Approved Minutes for the Division Committee Meeting
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Transport of Dangerous Goods
ST/SG/AC.10/1/Rev.16 (Vol.I) Recommendations on the TRANSPORT OF DANGEROUS GOODS Model Regulations Volume I Sixteenth revised edition UNITED NATIONS New York and Geneva, 2009 NOTE The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. ST/SG/AC.10/1/Rev.16 (Vol.I) Copyright © United Nations, 2009 All rights reserved. No part of this publication may, for sales purposes, be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, electrostatic, magnetic tape, mechanical, photocopying or otherwise, without prior permission in writing from the United Nations. UNITED NATIONS Sales No. E.09.VIII.2 ISBN 978-92-1-139136-7 (complete set of two volumes) ISSN 1014-5753 Volumes I and II not to be sold separately FOREWORD The Recommendations on the Transport of Dangerous Goods are addressed to governments and to the international organizations concerned with safety in the transport of dangerous goods. The first version, prepared by the United Nations Economic and Social Council's Committee of Experts on the Transport of Dangerous Goods, was published in 1956 (ST/ECA/43-E/CN.2/170). In response to developments in technology and the changing needs of users, they have been regularly amended and updated at succeeding sessions of the Committee of Experts pursuant to Resolution 645 G (XXIII) of 26 April 1957 of the Economic and Social Council and subsequent resolutions. -

A Confinement Strategy to Prepare N-Doped Reduced Graphene Oxide
Electronic Supplementary Material (ESI) for Nanoscale. This journal is © The Royal Society of Chemistry 2019 Supporting Information A confinement strategy to prepare N-doped reduced graphene oxide foams with desired monolithic structures for supercapacitors Daoqing Liu,a,b,# Qianwei Li, *c,# Si Li, a,b Jinbao Hou,d Huazhang Zhao *a,b a Department of Environmental Engineering, Peking University, Beijing 100871, China b The Key Laboratory of Water and Sediment Sciences, Ministry of Education, Beijing 100871, China c State Key Laboratory of Heavy Oil Processing, Beijing Key Laboratory of Oil and Gas Pollution Control, China University of Petroleum, 18 Fuxue Road, Changping District, Beijing 102249, China d College of Chemical Engineering, Beijing University of Chemical Technology, Beijing 100029, China # Co-first author. * Corresponding author. Tel/fax: +86-10-62758748; email address: [email protected] (H.Z. Zhao). S1 Experimental Methods 1. Preparation of graphite oxide (GO) Graphite oxide was prepared from natural graphite (Jinglong Co., Beijing, China) according to 1 a modified Hummers method : 120 mL 98 wt% H2SO4 was poured into a beaker containing a mixture of 5 g natural graphite and 2.5 g NaNO3, and then the mixture was stirred in an ice bath for 30 min. 15 g KMnO4 was added slowly into the mixture, which was allowed to react for 2 h at a temperature no more than 20°C. Then, the temperature was risen to 35°C, and the reaction was performed for another 2 h. After that, the reactant mixture was poured slowly into 360 mL distilled water under violent stirring condition so as to control the temperature no more than 90°C, followed by further reaction at 75°C for 1 h. -

Sections-A, B and C
Science IX Sample Paper 7 Solved www.rava.org.in CLASS IX (2019-20) SCIENCE (CODE 086) SAMPLE PAPER-7 Time : 3 Hours Maximum Marks : 80 General Instructions : (i) The question paper comprises of three sections-A, B and C. Attempt all the sections. (ii) All questions are compulsory. (iii) Internal choice is given in each sections. (iv) All questions in Section A are one-mark questions comprising MCQ, VSA type and assertion-reason type questions. They are to be answered in one word or in one sentence. (v) All questions in Section B are three-mark, short-answer type questions. These are to be answered in about 50-60 words each. (vi) All questions in Section C are five-mark, long-answer type questions. These are to be answered in about 80-90 words each. (vii) This question paper consists of a total of 30 questions. 