WO 2017/048727 Al 23 March 2017 (23.03.2017) P O P C T
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
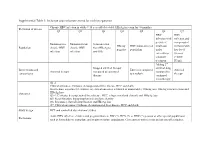
Inclusion and Exclusion Criteria for Each Key Question
Supplemental Table 1: Inclusion and exclusion criteria for each key question Chronic HBV infection in adults ≥ 18 year old (detectable HBsAg in serum for >6 months) Definition of disease Q1 Q2 Q3 Q4 Q5 Q6 Q7 HBV HBV infection with infection and persistent compensated Immunoactive Immunotolerant Seroconverted HBeAg HBV mono-infected viral load cirrhosis with Population chronic HBV chronic HBV from HBeAg to negative population under low level infection infection anti-HBe entecavir or viremia tenofovir (<2000 treatment IU/ml) Adding 2nd Stopped antiviral therapy antiviral drug Interventions and Entecavir compared Antiviral Antiviral therapy compared to continued compared to comparisons to tenofovir therapy therapy continued monotherapy Q1-2: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Intermediate outcomes (if evidence on clinical outcomes is limited or unavailable): HBsAg loss, HBeAg seroconversion and Outcomes HBeAg loss Q3-4: Cirrhosis, decompensated liver disease, HCC, relapse (viral and clinical) and HBsAg loss Q5: Renal function, hypophosphatemia and bone density Q6: Resistance, flare/decompensation and HBeAg loss Q7: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Study design RCT and controlled observational studies Acute HBV infection, children and pregnant women, HIV (+), HCV (+) or HDV (+) persons or other special populations Exclusions such as hemodialysis, transplant, and treatment failure populations. Co treatment with steroids and uncontrolled studies. Supplemental Table 2: Detailed Search Strategy: Ovid Database(s): Embase 1988 to 2014 Week 37, Ovid MEDLINE(R) In-Process & Other Non- Indexed Citations and Ovid MEDLINE(R) 1946 to Present, EBM Reviews - Cochrane Central Register of Controlled Trials August 2014, EBM Reviews - Cochrane Database of Systematic Reviews 2005 to July 2014 Search Strategy: # Searches Results 1 exp Hepatitis B/dt 26410 ("hepatitis B" or "serum hepatitis" or "hippie hepatitis" or "injection hepatitis" or 2 178548 "hepatitis type B").mp. -

Nnrtis – MK1439 New Classes • Maturation Inhibitors, LEDGINS, Etc
New Antiretrovirals and New Strategies Saye Khoo HIV Pharmacology Group Declaration of Interests •www.hiv-druginteractions.org & www.hep-druginteractions.org sponsorship from Janssen, ViiV, Abbott, Merck, BMS, Gilead, Boehringer, Vertex. Editorial content remains independent. •Research Grants: Merck, ViiV •Speakers bureau: Merck, Janssen, Abbott, Roche •Travel grants: Gilead, ViiV, BMS, Janssen •TaiLor trial (NIHR-funded) Antiretroviral Stewardship Myocardial Infarction Stroke Cancer Congnitive impairment Liver, renal etc Plan Now For Then • what to give ? • Minimise resistance • when to start ? • Minimise toxicity • how to manage ? • Preserve options • Normalise Immunity • Equip for future co-morbidity New Drugs, New Formulations, New Strategies Improvements on existing classes • TAF • dolutegravir and other integrases • new NNRTIs – MK1439 New Classes • Maturation inhibitors, LEDGINS, etc New Formulations • nanoformulations • mono- or dual therapy • LA injections or implants • targeting latent reservoirs New Strategies • NRTI-sparing, PI monotherapy • targeting latent reservoirs • targeting immune activation, cardiovascular risk • etc INSTIs NRTIs PIs NNRTIs Other Approved Dolutegravir Phase 3 TAF DRVc Doravirine TAF/FTC/EVGc (MK1349) Cenicriviroc RPV-LA BMS663068 Phase 2 GSK126744 Racivir ABC/3TC/DTG Amodoxovir TAF/FTC/DRVc Elvucitabine Doravirine (MK-1439) • Pharmacology – Potent - IC95 ~19 nM (50% human serum) – Once-daily dosing; T½ 10-16h – P450 metabolism (CYP3A4/5) • No significant inhibition/induction of CYP P450s • No significant -

