Copenhagen, a Play by Michael Frayn
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hendrik Antoon Lorentz's Struggle with Quantum Theory A. J
Hendrik Antoon Lorentz’s struggle with quantum theory A. J. Kox Archive for History of Exact Sciences ISSN 0003-9519 Volume 67 Number 2 Arch. Hist. Exact Sci. (2013) 67:149-170 DOI 10.1007/s00407-012-0107-8 1 23 Your article is published under the Creative Commons Attribution license which allows users to read, copy, distribute and make derivative works, as long as the author of the original work is cited. You may self- archive this article on your own website, an institutional repository or funder’s repository and make it publicly available immediately. 1 23 Arch. Hist. Exact Sci. (2013) 67:149–170 DOI 10.1007/s00407-012-0107-8 Hendrik Antoon Lorentz’s struggle with quantum theory A. J. Kox Received: 15 June 2012 / Published online: 24 July 2012 © The Author(s) 2012. This article is published with open access at Springerlink.com Abstract A historical overview is given of the contributions of Hendrik Antoon Lorentz in quantum theory. Although especially his early work is valuable, the main importance of Lorentz’s work lies in the conceptual clarifications he provided and in his critique of the foundations of quantum theory. 1 Introduction The Dutch physicist Hendrik Antoon Lorentz (1853–1928) is generally viewed as an icon of classical, nineteenth-century physics—indeed, as one of the last masters of that era. Thus, it may come as a bit of a surprise that he also made important contribu- tions to quantum theory, the quintessential non-classical twentieth-century develop- ment in physics. The importance of Lorentz’s work lies not so much in his concrete contributions to the actual physics—although some of his early work was ground- breaking—but rather in the conceptual clarifications he provided and his critique of the foundations and interpretations of the new ideas. -

Philosophical Rhetoric in Early Quantum Mechanics, 1925-1927
b1043_Chapter-2.4.qxd 1/27/2011 7:30 PM Page 319 b1043 Quantum Mechanics and Weimar Culture FA 319 Philosophical Rhetoric in Early Quantum Mechanics 1925–27: High Principles, Cultural Values and Professional Anxieties Alexei Kojevnikov* ‘I look on most general reasoning in science as [an] opportunistic (success- or unsuccessful) relationship between conceptions more or less defined by other conception[s] and helping us to overlook [danicism for “survey”] things.’ Niels Bohr (1919)1 This paper considers the role played by philosophical conceptions in the process of the development of quantum mechanics, 1925–1927, and analyses stances taken by key participants on four main issues of the controversy (Anschaulichkeit, quantum discontinuity, the wave-particle dilemma and causality). Social and cultural values and anxieties at the time of general crisis, as identified by Paul Forman, strongly affected the language of the debate. At the same time, individual philosophical positions presented as strongly-held principles were in fact flexible and sometimes reversible to almost their opposites. One can understand the dynamics of rhetorical shifts and changing strategies, if one considers interpretational debates as a way * Department of History, University of British Columbia, 1873 East Mall, Vancouver, British Columbia, Canada V6T 1Z1; [email protected]. The following abbreviations are used: AHQP, Archive for History of Quantum Physics, NBA, Copenhagen; AP, Annalen der Physik; HSPS, Historical Studies in the Physical Sciences; NBA, Niels Bohr Archive, Niels Bohr Institute, Copenhagen; NW, Die Naturwissenschaften; PWB, Wolfgang Pauli, Wissenschaftlicher Briefwechsel mit Bohr, Einstein, Heisenberg a.o., Band I: 1919–1929, ed. A. Hermann, K.V. -

Von Richthofen, Einstein and the AGA Estimating Achievement from Fame
Von Richthofen, Einstein and the AGA Estimating achievement from fame Every schoolboy has heard of Einstein; fewer have heard of Antoine Becquerel; almost nobody has heard of Nils Dalén. Yet they all won Nobel Prizes for Physics. Can we gauge a scientist’s achievements by his or her fame? If so, how? And how do fighter pilots help? Mikhail Simkin and Vwani Roychowdhury look for the linkages. “It was a famous victory.” We instinctively rank the had published. However, in 2001–2002 popular French achievements of great men and women by how famous TV presenters Igor and Grichka Bogdanoff published they are. But is instinct enough? And how exactly does a great man’s fame relate to the greatness of his achieve- ment? Some achievements are easy to quantify. Such is the case with fighter pilots of the First World War. Their achievements can be easily measured and ranked, in terms of their victories – the number of enemy planes they shot down. These aces achieved varying degrees of fame, which have lasted down to the internet age. A few years ago we compared1 the fame of First World War fighter pilot aces (measured in Google hits) with their achievement (measured in victories); and we found that We can estimate fame grows exponentially with achievement. fame from Google; Is the same true in other areas of excellence? Bagrow et al. have studied the relationship between can this tell us 2 achievement and fame for physicists . The relationship Manfred von Richthofen (in cockpit) with members of his so- about actual they found was linear. -