6. Who proposed the fluid mosaic model of protoplasm? [1] Section - A (a) Singer and Nicolson (b) Watson and Crick (c) Robert Hook (d) Robert Brown 1. Which of the following actions a force can do? [1] (a) Can move a stationary object. Ans : (a) Singer and Nicolson. (b) Can stop a moving object. or (c) Can change the speed of a moving object. Which of the following are complex tissues? (d) All of the above. (a) Xylem and Phloem Ans : (d) All of the above (b) Collenchyma and Sclerenchyma (c) Parenchyma and Collenchyma 2. Ozone layer protects us from which one of the (d) Xylem and Parenchyma following? [1] (a) X- rays. -

Chapter 2: Alkanes Alkanes from Carbon and Hydrogen
Chapter 2: Alkanes Alkanes from Carbon and Hydrogen •Alkanes are carbon compounds that contain only single bonds. •The simplest alkanes are hydrocarbons – compounds that contain only carbon and hydrogen. •Hydrocarbons are used mainly as fuels, solvents and lubricants: H H H H H H H H H H H H C H C C H C C C C H H C C C C C H H H C C H H H H H H CH2 H CH3 H H H H CH3 # of carbons boiling point range Use 1-4 <20 °C fuel (gasses such as methane, propane, butane) 5-6 30-60 solvents (petroleum ether) 6-7 60-90 solvents (ligroin) 6-12 85-200 fuel (gasoline) 12-15 200-300 fuel (kerosene) 15-18 300-400 fuel (heating oil) 16-24 >400 lubricating oil, asphalt Hydrocarbons Formula Prefix Suffix Name Structure H CH4 meth- -ane methane H C H H C H eth- -ane ethane 2 6 H3C CH3 C3H8 prop- -ane propane C4H10 but- -ane butane C5H12 pent- -ane pentane C6H14 hex- -ane hexane C7H16 hept- -ane heptane C8H18 oct- -ane octane C9H20 non- -ane nonane C10H22 dec- -ane decane Hydrocarbons Formula Prefix Suffix Name Structure H CH4 meth- -ane methane H C H H H H C2H6 eth- -ane ethane H C C H H H H C H prop- -ane propane 3 8 H3C C CH3 or H H H C H 4 10 but- -ane butane H3C C C CH3 or H H H C H 4 10 but- -ane butane? H3C C CH3 or CH3 HydHrydorcocaarrbobnos ns Formula Prefix Suffix Name Structure H CH4 meth- -ane methane H C H H H H C2H6 eth- -ane ethane H C C H H H H C3H8 prop- -ane propane H3C C CH3 or H H H C H 4 10 but- -ane butane H3C C C CH3 or H H H C H 4 10 but- -ane iso-butane H3C C CH3 or CH3 HydHrydoroccarbrobnsons Formula Prefix Suffix Name Structure H H -
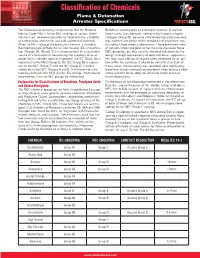
Classification of Chemicals
Classification of Chemicals Flame & Detonation Arrester Specifications PROTECTOSEAL ® The Protectoseal Company recommends that the National Butadiene would qualify as a Group D material. In each of Electric Code (NEC) Article 500, rankings of various chemi - these cases, the chemicals were primarly listed in a higher cals be used, whenever possible, to determine the suitability category (Group B), because of relatively high pressure read - of a detonation arrester for use with a particular chemical. ings noted in one phase of the standard test procedure con - When no NEC rating of the particular chemical is available, ducted by Underwriters Laboratories. These pressures were the International Electrotechnical Commission (IEC) classifica - of concern when categorizing the chemicals because these tion (Groups IIA, IIB and IIC) is recommended as a secondary NEC groupings are also used as standard indicators for the source of information for determining the suitability of an ar - design strength requirements of electrical boxes, apparatus, rester for its intended service. In general, the IEC Group IIA is etc. that must withstand the pressures generated by an igni - equivalent to the NEC Group D; the IEC Group IIB is equiva - tion within the container. It should be noted that, in each of lent to the NEC Group C; and the IEC Group IIC includes these cases, the test pressures recorded were significantly chemicals in the NEC Groups A and B. In the event of a dis - lower than those commonly encountered when testing a deto - crepancy between the NEC and the IEC ratings, Protectoseal nation arrester for its ability to withstand stable and over - recommends that the NEC groups be referenced. -