Classification Decisions Taken by the Harmonized System Committee from the 47Th to 60Th Sessions (2011
CLASSIFICATION DECISIONS TAKEN BY THE HARMONIZED SYSTEM COMMITTEE FROM THE 47TH TO 60TH SESSIONS (2011 - 2018) WORLD CUSTOMS ORGANIZATION Rue du Marché 30 B-1210 Brussels Belgium November 2011 Copyright © 2011 World Customs Organization. All rights reserved. Requests and inquiries concerning translation, reproduction and adaptation rights should be addressed to [email protected]. D/2011/0448/25 The following list contains the classification decisions (other than those subject to a reservation) taken by the Harmonized System Committee ( 47th Session – March 2011) on specific products, together with their related Harmonized System code numbers and, in certain cases, the classification rationale. Advice Parties seeking to import or export merchandise covered by a decision are advised to verify the implementation of the decision by the importing or exporting country, as the case may be. HS codes Classification No Product description Classification considered rationale 1. Preparation, in the form of a powder, consisting of 92 % sugar, 6 % 2106.90 GRIs 1 and 6 black currant powder, anticaking agent, citric acid and black currant flavouring, put up for retail sale in 32-gram sachets, intended to be consumed as a beverage after mixing with hot water. 2. Vanutide cridificar (INN List 100). 3002.20 3. Certain INN products. Chapters 28, 29 (See “INN List 101” at the end of this publication.) and 30 4. Certain INN products. Chapters 13, 29 (See “INN List 102” at the end of this publication.) and 30 5. Certain INN products. Chapters 28, 29, (See “INN List 103” at the end of this publication.) 30, 35 and 39 6. Re-classification of INN products. -

HIV/AIDS Technologies: a Review of Progress to Date and Current Prospects
Working Paper No.6 HIV/AIDS Technologies: A review of progress to date and current prospects COMMISSIONED BY: aids2031 Science and Technology Working Group AUTHORED BY: KEITH ALCORN NAM Publications Disclaimer: The views expressed in this paper are those of the author(s) and do not necessarily reflect the official policy, position, or opinions of the wider aids2031 initiative or partner organizations aids2031 Science and Technology working group A review of progress to date and current prospects October 2008 Acronyms 3TC lamivudine ANRS Agènce Nationale de Récherche sur la Sida ART Antiretroviral therapy ARV Antiretroviral AZT azidothymidine or zidovudine bDNA branched DNA CDC US Centers for Disease Control CHER Children with HIV Early Antiretroviral therapy (study) CTL Cytotoxic T-lymphocyte D4T stavudine DSMB Data and Safety Monitoring Board EFV Efavirenz ELISA Enzyme Linked Immunosorbent Assay FDC Fixed-dose combination FTC Emtricitabine HAART Highly Active Antiretroviral Therapy HBAC Home-based AIDS care HCV Hepatitis C virus HPTN HIV Prevention Trials Network HSV-2 Herpes simplex virus type 2 IAVI International AIDS Vaccine Initiative IL-2 Interleukin-2 LED Light-emitting diode LPV/r Lopinavir/ritonavir MIRA Methods for Improving Reproductive Health in Africa trial MSF Médecins sans Frontières MSM Men who have sex with men MVA Modified vaccinia Ankara NIH US National Institutes of Health NRTI Nucleoside reverse transcriptase inhibitor NNRTI Non-nucleoside reverse transcriptase inhibitor OBT Optimised background therapy PCR Polymerase -

