Supplementary Materials
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Transcriptome Response of High- and Low-Light-Adapted Prochlorococcus Strains to Changing Iron Availability
Transcriptome response of high- and low-light-adapted Prochlorococcus strains to changing iron availability The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation Thompson, Anne W et al. “Transcriptome Response of High- and Low-light-adapted Prochlorococcus Strains to Changing Iron Availability.” ISME Journal (2011), 1-15. As Published http://dx.doi.org/10.1038/ismej.2011.49 Publisher Nature Publishing Group Version Author's final manuscript Citable link http://hdl.handle.net/1721.1/64705 Terms of Use Creative Commons Attribution-Noncommercial-Share Alike 3.0 Detailed Terms http://creativecommons.org/licenses/by-nc-sa/3.0/ Title: Transcriptome response of high- and low-light adapted Prochlorococcus strains to changing iron availability Running title: Prochlorococcus response to iron stress 5 Contributors: Anne W. Thompson1, Katherine Huang1, Mak A. Saito* 2, Sallie W. Chisholm* 1, 3 10 1 MIT Department of Civil and Environmental Engineering 2 Woods Hole Oceanographic Institution – Department of Marine Chemistry and Geochemistry 3 MIT Department of Biology 15 * To whom correspondence should be addressed: E-mail: [email protected] and [email protected] Subject Category: Microbial population and community ecology 20 Abstract Prochlorococcus contributes significantly to ocean primary productivity. The link between primary productivity and iron in specific ocean regions is well established and iron-limitation of Prochlorococcus cell division rates in these regions has been 25 demonstrated. However, the extent of ecotypic variation in iron metabolism among Prochlorococcus and the molecular basis for differences is not understood. Here, we examine the growth and transcriptional response of Prochlorococcus strains, MED4 and MIT9313, to changing iron concentrations. -

A Minimum-Labeling Approach for Reconstructing Protein Networks Across Multiple Conditions
A Minimum-Labeling Approach for Reconstructing Protein Networks across Multiple Conditions Arnon Mazza1, Irit Gat-Viks2, Hesso Farhan3, and Roded Sharan1 1 Blavatnik School of Computer Science, Tel Aviv University, Tel Aviv 69978, Israel. Email: [email protected]. 2 Dept. of Cell Research and Immunology, Tel Aviv University, Tel Aviv 69978, Israel. 3 Biotechnology Institute Thurgau, University of Konstanz, Unterseestrasse 47, CH-8280 Kreuzlingen, Switzerland. Abstract. The sheer amounts of biological data that are generated in recent years have driven the development of network analysis tools to fa- cilitate the interpretation and representation of these data. A fundamen- tal challenge in this domain is the reconstruction of a protein-protein sub- network that underlies a process of interest from a genome-wide screen of associated genes. Despite intense work in this area, current algorith- mic approaches are largely limited to analyzing a single screen and are, thus, unable to account for information on condition-specific genes, or reveal the dynamics (over time or condition) of the process in question. Here we propose a novel formulation for network reconstruction from multiple-condition data and devise an efficient integer program solution for it. We apply our algorithm to analyze the response to influenza in- fection in humans over time as well as to analyze a pair of ER export related screens in humans. By comparing to an extant, single-condition tool we demonstrate the power of our new approach in integrating data from multiple conditions in a compact and coherent manner, capturing the dynamics of the underlying processes. 1 Introduction With the increasing availability of high-throughput data, network biol- arXiv:1307.7803v1 [q-bio.QM] 30 Jul 2013 ogy has become the method of choice for filtering, interpreting and rep- resenting these data. -

Conserved and Novel Properties of Clathrin-Mediated Endocytosis in Dictyostelium Discoideum" (2012)
Rockefeller University Digital Commons @ RU Student Theses and Dissertations 2012 Conserved and Novel Properties of Clathrin- Mediated Endocytosis in Dictyostelium Discoideum Laura Macro Follow this and additional works at: http://digitalcommons.rockefeller.edu/ student_theses_and_dissertations Part of the Life Sciences Commons Recommended Citation Macro, Laura, "Conserved and Novel Properties of Clathrin-Mediated Endocytosis in Dictyostelium Discoideum" (2012). Student Theses and Dissertations. Paper 163. This Thesis is brought to you for free and open access by Digital Commons @ RU. It has been accepted for inclusion in Student Theses and Dissertations by an authorized administrator of Digital Commons @ RU. For more information, please contact [email protected]. CONSERVED AND NOVEL PROPERTIES OF CLATHRIN- MEDIATED ENDOCYTOSIS IN DICTYOSTELIUM DISCOIDEUM A Thesis Presented to the Faculty of The Rockefeller University in Partial Fulfillment of the Requirements for the degree of Doctor of Philosophy by Laura Macro June 2012 © Copyright by Laura Macro 2012 CONSERVED AND NOVEL PROPERTIES OF CLATHRIN- MEDIATED ENDOCYTOSIS IN DICTYOSTELIUM DISCOIDEUM Laura Macro, Ph.D. The Rockefeller University 2012 The protein clathrin mediates one of the major pathways of endocytosis from the extracellular milieu and plasma membrane. Clathrin functions with a network of interacting accessory proteins, one of which is the adaptor complex AP-2, to co-ordinate vesicle formation. Disruption of genes involved in clathrin-mediated endocytosis causes embryonic lethality in multicellular animals suggesting that clathrin-mediated endocytosis is a fundamental cellular process. However, loss of clathrin-mediated endocytosis genes in single cell eukaryotes, such as S.cerevisiae (yeast), does not cause lethality, suggesting that clathrin may convey specific advantages for multicellularity. -