Measurements of Higher Alkanes Using NO Chemical Ionization in PTR-Tof-MS
Atmos. Chem. Phys., 20, 14123–14138, 2020 https://doi.org/10.5194/acp-20-14123-2020 © Author(s) 2020. This work is distributed under the Creative Commons Attribution 4.0 License. Measurements of higher alkanes using NOC chemical ionization in PTR-ToF-MS: important contributions of higher alkanes to secondary organic aerosols in China Chaomin Wang1,2, Bin Yuan1,2, Caihong Wu1,2, Sihang Wang1,2, Jipeng Qi1,2, Baolin Wang3, Zelong Wang1,2, Weiwei Hu4, Wei Chen4, Chenshuo Ye5, Wenjie Wang5, Yele Sun6, Chen Wang3, Shan Huang1,2, Wei Song4, Xinming Wang4, Suxia Yang1,2, Shenyang Zhang1,2, Wanyun Xu7, Nan Ma1,2, Zhanyi Zhang1,2, Bin Jiang1,2, Hang Su8, Yafang Cheng8, Xuemei Wang1,2, and Min Shao1,2 1Institute for Environmental and Climate Research, Jinan University, 511443 Guangzhou, China 2Guangdong-Hongkong-Macau Joint Laboratory of Collaborative Innovation for Environmental Quality, 511443 Guangzhou, China 3School of Environmental Science and Engineering, Qilu University of Technology (Shandong Academy of Sciences), 250353 Jinan, China 4State Key Laboratory of Organic Geochemistry and Guangdong Key Laboratory of Environmental Protection and Resources Utilization, Guangzhou Institute of Geochemistry, Chinese Academy of Sciences, 510640 Guangzhou, China 5State Joint Key Laboratory of Environmental Simulation and Pollution Control, College of Environmental Sciences and Engineering, Peking University, 100871 Beijing, China 6State Key Laboratory of Atmospheric Boundary Physics and Atmospheric Chemistry, Institute of Atmospheric Physics, Chinese -

Cycloalkanes, Cycloalkenes, and Cycloalkynes
CYCLOALKANES, CYCLOALKENES, AND CYCLOALKYNES any important hydrocarbons, known as cycloalkanes, contain rings of carbon atoms linked together by single bonds. The simple cycloalkanes of formula (CH,), make up a particularly important homologous series in which the chemical properties change in a much more dramatic way with increasing n than do those of the acyclic hydrocarbons CH,(CH,),,-,H. The cyclo- alkanes with small rings (n = 3-6) are of special interest in exhibiting chemical properties intermediate between those of alkanes and alkenes. In this chapter we will show how this behavior can be explained in terms of angle strain and steric hindrance, concepts that have been introduced previously and will be used with increasing frequency as we proceed further. We also discuss the conformations of cycloalkanes, especially cyclo- hexane, in detail because of their importance to the chemistry of many kinds of naturally occurring organic compounds. Some attention also will be paid to polycyclic compounds, substances with more than one ring, and to cyclo- alkenes and cycloalkynes. 12-1 NOMENCLATURE AND PHYSICAL PROPERTIES OF CYCLOALKANES The IUPAC system for naming cycloalkanes and cycloalkenes was presented in some detail in Sections 3-2 and 3-3, and you may wish to review that ma- terial before proceeding further. Additional procedures are required for naming 446 12 Cycloalkanes, Cycloalkenes, and Cycloalkynes Table 12-1 Physical Properties of Alkanes and Cycloalkanes Density, Compounds Bp, "C Mp, "C diO,g ml-' propane cyclopropane butane cyclobutane pentane cyclopentane hexane cyclohexane heptane cycloheptane octane cyclooctane nonane cyclononane "At -40". bUnder pressure. polycyclic compounds, which have rings with common carbons, and these will be discussed later in this chapter. -
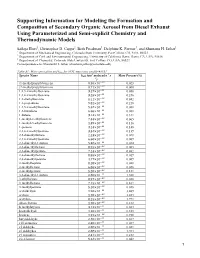
Supporting Information for Modeling the Formation and Composition Of