Wolfgang Pauli Niels Bohr Paul Dirac Max Planck Richard Feynman
Wolfgang Pauli Niels Bohr Paul Dirac Max Planck Richard Feynman Louis de Broglie Norman Ramsey Willis Lamb Otto Stern Werner Heisenberg Walther Gerlach Ernest Rutherford Satyendranath Bose Max Born Erwin Schrödinger Eugene Wigner Arnold Sommerfeld Julian Schwinger David Bohm Enrico Fermi Albert Einstein Where discovery meets practice Center for Integrated Quantum Science and Technology IQ ST in Baden-Württemberg . Introduction “But I do not wish to be forced into abandoning strict These two quotes by Albert Einstein not only express his well more securely, develop new types of computer or construct highly causality without having defended it quite differently known aversion to quantum theory, they also come from two quite accurate measuring equipment. than I have so far. The idea that an electron exposed to a different periods of his life. The first is from a letter dated 19 April Thus quantum theory extends beyond the field of physics into other 1924 to Max Born regarding the latter’s statistical interpretation of areas, e.g. mathematics, engineering, chemistry, and even biology. beam freely chooses the moment and direction in which quantum mechanics. The second is from Einstein’s last lecture as Let us look at a few examples which illustrate this. The field of crypt it wants to move is unbearable to me. If that is the case, part of a series of classes by the American physicist John Archibald ography uses number theory, which constitutes a subdiscipline of then I would rather be a cobbler or a casino employee Wheeler in 1954 at Princeton. pure mathematics. Producing a quantum computer with new types than a physicist.” The realization that, in the quantum world, objects only exist when of gates on the basis of the superposition principle from quantum they are measured – and this is what is behind the moon/mouse mechanics requires the involvement of engineering. -

11/03/11 110311 Pisp.Doc Physics in the Interest of Society 1
1 _11/03/11_ 110311 PISp.doc Physics in the Interest of Society Physics in the Interest of Society Richard L. Garwin IBM Fellow Emeritus IBM, Thomas J. Watson Research Center Yorktown Heights, NY 10598 www.fas.org/RLG/ www.garwin.us [email protected] Inaugural Lecture of the Series Physics in the Interest of Society Massachusetts Institute of Technology November 3, 2011 2 _11/03/11_ 110311 PISp.doc Physics in the Interest of Society In preparing for this lecture I was pleased to reflect on outstanding role models over the decades. But I felt like the centipede that had no difficulty in walking until it began to think which leg to put first. Some of these things are easier to do than they are to describe, much less to analyze. Moreover, a lecture in 2011 is totally different from one of 1990, for instance, because of the instant availability of the Web where you can check or supplement anything I say. It really comes down to the comment of one of Elizabeth Taylor later spouses-to-be, when asked whether he was looking forward to his wedding, and replied, “I know what to do, but can I make it interesting?” I’ll just say first that I think almost all Physics is in the interest of society, but I take the term here to mean advising and consulting, rather than university, national lab, or contractor research. I received my B.S. in physics from what is now Case Western Reserve University in Cleveland in 1947 and went to Chicago with my new wife for graduate study in Physics. -