Supplementary Methods
Supplementary methods Human lung tissues and tissue microarray (TMA) All human tissues were obtained from the Lung Cancer Specialized Program of Research Excellence (SPORE) Tissue Bank at the M.D. Anderson Cancer Center (Houston, TX). A collection of 26 lung adenocarcinomas and 24 non-tumoral paired tissues were snap-frozen and preserved in liquid nitrogen for total RNA extraction. For each tissue sample, the percentage of malignant tissue was calculated and the cellular composition of specimens was determined by histological examination (I.I.W.) following Hematoxylin-Eosin (H&E) staining. All malignant samples retained contained more than 50% tumor cells. Specimens resected from NSCLC stages I-IV patients who had no prior chemotherapy or radiotherapy were used for TMA analysis by immunohistochemistry. Patients who had smoked at least 100 cigarettes in their lifetime were defined as smokers. Samples were fixed in formalin, embedded in paraffin, stained with H&E, and reviewed by an experienced pathologist (I.I.W.). The 413 tissue specimens collected from 283 patients included 62 normal bronchial epithelia, 61 bronchial hyperplasias (Hyp), 15 squamous metaplasias (SqM), 9 squamous dysplasias (Dys), 26 carcinomas in situ (CIS), as well as 98 squamous cell carcinomas (SCC) and 141 adenocarcinomas. Normal bronchial epithelia, hyperplasia, squamous metaplasia, dysplasia, CIS, and SCC were considered to represent different steps in the development of SCCs. All tumors and lesions were classified according to the World Health Organization (WHO) 2004 criteria. The TMAs were prepared with a manual tissue arrayer (Advanced Tissue Arrayer ATA100, Chemicon International, Temecula, CA) using 1-mm-diameter cores in triplicate for tumors and 1.5 to 2-mm cores for normal epithelial and premalignant lesions. -
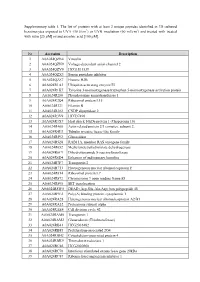
Supplementary Table 1. the List of Proteins with at Least 2 Unique
Supplementary table 1. The list of proteins with at least 2 unique peptides identified in 3D cultured keratinocytes exposed to UVA (30 J/cm2) or UVB irradiation (60 mJ/cm2) and treated with treated with rutin [25 µM] or/and ascorbic acid [100 µM]. Nr Accession Description 1 A0A024QZN4 Vinculin 2 A0A024QZN9 Voltage-dependent anion channel 2 3 A0A024QZV0 HCG1811539 4 A0A024QZX3 Serpin peptidase inhibitor 5 A0A024QZZ7 Histone H2B 6 A0A024R1A3 Ubiquitin-activating enzyme E1 7 A0A024R1K7 Tyrosine 3-monooxygenase/tryptophan 5-monooxygenase activation protein 8 A0A024R280 Phosphoserine aminotransferase 1 9 A0A024R2Q4 Ribosomal protein L15 10 A0A024R321 Filamin B 11 A0A024R382 CNDP dipeptidase 2 12 A0A024R3V9 HCG37498 13 A0A024R3X7 Heat shock 10kDa protein 1 (Chaperonin 10) 14 A0A024R408 Actin related protein 2/3 complex, subunit 2, 15 A0A024R4U3 Tubulin tyrosine ligase-like family 16 A0A024R592 Glucosidase 17 A0A024R5Z8 RAB11A, member RAS oncogene family 18 A0A024R652 Methylenetetrahydrofolate dehydrogenase 19 A0A024R6C9 Dihydrolipoamide S-succinyltransferase 20 A0A024R6D4 Enhancer of rudimentary homolog 21 A0A024R7F7 Transportin 2 22 A0A024R7T3 Heterogeneous nuclear ribonucleoprotein F 23 A0A024R814 Ribosomal protein L7 24 A0A024R872 Chromosome 9 open reading frame 88 25 A0A024R895 SET translocation 26 A0A024R8W0 DEAD (Asp-Glu-Ala-Asp) box polypeptide 48 27 A0A024R9E2 Poly(A) binding protein, cytoplasmic 1 28 A0A024RA28 Heterogeneous nuclear ribonucleoprotein A2/B1 29 A0A024RA52 Proteasome subunit alpha 30 A0A024RAE4 Cell division cycle 42 31 -