Supporting Information for Modeling the Formation and Composition of Secondary Organic Aerosol from Diesel Exhaust Using Parameterized and Semi-explicit Chemistry and Thermodynamic Models Sailaja Eluri1, Christopher D. Cappa2, Beth Friedman3, Delphine K. Farmer3, and Shantanu H. Jathar1 1 Department of Mechanical Engineering, Colorado State University, Fort Collins, CO, USA, 80523 2 Department of Civil and Environmental Engineering, University of California Davis, Davis, CA, USA, 95616 3 Department of Chemistry, Colorado State University, Fort Collins, CO, USA, 80523 Correspondence to: Shantanu H. Jathar ([email protected]) Table S1: Mass speciation and kOH for VOC emissions profile #3161 3 -1 - Species Name kOH (cm molecules s Mass Percent (%) 1) (1-methylpropyl) benzene 8.50×10'() 0.023 (2-methylpropyl) benzene 8.71×10'() 0.060 1,2,3-trimethylbenzene 3.27×10'(( 0.056 1,2,4-trimethylbenzene 3.25×10'(( 0.246 1,2-diethylbenzene 8.11×10'() 0.042 1,2-propadiene 9.82×10'() 0.218 1,3,5-trimethylbenzene 5.67×10'(( 0.088 1,3-butadiene 6.66×10'(( 0.088 1-butene 3.14×10'(( 0.311 1-methyl-2-ethylbenzene 7.44×10'() 0.065 1-methyl-3-ethylbenzene 1.39×10'(( 0.116 1-pentene 3.14×10'(( 0.148 2,2,4-trimethylpentane 3.34×10'() 0.139 2,2-dimethylbutane 2.23×10'() 0.028 2,3,4-trimethylpentane 6.60×10'() 0.009 2,3-dimethyl-1-butene 5.38×10'(( 0.014 2,3-dimethylhexane 8.55×10'() 0.005 2,3-dimethylpentane 7.14×10'() 0.032 2,4-dimethylhexane 8.55×10'() 0.019 2,4-dimethylpentane 4.77×10'() 0.009 2-methylheptane 8.28×10'() 0.028 2-methylhexane 6.86×10'() -

Process for the Preparation of (11Α,13E,15S)-11,15-Dihydroxy-9-Oxoprost-13-En-1-Oic Acid
Technical Disclosure Commons Defensive Publications Series July 2020 Process for the preparation of (11α,13E,15S)-11,15-dihydroxy-9-oxoprost-13-en-1-oic acid Srinivasan Thirumalai Rajan Follow this and additional works at: https://www.tdcommons.org/dpubs_series Recommended Citation Srinivasan Thirumalai Rajan, "Process for the preparation of (11α,13E,15S)-11,15-dihydroxy-9-oxoprost-13-en-1-oic acid", Technical Disclosure Commons, (July 10, 2020) https://www.tdcommons.org/dpubs_series/3418 This work is licensed under a Creative Commons Attribution 4.0 License. This Article is brought to you for free and open access by Technical Disclosure Commons. It has been accepted for inclusion in Defensive Publications Series by an authorized administrator of Technical Disclosure Commons. : Process for the preparation of (11?,13E,15S)-11,15-dihydroxy-9-ox Process for the preparation of (11α,13E,15S)-11,15-dihydroxy-9-oxoprost-13-en- 1-oic acid Field of the invention: 5 The present application relates a process for the preparation of (11α,13E,15S)- 11,15-dihydroxy-9-oxoprost-13-en-1-oic acid. Formula-1 10 Background of the invention: (11α,13E,15S)-11,15-dihydroxy-9-oxoprost-13-en-1-oic acid is generally known as Prostaglandin E1 (PGE1) or Alprostadil. Alprostadil was approved in US and Europe under the brand name of CAVERJECT® and indicated for the treatment 15 of erectile dysfunction due to neurogenic, vasculogenic, psychogenic, or mixed etiology. Biosynthetic PGE1, is formed from dihomo-γ-linolenic acid was disclosed in Prostaglandin 20, 187, 1980. Syntheses of PGE1 was disclosed in J. -

Vapor Pressures and Vaporization Enthalpies of the N-Alkanes from 2 C21 to C30 at T ) 298.15 K by Correlation Gas Chromatography
BATCH: je1a04 USER: jeh69 DIV: @xyv04/data1/CLS_pj/GRP_je/JOB_i01/DIV_je0301747 DATE: October 17, 2003 1 Vapor Pressures and Vaporization Enthalpies of the n-Alkanes from 2 C21 to C30 at T ) 298.15 K by Correlation Gas Chromatography 3 James S. Chickos* and William Hanshaw 4 Department of Chemistry and Biochemistry, University of MissourisSt. Louis, St. Louis, Missouri 63121 5 6 The temperature dependence of gas chromatographic retention times for n-heptadecane to n-triacontane 7 is reported. These data are used to evaluate the vaporization enthalpies of these compounds at T ) 298.15 8 K, and a protocol is described that provides vapor pressures of these n-alkanes from T ) 298.15 to 575 9 K. The vapor pressure and vaporization enthalpy results obtained are compared with existing literature 10 data where possible and found to be internally consistent. Sublimation enthalpies for n-C17 to n-C30 are 11 calculated by combining vaporization enthalpies with fusion enthalpies and are compared when possible 12 to direct measurements. 13 14 Introduction 15 The n-alkanes serve as excellent standards for the 16 measurement of vaporization enthalpies of hydrocarbons.1,2 17 Recently, the vaporization enthalpies of the n-alkanes 18 reported in the literature were examined and experimental 19 values were selected on the basis of how well their 20 vaporization enthalpies correlated with their enthalpies of 21 transfer from solution to the gas phase as measured by gas 22 chromatography.3 A plot of the vaporization enthalpies of 23 the n-alkanes as a function of the number of carbon atoms 24 is given in Figure 1. -