On the Linkage Between Planck's Quantum and Maxwell's Equations
The Finnish Society for Natural Philosophy 25 years, K.V. Laurikainen Honorary Symposium, Helsinki 11.-12.11.2013 On the Linkage between Planck's Quantum and Maxwell's Equations Tuomo Suntola Physics Foundations Society, www.physicsfoundations.org Abstract The concept of quantum is one of the key elements of the foundations in modern physics. The term quantum is primarily identified as a discrete amount of something. In the early 20th century, the concept of quantum was needed to explain Max Planck’s blackbody radiation law which suggested that the elec- tromagnetic radiation emitted by atomic oscillators at the surfaces of a blackbody cavity appears as dis- crete packets with the energy proportional to the frequency of the radiation in the packet. The derivation of Planck’s equation from Maxwell’s equations shows quantizing as a property of the emission/absorption process rather than an intrinsic property of radiation; the Planck constant becomes linked to primary electrical constants allowing a unified expression of the energies of mass objects and electromagnetic radiation thereby offering a novel insight into the wave nature of matter. From discrete atoms to a quantum of radiation Continuity or discontinuity of matter has been seen as a fundamental question since antiquity. Aristotle saw perfection in continuity and opposed Democritus’s ideas of indivisible atoms. First evidences of atoms and molecules were obtained in chemistry as multiple proportions of elements in chemical reac- tions by the end of the 18th and in the early 19th centuries. For physicist, the idea of atoms emerged for more than half a century later, most concretely in statistical thermodynamics and kinetic gas theory that converted the mole-based considerations of chemists into molecule-based considerations based on con- servation laws and probabilities. -
![I. I. Rabi Papers [Finding Aid]. Library of Congress. [PDF Rendered Tue Apr](https://docslib.b-cdn.net/cover/8589/i-i-rabi-papers-finding-aid-library-of-congress-pdf-rendered-tue-apr-428589.webp)
I. I. Rabi Papers [Finding Aid]. Library of Congress. [PDF Rendered Tue Apr
I. I. Rabi Papers A Finding Aid to the Collection in the Library of Congress Manuscript Division, Library of Congress Washington, D.C. 1992 Revised 2010 March Contact information: http://hdl.loc.gov/loc.mss/mss.contact Additional search options available at: http://hdl.loc.gov/loc.mss/eadmss.ms998009 LC Online Catalog record: http://lccn.loc.gov/mm89076467 Prepared by Joseph Sullivan with the assistance of Kathleen A. Kelly and John R. Monagle Collection Summary Title: I. I. Rabi Papers Span Dates: 1899-1989 Bulk Dates: (bulk 1945-1968) ID No.: MSS76467 Creator: Rabi, I. I. (Isador Isaac), 1898- Extent: 41,500 items ; 105 cartons plus 1 oversize plus 4 classified ; 42 linear feet Language: Collection material in English Location: Manuscript Division, Library of Congress, Washington, D.C. Summary: Physicist and educator. The collection documents Rabi's research in physics, particularly in the fields of radar and nuclear energy, leading to the development of lasers, atomic clocks, and magnetic resonance imaging (MRI) and to his 1944 Nobel Prize in physics; his work as a consultant to the atomic bomb project at Los Alamos Scientific Laboratory and as an advisor on science policy to the United States government, the United Nations, and the North Atlantic Treaty Organization during and after World War II; and his studies, research, and professorships in physics chiefly at Columbia University and also at Massachusetts Institute of Technology. Selected Search Terms The following terms have been used to index the description of this collection in the Library's online catalog. They are grouped by name of person or organization, by subject or location, and by occupation and listed alphabetically therein. -

The Splendors (And No Miseries) of Mass Spectrometry
Image: David Monniaux, Wikipedia Image: Wired Center for Industrial Mathematics Image: Wikipedia Image: ION-TOF.com The Splendors (and no Miseries) of Mass Spectrometry Theodore Alexandrov Center for Industrial Mathematics, University of Bremen Winterseminar, Alghero, Sardinia, Italy 23 Sep 2009 Image: ION-TOF GmbH Image: www.electrotherapymuseum.com Center for Industrial Did you know that? Mathematics Mass spectrometry (MS) is a technique of analytical chemistry that identifies the elemental composition of sample based on mass-to- charge ratio of charged particles. Did you know that MS is used to – Detect and identify the use of drugs of abuse (dopings) in athletes – Identification of explosives and analysis of explosives in postblast residues – Analyse suspicious powders following the post 9/11 anthrax scare – Monitor the breath of patients by anesthesiologists during surgery – and … 2of 14 Center for Industrial T. Rex: just a big chicken? Mathematics A sample of a bone of Tyrannosaurus rex was discovered in 2000 (sample MOR 1125) 68 million years old Soft tissues were inside the bone A sample was demineralized and studied in a mass-spectrometer Several fragments were found which are very similar to collagen—the most common protein found in bones—from birds, specifically chickens! Wired, 22.06.09 (http://www.wired.com/medtech/genetics/magazine/17-07/ff_originofspecies) 3of 14 Center for Industrial T. Rex: just a big chicken? Mathematics 4of 14 Center for Industrial Early history of MS Mathematics Eugen Goldstein (Potsdam) 1886: invented Kanalstrahlen (anode/channel/positive rays) (-) Image: Michael Hedenus, Der Komet in der Entladungsröhre (+) Image: www.electrotherapymuseum.com Wilhelm Wien (RWTH Aachen,Würzburg,Munich) experimented with anode rays in a magnetic field separated canal rays (ions) according to their mass-to-charge ratio It might be expedient to “abandon the terms cathod rays, canal rays and positive light and to speak only of positive and negative particles". -
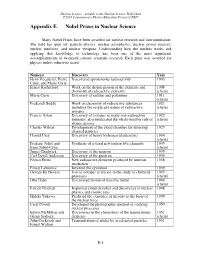
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