Host Cell Factors Necessary for Influenza a Infection: Meta-Analysis of Genome Wide Studies
Host Cell Factors Necessary for Influenza A Infection: Meta-Analysis of Genome Wide Studies Juliana S. Capitanio and Richard W. Wozniak Department of Cell Biology, Faculty of Medicine and Dentistry, University of Alberta Abstract: The Influenza A virus belongs to the Orthomyxoviridae family. Influenza virus infection occurs yearly in all countries of the world. It usually kills between 250,000 and 500,000 people and causes severe illness in millions more. Over the last century alone we have seen 3 global influenza pandemics. The great human and financial cost of this disease has made it the second most studied virus today, behind HIV. Recently, several genome-wide RNA interference studies have focused on identifying host molecules that participate in Influen- za infection. We used nine of these studies for this meta-analysis. Even though the overlap among genes identified in multiple screens was small, network analysis indicates that similar protein complexes and biological functions of the host were present. As a result, several host gene complexes important for the Influenza virus life cycle were identified. The biological function and the relevance of each identified protein complex in the Influenza virus life cycle is further detailed in this paper. Background and PA bound to the viral genome via nucleoprotein (NP). The viral core is enveloped by a lipid membrane derived from Influenza virus the host cell. The viral protein M1 underlies the membrane and anchors NEP/NS2. Hemagglutinin (HA), neuraminidase Viruses are the simplest life form on earth. They parasite host (NA), and M2 proteins are inserted into the envelope, facing organisms and subvert the host cellular machinery for differ- the viral exterior. -

Produktinformation
Produktinformation Diagnostik & molekulare Diagnostik Laborgeräte & Service Zellkultur & Verbrauchsmaterial Forschungsprodukte & Biochemikalien Weitere Information auf den folgenden Seiten! See the following pages for more information! Lieferung & Zahlungsart Lieferung: frei Haus Bestellung auf Rechnung SZABO-SCANDIC Lieferung: € 10,- HandelsgmbH & Co KG Erstbestellung Vorauskassa Quellenstraße 110, A-1100 Wien T. +43(0)1 489 3961-0 Zuschläge F. +43(0)1 489 3961-7 [email protected] • Mindermengenzuschlag www.szabo-scandic.com • Trockeneiszuschlag • Gefahrgutzuschlag linkedin.com/company/szaboscandic • Expressversand facebook.com/szaboscandic SAN TA C RUZ BI OTEC HNOL OG Y, INC . COPG siRNA (h): sc-37254 BACKGROUND STORAGE AND RESUSPENSION Membrane and vesicular trafficking in the early secretory pathway are medi - Store lyophilized siRNA duplex at -20° C with desiccant. Stable for at least ated by non-clathrin COP (coat protein) I-coated vesicles. COPI-coated vesi cles one year from the date of shipment. Once resuspended, store at -20° C, mediate retrograde transport from the Golgi back to the ER and intra-Golgi avoid contact with RNAses and repeated freeze thaw cycles. transport. The cytosolic precursor of the COPI coat, the heptameric coatomer Resuspend lyophilized siRNA duplex in 330 µl of the RNAse-free water complex, is composed of two subcomplexes. The first consists of the COPB, pro vided. Resuspension of the siRNA duplex in 330 µl of RNAse-free water COPG, COPD and COPZ subunits (also known as -, -, - and -COP, respec - β γ δ ζ makes a 10 µM solution in a 10 µM Tris-HCl, pH 8.0, 20 mM NaCl, 1 mM tively), which are distantly homologous to AP Clathrin adaptor subunits. -