Multi-Class Immune-Based Therap I Es Co Combination Drugs AZ T
m- IMMUNE-BASED THERAP o /R) c VIR, A LPV ) MULTI-CLASSA NFV VIR, A S T N (INDIN COMBINATION DRUGSA VIR, REZIST AZ R 754) P A ) AVX C + Inhibitors Protease RTV VIR/RITON ) CRIXIV AB A OM (KIVEXA, COMBIVIR (ZIDOVUDINE + LAMIVUDINE, AZT + 3TC) EMTRIVA Protease754, Inhibitors C VIR, (EMTRICITABINE, FTC) EPIVIR (LAMIVUDINE, 3TC) EPZICOM (KIVEXA, TPV T A PZI HE EPT (NELFIN SPD OVIR DISOPROXIL ABACAVIR + LAMIVUDINE, ABC + 3TC) RETROVIR (ZIDOVUDINE, AZT, E HIBITO F VIR, , LOPIN ZDV) TRIZIVIR (ABACAVIR + ZIDOVUDINE + LAMIVUDINE, ABC + AC A A S IR N IN TRUGGLE FOR AZT + 3TC) TRUVADA (TENOFOVIR DF + EMTRICITABINE, TDF + V ENO A ) T ABINE ( ABINE FTC) VIDEX & VIDEX EC (DIDANOSINE, DDI) VIREAD (TENOFOVIR T TV I THE (ALUVI C DISOPROXIL FUMARATE, TDF) ZERIT (STAVUDINE, D4T) ZIAGEN A (ABACAVIR, ABC) RACIVIR (RCV) AMDOXOVIR (AMDX, DAPD) ORVIR (RITON N PRI VIR, A ) IREAD ( A A V APRICITABINE (SPD754, AVX754)ELVUCITABINE (ACH- TORS ) LETR N ) A I 126,443, BETA-L-FD4C) COMBIVIR (ZIDOVUDINE + FPV A Z I LAMIVUDINE, AZT + 3TC) EMTRIVA (EMTRICITABINE, ) K A ) APTIVUS (TIPR DAPD T VIR, IPTASE IPTASE , A FTC) EPIVIR (LAMIVUDINE, 3TC) EPZICOM (KIVEXA, A PV A SQV CCESS TO ABACAVIR + LAMIVUDINE, ABC + 3TC) RETROVIR R Z ( PIVIR (LAMIVUDINE, 3TC) B A This book documents the struggle that has been faced by those E (ZIDOVUDINE, AZT, ZDV) TRIZIVIR (ABACAVIR + T AMDX VIR, VIR, A A A I STRUGGLEA requiring treatment for HIV/AIDS in India, and those affected ZIDOVUDINE + LAMIVUDINE, ABC + AZT + 3TC) MPREN AVIR + ZIDOVUDINE + LAMIVUDINE, SC EY A C TRUVADA (TENOFOVIR DF + EMTRICITABINE, by HIV/AIDS, since the first recorded incidence of HIV/AIDS in FOR R N ) TDF + FTC) VIDEX & VIDEX EC (DIDANOSINE, QUIN India in 1986. -

AVCC Figure Template
Schinazi 11/2/08 14:00 Page 343 Antiviral Chemistry & Chemotherapy 18:343–346 Short communication Cellular pharmacology of 9-(β-D-1,3-dioxolan-4-yl) guanine and its lack of drug interactions with zidovudine in primary human lymphocytes Brenda I Hernandez-Santiago, Aleksandr Obikhod, Emilie Fromentin, Selwyn J Hurwitz and Raymond F Schinazi* Center for AIDS Research, Laboratory of Biochemical Pharmacology, Department of Pediatrics, Emory University School of Medicine, and Veterans Affairs Medical Center, Decatur, GA 30033, USA *Corresponding author: Tel: 404 728 7711; Fax: 404 728 7726; E-mail: [email protected] Amdoxovir, currently in Phase II clinical trials, is different resistance mutations, co-formulation of rapidly converted to 9-(β-D-1,3-dioxolan-4- the these two drugs is an attractive proposition. A yl)guanine (DXG) by adenosine deaminase in vitro combination study between DXG and ZDV and in humans. The cellular pharmacology of DXG showed no reduction of DXG-TP or ZDV-TP. Taken in primary human lymphocytes, including together, these results suggest that an appropri- dose–response relationships, intracellular half-life ately designed DXG prodrug could be given once of DXG triphosphate (DXG-TP), and combination a day and that co-formulation with ZDV might be studies were determined. DXG produced high a possibility. levels of DXG-TP with a long half-life (16 h) in acti- vated human peripheral blood mononuclear cells. Keywords: cellular pharmacology, DXG, nucleoside Since zidovudine (ZDV) and DXG select for analogues, NRTI The emergence of resistant HIV strains during therapy has inhibitor of HIV-1, HIV-2 and hepatitis B virus (HBV) made it a major challenge to develop drugs that delay, in human cell lines. -