Wien Peaks, Planck Distribution Function and Its Decomposition, The
Applied Physics Research; Vol. 12, No. 4; 2020 ISSN 1916-9639 E-ISSN 1916-9647 Published by Canadian Center of Science and Education How to Understand the Planck´s Oscillators? Wien Peaks, Planck Distribution Function and Its Decomposition, the Bohm Sheath Criterion, Plasma Coupling Constant, the Barrier of Determinacy, Hubble Cooling Constant. (24.04.2020) Jiří Stávek1 1 Bazovského 1228, 163 00 Prague, Czech republic Correspondence: Jiří Stávek, Bazovského 1228, 163 00 Prague, Czech republic. E-mail: [email protected] Received: April 23, 2020 Accepted: June 26, 2020 Online Published: July 31, 2020 doi:10.5539/apr.v12n4p63 URL: http://dx.doi.org/10.5539/apr.v12n4p63 Abstract In our approach we have combined knowledge of Old Masters (working in this field before the year 1905), New Masters (working in this field after the year 1905) and Dissidents under the guidance of Louis de Broglie and David Bohm. Based on the great works of Wilhelm Wien and Max Planck we have presented a new look on the “Wien Peaks” and the Planck Distribution Function and proposed the “core-shell” model of the photon. There are known many “Wien Peaks” defined for different contexts. We have introduced a thermodynamic approach to define the Wien Photopic Peak at the wavelength λ = 555 nm and the Wien Scotopic Peak at the wavelength λ = 501 nm to document why Nature excellently optimized the human vision at those wavelengths. There could be discovered many more the so-called Wien Thermodynamic Peaks for other physical and chemical processes. We have attempted to describe the so-called Planck oscillators as coupled oscillations of geons and dyons. -

Quantum Theory, Quantum Mechanics) Part 1
Quantum physics (quantum theory, quantum mechanics) Part 1 1 Outline Introduction Problems of classical physics Black-body Radiation experimental observations Wien’s displacement law Stefan – Boltzmann law Rayleigh - Jeans Wien’s radiation law Planck’s radiation law photoelectric effect observation studies Einstein’s explanation Quantum mechanics Features postulates Summary Quantum Physics 2 Question: What do these have in common? lasers solar cells transistors computer chips CCDs in digital cameras Ipods superconductors ......... Answer: They are all based on the quantum physics discovered in the 20th century. 3 “Classical” vs “modern” physics 4 Why Quantum Physics? “Classical Physics”: developed in 15th to 20th century; provides very successful description “macroscopic phenomena, i.e. behavior of “every day, ordinary objects” o motion of trains, cars, bullets,…. o orbit of moon, planets o how an engine works,.. o Electrical and magnetic phenomena subfields: mechanics, thermodynamics, electrodynamics, “There is nothing new to be discovered in physics now. All that remains is more and more precise measurement.” 5 --- William Thomson (Lord Kelvin), 1900 Why Quantum Physics? – (2) Quantum Physics: developed early 20th century, in response to shortcomings of classical physics in describing certain phenomena (blackbody radiation, photoelectric effect, emission and absorption spectra…) describes microscopic phenomena, e.g. behavior of atoms, photon-atom scattering and flow of the electrons in a semiconductor. -

The Early History of the Niels Bohr Institute
CHAPTER 4.1 Birthplace of a new physics - the early history of the Niels Bohr Institute Peter Robertson* Abstract SCI.DAN.M. The foundation in 1921 of the Niels Bohr Institute in Copenhagen was to prove an important event in the I birth of modern physics. From its modest beginnings as • ONE a small three-storey building and a handful of physicists, HUNDRED the Institute underwent a rapid expansion over the fol lowing years. Under Bohr’s leadership, the Institute provided the principal centre for the emergence of quan YEARS tum mechanics and a new understanding of Nature at OF the atomic level. Over sixty physicists from 17 countries THE came to collaborate with the Danish physicists at the In BOHR stitute during its first decade. The Bohr Institute was the ATOM: first truly international centre in physics and, indeed, one of the first in any area of science. The Institute pro PROCEEDINGS vided a striking demonstration of the value of interna tional cooperation in science and it inspired the later development of similar centres elsewhere in Europe and FROM the United States. In this article I will discuss the origins and early development of the Institute and examine the A CONFERENCE reasons why it became such an important centre in the development of modern physics. Keywords: Niels Bohr Institute; development of quan tum mechanics; international cooperation in science. * School of Physics, University of Melbourne, Melbourne, Vic. 3010, Australia. Email: [email protected]. 481 PETER ROBERTSON SCI.DAN.M. I 1. Planning and construction of the Institute In 1916 Niels Bohr returned home to Copenhagen over four years since his first visit to Cambridge, England, and two years after a second visit to Manchester, working in the group led by Ernest Ru therford.