Supplemental Data 5-21-18
Supplemental Methods and Data Androgen receptor polyglutamine expansion drives age-dependent quality control defects and muscle dysfunction Samir R. Nath1,2,3, Zhigang Yu1, Theresa A. Gipson4, Gregory B. Marsh5, Eriko Yoshidome1, Diane M. Robins6, Sokol V. Todi5, David E. Housman4, Andrew P. Lieberman1 1 Department of Pathology, University of Michigan Medical School, Ann Arbor, MI 48109 2 Medical Scientist Training Program, University of Michigan Medical School, Ann Arbor, MI 48109 3 Cellular and Molecular Biology Graduate Program, University of Michigan Medical School, Ann Arbor, MI 48109 4 Koch Institute for Integrative Cancer Research, Massachusetts Institute of Technology, Cambridge, MA 02139 5 Department of Pharmacology, Wayne State University School of Medicine, Detroit, MI 48201 6 Department of Human Genetics, University of Michigan Medical School, Ann Arbor, MI 48109 Supplemental Methods qPCR For Drosophila samples, total RNA was extracted from adult fly heads using TRIzol. Fifteen heads were used per sample. Extracted RNA was treated with TURBO DNAse (Ambion) to eliminate contaminating DNA, and reverse transcription was carried out as indicated above. RNA levels were quantified using StepOnePlus Real-Time PCR System with Fast SYBR Green Master Mix (Applied Biosystems). rp49 was used as the internal control. Each round of qRT-PCR was conducted in technical triplicates. A total of three independent repeats was conducted. Fly histology: For histological preparation (75, 76), wings and proboscises of adult flies were removed and bodies were fixed overnight in 2% glutaraldehyde/2% paraformaldehyde in Tris- buffered saline with 0.1% Triton X-100, rotating at 4˚C. Fixed bodies were subsequently dehydrated by using a series of 30%, 50%, 75%, and 100% ethanol/propylene oxide. -

COPG Antibody (C-Term) Affinity Purified Rabbit Polyclonal Antibody (Pab) Catalog # Ap19908b
苏州工业园区双圩路9号1幢 邮 编 : 215000 电 话 : 0512-88856768 COPG Antibody (C-term) Affinity Purified Rabbit Polyclonal Antibody (Pab) Catalog # AP19908b Specification COPG Antibody (C-term) - Product info Application WB Primary Accession Q9Y678 Other Accession NP_057212.1 Reactivity Human Host Rabbit Clonality Polyclonal Isotype Rabbit Ig Clone Names RB41787 Calculated MW 97718 COPG Antibody (C-term) - Additional info Gene ID 22820 Other Names Coatomer subunit gamma-1, Gamma-1-coat protein, Gamma-1-COP, COPG1, COPG All lanes : Anti-COPG Antibody (C-term) Target/Specificity at 1:1000 dilution Lane 1: A2058 whole This COPG antibody is generated from rabbits immunized with cell lysate Lane 2: K562 whole cell lysate a KLH conjugated synthetic peptide between 721-750 amino Lysates/proteins at 20 µg per lane. acids from the C-terminal region of human COPG. Secondary Goat Anti-Rabbit IgG, (H+L), Peroxidase conjugated at 1/10000 Dilution dilution. Predicted band size : 98 kDa WB~~1:1000 Blocking/Dilution buffer: 5% NFDM/TBST. Format Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. Storage Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. Precautions COPG Antibody (C-term) is for research use only and not for use in diagnostic or therapeutic procedures. COPG Antibody (C-term) - Protein Information Name COPG1 Synonyms COPG Function The coatomer is a cytosolic protein complex that binds to dilysine motifs and reversibly associates with Golgi non-clathrin- coated vesicles, which further mediate biosynthetic protein transport from the ER, via the Golgi up to the trans Golgi network. -

Colon Cancer and Its Molecular Subsystems: Network Approaches to Dissecting Driver Gene Biology
COLON CANCER AND ITS MOLECULAR SUBSYSTEMS: NETWORK APPROACHES TO DISSECTING DRIVER GENE BIOLOGY by VISHAL N. PATEL Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy Department of Genetics CASE WESTERN RESERVE UNIVERSITY August 2011 Case Western Reserve University School of Graduate Studies We hereby approve the dissertation* of Vishal N. Patel, candidate for the degree of Doctor of Philosophy on July 6, 2011. Committee Chair: Georgia Wiesner Mark R. Chance Sudha Iyengar Mehmet Koyuturk * We also certify that written approval has been obtained for any proprietary material contained therein 2 Table of Contents I. List of Tables 6 II. List of Figures 7 III. List of Abbreviations 8 IV. Glossary 10 V. Abstract 14 VI. Colon Cancer and its Molecular Subsystems 15 Construction, Interpretation, and Validation a. Colon Cancer i. Etiology ii. Development iii. The Pathway Paradigm iv. Cancer Subtypes and Therapies b. Molecular Subsystems i. Introduction ii. Construction iii. Interpretation iv. Validation c. Summary VII. Prostaglandin dehydrogenase signaling 32 One driver and an unknown path a. Introduction i. Colon Cancer ii. Mass spectrometry 3 b. Methods c. Results d. Discussion e. Summary VIII. Apc signaling with Candidate Drivers 71 Two drivers, many paths a. Introduction b. Methods c. Results and Discussion d. Summary IX. Apc-Cdkn1a signaling 87 Two drivers, one path a. Introduction b. Methods c. Results i. Driver Gene Network Prediction ii. Single Node Perturbations 1. mRNA profiling 2. Proteomic profiling d. Discussion e. Summary X. Molecular Subsystems in Cancer 114 The Present to the Future XI. Appendix I 119 Prostaglandin dehydrogenase associated data XII. -

