-like, nearly half exerted from calyx Flowering Time: September to November Light Requirement: Full sun to partial shade Wetland Indicator Status: FAC – similar likelihood of occurring in both wetlands and non-wetlands Habitat: Wet longleaf pine flatwoods savannahs and hillside seepage bogs. Threats: • Conversion of habitat to pine plantations (bedding, dense tree spacing, etc.) • Residential and commercial development • Fire exclusion, allowing invasion of habitat by woody species • Hydrologic alteration directly (e.g. ditching) and indirectly (fire suppression allowing higher tree density and more large-diameter trees) Beneficial Management Practices: • Thinning (during very dry periods), targeting off-site species such as loblolly and slash pines for removal • Prescribed burning, establishing a regime consisting of mostly growing season (May-June) burns Rare Plants of Louisiana LA River Basins: Pearl, Pontchartrain, Mermentau, Calcasieu, Sabine Side view of flower. Photo by John Hays References: Godfrey, R. K. and J. W. Wooten. -

Epilobium Hirsutum
Epilobium hirsutum COMMON NAME Great willowherb FAMILY Onagraceae AUTHORITY Epilobium hirsutum L. FLORA CATEGORY Vascular – Exotic STRUCTURAL CLASS Herbs - Dicotyledons other than Composites CHROMOSOME NUMBER 2n = 36 BRIEF DESCRIPTION Stout, rhizomatous herb with erect stems up to 2 m tall. All parts (except Travis Wetland Nature Heritage Park. flowers) conspicuously hairy. Leaves lanceolate to narrowly oblong, Photographer: Colin Meurk coarsely toothed light green, green to grey-green. Flowers conspicous, up to 10 mm diameter, petals dark pink to rose-purple, stigma white, exserted. DISTRIBUTION Naturalised to New Zealand. As of 2019 only known from the South Island from wetlands in North Canterbury HABITAT Wetlands margins and damp ground in wasteland and urban areas. FEATURES Foliage - abaxial leaf surface and stems - Semi-aquatic, softly-hairy, rhizomatous, much branched herb up to 2 m Travis Wetland Nature Heritage Park. Photographer: James Bailey tall. Stems erect, woody near base, densely, softly pubescent; hairs in admixtures of . non-glandular and eglandular. Leaves opposite to subopposite, 30‒60 ×10‒26 mm, light green, green to grey-green, surfaces densely, finely pubescent (hairs eglandular), lanceolate to narrowly oblong, bases clasping stem and slightly decurrent with stem, apices acute, margins coarsely to finely toothed in upper ‒½. Infloresences borne in upper third of stem, with one flower per leaf axil. Flowers erect up to 10 mm diameter; ovaries erect or outwardly falcate, 30‒50 mm long, densely strigulose, dull green-green or maroon, borne on a pedicel 4‒20 mm long. Hypanthium 1.8–2.8 × 1.6–3.2 mm, bearing a conspicuous rim of white hairs near apex. -

An Assessment of Exotic Species in the Tonle Sap Biosphere Reserve
AN ASSESSMENT OF EXOTIC SPECIES IN THE TONLE SAP BIOSPHERE RESERVE AND ASSOCIATED THREATS TO BIODIVERSITY A RESOURCE DOCUMENT FOR THE MANAGEMENT OF INVASIVE ALIEN SPECIES December 2006 Robert van Zalinge (compiler) This publication is a technical output of the UNDP/GEF-funded Tonle Sap Conservation Project Executive Summary Introduction This report is mainly a literature review. It attempts to put together all the available information from recent biological surveys, and environmental and resource use studies in the Tonle Sap Biosphere Reserve (TSBR) in order to assess the status of exotic species and report any information on their abundance, distribution and impact. For those exotic species found in the TSBR, it is examined whether they can be termed as being an invasive alien species (IAS). IAS are exotic species that pose a threat to native ecosystems, economies and/or human health. It is widely believed that IAS are the second most significant threat to biodiversity worldwide, following habitat destruction. In recognition of the threat posed by IAS the Convention on Biological Diversity puts forward the following strategy to all parties in Article 8h: “each contracting party shall as far as possible and as appropriate: prevent the introduction of, control, or eradicate those alien species which threaten ecosystems, habitats or species”. The National Assembly of Cambodia ratified the Convention on Biological Diversity in 1995. After reviewing the status of exotic species in the Tonle Sap from the literature, as well as the results from a survey based on questionnaires distributed among local communities, the main issues are discussed, possible strategies to combat the spread of alien species that are potentially invasive are examined, and recommendations are made to facilitate the implementation of a strategy towards reducing the impact of these species on the TSBR ecosystem. -