This Project Has Been Supported with Unrestriced Grants from Abbvie Gilead Sciences HEXAL Janssen-Cilag MSD Viiv Healthcare By
This project has been supported with unrestriced grants from AbbVie Gilead Sciences HEXAL Janssen-Cilag MSD ViiV Healthcare By Marcus Altfeld, Hamburg/Boston (USA) Achim Barmeyer, Dortmund Georg Behrens, Hannover Dirk Berzow, Hamburg Christoph Boesecke, Bonn Patrick Braun, Aachen Thomas Buhk, Hamburg Rob Camp, Barcelona (Spain/USA) Rika Draenert, Munich Christian Eggers, Linz (Austria) Stefan Esser, Essen Gerd Fätkenheuer, Cologne Gunar Günther, Windhoek (Namibia) Thomas Harrer, Erlangen Christian Herzmann, Borstel Christian Hoffmann, Hamburg Heinz-August Horst, Kiel Martin Hower, Dortmund Christoph Lange, Borstel Thore Lorenzen, Hamburg Tim Niehues, Krefeld Christian Noah, Hamburg Ramona Pauli, Munich Ansgar Rieke, Koblenz Jürgen Kurt Rockstroh, Bonn Thorsten Rosenkranz, Hamburg Bernhard Schaaf, Dortmund Ulrike Sonnenberg-Schwan, Munich Christoph D. Spinner, Munich Thomas Splettstoesser (Figures), Berlin Matthias Stoll, Hannover Hendrik Streeck, Essen/Boston (USA) Jan Thoden, Freiburg Markus Unnewehr, Dortmund Mechthild Vocks-Hauck, Berlin Jan-Christian Wasmuth, Bonn Michael Weigel, Schweinfurt Thomas Weitzel, Santiago (Chile) Eva Wolf, Munich HIV 2015/16 www.hivbook.com Edited by Christian Hoffmann and Jürgen K. Rockstroh Medizin Fokus Verlag IV Christian Hoffmann, M.D., Ph.D. ICH Stadtmitte (Infektionsmedizinisches Centrum Hamburg) Glockengiesserwall 1 20095 Hamburg, Germany Phone: + 49 40 2800 4200 Fax: + 49 40 2800 42020 [email protected] Jürgen K. Rockstroh, M.D., Ph.D. Department of Medicine I University of Bonn Sigmund-Freud-Strasse 25 53105 Bonn, Germany Phone: + 49 228 287 6558 Fax: + 49 228 287 5034 [email protected] HIV Medicine is an ever-changing field. The editors and authors of HIV 2015/16 have made every effort to provide information that is accurate and complete as of the date of publication. -

Wo 2009/148600 A2
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 10 December 2009 (10.12.2009) WO 2009/148600 A2 (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every C07C 311/41 (2006.01) A61K 31/661 (2006.01) kind of national protection available): AE, AG, AL, AM, A61K 31/63 (2006.01) C07F 9/141 (2006.01) AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, (21) International Application Number: DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, PCT/US2009/003391 HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, (22) International Filing Date: KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, 4 June 2009 (04.06.2009) ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PG, PH, PL, PT, RO, RS, RU, SC, SD, SE, (25) Filing Language: English SG, SK, SL, SM, ST, SV, SY, TJ, TM, TN, TR, TT, TZ, (26) Publication Language: English UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: (84) Designated States (unless otherwise indicated, for every 61/059,556 6 June 2008 (06.06.2008) US kind of regional protection available): ARIPO (BW, GH, GM, KE, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, ZM, (71) Applicant (for all designated States except US): CON¬ ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, CERT PHARMACEUTICALS, INC. -