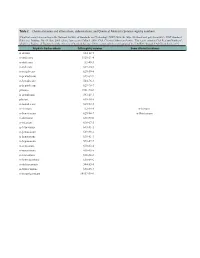
Table 2. Chemical Names and Alternatives, Abbreviations, and Chemical Abstracts Service Registry Numbers
Table 2. Chemical names and alternatives, abbreviations, and Chemical Abstracts Service registry numbers. [Final list compiled according to the National Institute of Standards and Technology (NIST) Web site (http://webbook.nist.gov/chemistry/); NIST Standard Reference Database No. 69, June 2005 release, last accessed May 9, 2008. CAS, Chemical Abstracts Service. This report contains CAS Registry Numbers®, which is a Registered Trademark of the American Chemical Society. CAS recommends the verification of the CASRNs through CAS Client ServicesSM] Aliphatic hydrocarbons CAS registry number Some alternative names n-decane 124-18-5 n-undecane 1120-21-4 n-dodecane 112-40-3 n-tridecane 629-50-5 n-tetradecane 629-59-4 n-pentadecane 629-62-9 n-hexadecane 544-76-3 n-heptadecane 629-78-7 pristane 1921-70-6 n-octadecane 593-45-3 phytane 638-36-8 n-nonadecane 629-92-5 n-eicosane 112-95-8 n-Icosane n-heneicosane 629-94-7 n-Henicosane n-docosane 629-97-0 n-tricosane 638-67-5 n-tetracosane 643-31-1 n-pentacosane 629-99-2 n-hexacosane 630-01-3 n-heptacosane 593-49-7 n-octacosane 630-02-4 n-nonacosane 630-03-5 n-triacontane 638-68-6 n-hentriacontane 630-04-6 n-dotriacontane 544-85-4 n-tritriacontane 630-05-7 n-tetratriacontane 14167-59-0 Table 2. Chemical names and alternatives, abbreviations, and Chemical Abstracts Service registry numbers.—Continued [Final list compiled according to the National Institute of Standards and Technology (NIST) Web site (http://webbook.nist.gov/chemistry/); NIST Standard Reference Database No. -

Chemical Communications COMMUNICATION
Please do not adjust margins Chemical Communications COMMUNICATION Multielectron C–H Photoactivation with an Sb(V) Oxo Corrole Christopher M. Lemon,† Andrew G. Maher, Anthony R. Mazzotti, David C. Powers,‡ Miguel I. Received 00th January 20xx, Gonzalez, and Daniel G. Nocera* Accepted 00th January 20xx DOI: 10.1039/x0xx00000x www.rsc.org/ Pnictogen complexes are ideal for mediating multi-electron chemical reactions in two-electron steps. We report a Sb(V) bis-μ- oxo corrole that photochemically oxidises the C–H bonds of organic substrates. In the case of toluene, the substrate is oxidised to benzaldehyde, a rare example of a four-electron photoreaction. Nature utilizes the cytochrome P450 family of enzymes to perform a variety of aerobic oxidations including hydrocarbon 1 hydroxylation, alkene epoxidation, and N- and S-oxidation. Figure 1. Synthesis of the Sb(V) oxo dimer 2 (Ar = C6F5), which can be prepared using Antimony porphyrins have garnered interest as cytochrome various oxidants: H2O2 • urea (74% yield), PhI(OAc)2/O2 (56% yield), or PhIO (71% yield). P450 mimics,2–4 owing to similarities between the absorption5 and magnetic circular dichroism6 spectra of Sb(III) porphyrins As an alternative to manipulating the Sb–X bond by outer- and the CO adduct of cytochrome P450. Augmenting these sphere electron transfer, we have developed a direct halogen spectroscopic relationships, antimony porphyrins mediate a multielectron photochemistry by exploiting the Sb(III)/Sb(V)X2 23 variety of photochemical oxidations7,8 including alkene couple. This approach complements M–X photoactivation at n+2 n… n+2 epoxidation9,10 and hydrocarbon hydroxylation.11,12 These the M X2 centre of two-electron mixed-valence (M M ) 24–27 reactions demonstrate that main group complexes can serve complexes with second- and third-row transition metals.