Dopamine Receptor D3 Regulates Endocytic Sorting by a Prazosin-Sensitive Interaction with the Coatomer COPI
Dopamine receptor D3 regulates endocytic sorting by a Prazosin-sensitive interaction with the coatomer COPI Xin Zhanga,b,c, Wenchao Wanga,c, Anne V. Bedigiana,b, Margaret L. Coughlind, Timothy J. Mitchisond, and Ulrike S. Eggerta,b,e,1 aDana-Farber Cancer Institute, bDepartment of Biological Chemistry and Molecular Pharmacology, and dDepartment of Systems Biology, Harvard Medical School, Boston, MA 02115; cHigh Magnetic Field Laboratory, Chinese Academy of Sciences, Hefei, 230031, People’s Republic of China; and eDepartment of Chemistry and Randall Division of Cell and Molecular Biophysics, King’s College London, London SE1 1UL, United Kingdom Edited* by Christopher T. Walsh, Harvard Medical School, Boston, MA, and approved June 19, 2012 (received for review May 8, 2012) Macromolecules enter cells by endocytosis and are sorted to different Prazosin, which we describe here as a tool for endocytosis cellular destinations in early/sorting endosomes. The mechanism and research, is an important drug that has been used clinically for regulation of sorting are poorly understood, although transitions decades to treat hypertension, prostate hyperplasia, post- between vesicular and tubular endosomes are important. We found traumatic stress disorder, and scorpion stings (11). Its primary that the antihypertensive drug Prazosin inhibits endocytic sorting by known mechanism is to antagonize α1-adrenergic receptors, an off-target perturbation of the G protein-coupled receptor dopa- a subfamily of GPCRs. GPCR receptor drugs often bind to mine receptor D3 (DRD3). Prazosin is also a potent cytokinesis inhib- GPCRs other than the primary target, and such off-target itor, likely as a consequence of its effects on endosomes. -

COPG (A-10): Sc-393977
SANTA CRUZ BIOTECHNOLOGY, INC. COPG (A-10): sc-393977 BACKGROUND APPLICATIONS Membrane and vesicular trafficking in the early secretory pathway are COPG (A-10) is recommended for detection of COPG of mouse, rat and mediated by non-clathrin COP (coat protein) I-coated vesicles. COPI-coated human origin by Western Blotting (starting dilution 1:100, dilution range vesicles mediate retrograde transport from the Golgi back to the ER and 1:100-1:1000), immunoprecipitation [1-2 µg per 100-500 µg of total protein intra-Golgi transport. The cytosolic precursor of the COPI coat, the heptameric (1 ml of cell lysate)], immunofluorescence (starting dilution 1:50, dilution coatomer complex, is composed of two subcomplexes. The first consists of range 1:50-1:500), immunohistochemistry (including paraffin-embedded the COPB, COPG, COPD and COPZ subunits (also known as β-, γ-, δ- and sections) (starting dilution 1:50, dilution range 1:50-1:500) and solid phase ζ-COP), which are distantly homologous to AP clathrin adaptor subunits. ELISA (starting dilution 1:30, dilution range 1:30-1:3000). The second consists of the COPA, β’-COP and COPE subunits (also known Suitable for use as control antibody for COPG siRNA (h): sc-37254, COPG as α-COP, COPP and ε-COP, respectively). The COPG (γCOP) subunit of the siRNA (m): sc-35092, COPG shRNA Plasmid (h): sc-37254-SH, COPG shRNA coatomer is believed to mediate the binding to the cytoplasmic dilysine Plasmid (m): sc-35092-SH, COPG shRNA (h) Lentiviral Particles: sc-37254-V motifs of membrane proteins. COPG has the highest level of expression in and COPG shRNA (m) Lentiviral Particles: sc-35092-V.