Epilobium Hirsutum L. (Onagraceae): a New Distributional Report for Northern Haryana, India
Journal on New Biological Reports ISSN 2319 – 1104 (Online) JNBR 5(3) 178 – 179 (2016) Published by www.researchtrend.net Epilobium hirsutum L. (Onagraceae): A New Distributional Report for Northern Haryana, India Inam Mohammed & Amarjit Singh* Guru Nanak Khalsa College, Yamuna Nagar,Haryana,India. *Corresponding author: [email protected] | Received: 24 November2016 | Accepted: 26 December 2016 | ABSTRACT The paper presents a taxon namely Epilobium hirsutum L. a showy member of Onagraceae and a Eurasian species collected and identified for the first time from district Yamuna nagar, Nothern Haryana. Key Words: Epilobium, Eurasian, Northern Haryana. INTRODUCTION The genus Epilobium L. with more than 300 specimens from Meerut region near Ganga canal. species occurs in all continents relatively at high Malik et al (2014) collected and identified this altitudes. Clarke (1879) described 12 species under species from district Saharanpur of Uttar Pradesh. the genus Epilobium from East and North- East During the survey of district Yamuna Nagar of Himalaya. Raven (1962) recognized 37 species, Haryana, authors reported three sites of this which include 13 new taxa from the Himalayan species, covering an estimated 325 km square area. region and recorded 31 taxa from India. In the 20th Collected specimens were critically examined with century Peter H. Raven (1976) has researched relevant literature and identified with the help of phylogeny and systematics of willow herbs DD herbarium and BSD herbarium Dehradun. extensively. Giri and Banerjee (1984) wrote Identified specimens have been deposited in identification and distributional note on a few herbarium of Guru Nanak Khalsa College, Yamuna species of Epilobium. In Western Himalayas E. -

TAXON:Fuchsia Magellanica Lam. SCORE:18.0 RATING:High Risk
TAXON: Fuchsia magellanica Lam. SCORE: 18.0 RATING: High Risk Taxon: Fuchsia magellanica Lam. Family: Onagraceae Common Name(s): earring flower Synonym(s): Fuchsia gracilis Lindl. hardy fuchsia Fuchsia macrostemma Ruiz & Pav. kulapepeiao lady's eardrops Assessor: Chuck Chimera Status: Assessor Approved End Date: 9 Jul 2021 WRA Score: 18.0 Designation: H(HPWRA) Rating: High Risk Keywords: Smothering Shrub, Environmental Weed, Self-Compatible, Spreads Vegetatively, Bird- Dispersed Qsn # Question Answer Option Answer 101 Is the species highly domesticated? y=-3, n=0 n 102 Has the species become naturalized where grown? 103 Does the species have weedy races? Species suited to tropical or subtropical climate(s) - If 201 island is primarily wet habitat, then substitute "wet (0-low; 1-intermediate; 2-high) (See Appendix 2) High tropical" for "tropical or subtropical" 202 Quality of climate match data (0-low; 1-intermediate; 2-high) (See Appendix 2) High 203 Broad climate suitability (environmental versatility) y=1, n=0 y Native or naturalized in regions with tropical or 204 y=1, n=0 y subtropical climates Does the species have a history of repeated introductions 205 y=-2, ?=-1, n=0 y outside its natural range? 301 Naturalized beyond native range y = 1*multiplier (see Appendix 2), n= question 205 y 302 Garden/amenity/disturbance weed n=0, y = 1*multiplier (see Appendix 2) n 303 Agricultural/forestry/horticultural weed n=0, y = 2*multiplier (see Appendix 2) n 304 Environmental weed n=0, y = 2*multiplier (see Appendix 2) y 305 Congeneric weed n=0, y = 1*multiplier (see Appendix 2) y 401 Produces spines, thorns or burrs y=1, n=0 n 402 Allelopathic 403 Parasitic y=1, n=0 n 404 Unpalatable to grazing animals y=1, n=-1 n 405 Toxic to animals y=1, n=0 n 406 Host for recognized pests and pathogens 407 Causes allergies or is otherwise toxic to humans y=1, n=0 n Creation Date: 9 Jul 2021 (Fuchsia magellanica Lam. -

Attracting Hummingbirds.’ Documents/Attractinghummingbirdsfs-1046April2015.Pdf
Tips on Gardening for Hummingbirds William Proebsting ([email protected]) Our breeding/resident hummingbirds in the Willamette Valley are Rufous Hummingbird and Anna’s Hummingbird. Rufous nest in almost any western habitat, but we typically find them here in brush to open forest. Anna’s has recently expanded its range to Oregon. It has a strong association with human habitation. Hummingbirds feed on nectar, tree sap and insects at sapsucker holes, insects and spiders, as well as sugar water at feeders. Our ideal hummingbird garden provides a diversity of feeding opportunities. This talk will center broadly on garden design and a number of shrubs and perennials that hummingbirds feed on. In general, think of a diversity of native plants, whether you are gardening for all birds or focused on hummingbirds. We will discuss a number of non-native plants that hummingbirds use, as well. This approach will build a general garden environment in which hummingbirds find the resources they need: 1) Develop structural diversity. Key considerations are A) to incorporate both vertical and horizontal elements in your garden design, i.e. trees and shrubs. If you have a small plot to work with, then key off nearby trees in the alley or along the property line. B) avoid large gaps between plants, by growing connectivity in three dimensions between trees to big shrubs to small shrubs to perennials and ground covers. C) use dead wood, small snags, brush piles and leaves where you can. 2) Develop biological diversity by using as many species as possible. This diversity provides an array of plant products, especially flowers and fruits over a longer period of time.