What's in the Pipeline: New HIV Drugs, Vaccines, Microbicides, HCV And
What’s in the Pipeline: New HIV Drugs, Vaccines, Microbicides, HCV and TB Treatments in Clinical Trials by Rob Camp, Richard Jefferys, Tracy Swan & Javid Syed edited by Mark Harrington & Bob Huff Treatment Action Group New York, NY, USA July 2005 e thymidine • BI-201 • Racivir (PSI 5004) • TMC-278 • Diarylpyrimidine (DAPY) • 640385 • Reverset (D-D4FC) • JTK-303 • UK-427 (maraviroc) • Amdoxovir • AMD-070 • Vicriviroc LIPO-5 • GTU-Multi-HIV • pHIS-HIV-B • rFPV-HIV-B • ADMVA • GSK Protein HIV Vaccine TBC-M335 (MVA) • TBC-F357 (FPV) • TBC-F349 (FPV) • LIPO-4T (LPHIV-1) • LFn-p24 • H G • Oligomeric gp140/MF59 • VRC-HIVDNA-009-00-VP • PolyEnv1 • ISS P-001 • EP HIV- • BufferGel • Lactin-V • Protected Lactobacilli in combination with BZK • Tenofovir/PMPA G ulose acetate/CAP) • Lime Juice • TMC120 • UC-781 • VivaGel (SPL7013 gel) • ALVAC Ad5 • Autologous dendritic cells pulsed w/ALVAC • Autologous dendritic cell HIV vaccination x • Tat vaccine • GTU-nef DNA vaccine • Interleukin-2 (IL-2) • HE2000 • Pegasys (peginter L-4/IL-13 trap • Serostim • Tucaresol • MDX-010 anti-CTLA4 antibody • Cyclosporine A • 496 • HGTV43 • M87o • Vertex • VX-950 • Idenix • Valopicitabine (NM283) • JTK-003 mplant • Albuferon • Celgosivir (MBI-3253) • IC41 • INN0101 • Tarvicin • ANA971 (oral) floxacin, Tequin • J, TMC207 (ex R207910) • LL-3858 • M, moxifloxacin, Avelox • PA-824 Acknowledgements. Thanks to our intrepid editors, Bob Huff, copy-editor Andrea Dailey, and proof-reader Jen Curry, to awesome layout expert Lei Chou, to webmaster Joel Beard, to Joe McConnell for handling administrative matters related to the report, and most of all to the board and supporters of TAG for making our work possible. -

Synthetic Compounds with 2-Amino-1, 3, 4-Thiadiazole Moiety
molecules Review Synthetic Compounds with 2-Amino-1,3,4-Thiadiazole Moiety Against Viral Infections Georgeta Serban Pharmaceutical Chemistry Department, Faculty of Medicine and Pharmacy, University of Oradea, 29 Nicolae Jiga, 410028 Oradea, Romania; [email protected]; Tel.: +4-0756-276-377 Academic Editor: Antonio Carta Received: 29 January 2020; Accepted: 18 February 2020; Published: 19 February 2020 Abstract: Viral infections have resulted in millions of victims in human history. Although great efforts have been made to find effective medication, there are still no drugs that truly cure viral infections. There are currently approximately 90 drugs approved for the treatment of human viral infections. As resistance toward available antiviral drugs has become a global threat to health, there is an intrinsic need to identify new scaffolds that are useful in discovering innovative, less toxic and highly active antiviral agents. 1,3,4-Thiadiazole derivatives have been extensively studied due to their pharmacological profile, physicochemical and pharmacokinetic properties. This review provides an overview of the various synthetic compounds containing the 2-amino-1,3,4-thiadiazole moiety that has been evaluated for antiviral activity against several viral strains and could be considered possible prototypes for the development of new antiviral drugs. Keywords: 2-amino-1,3,4-thiadiazole; viral infections; antiviral agents; drug resistance; inhibitory concentration; mechanism of action 1. Human Viral Infections Viruses are the smallest among -

Background Paper 6.7 Human Immunodeficiency Virus (HIV)/ Acquired Immune Deficiency Syndromes (AIDS)
Priority Medicines for Europe and the World "A Public Health Approach to Innovation" Update on 2004 Background Paper Written by Warren Kaplan Background Paper 6.7 Human Immunodeficiency Virus (HIV)/ Acquired Immune Deficiency Syndromes (AIDS) By Warren Kaplan, Ph.D., JD, MPH 15 February 2013 Update on 2004 Background Paper, BP 6.7 HIV/AIDS Table of Contents What is new since 2004? ..................................................................................................................................... 4 1. Introduction ................................................................................................................................................. 7 2. What are the Epidemiological Trends for Europe and the World? ................................................... 7 2.1 Western and Central Europe ............................................................................................................. 7 2.2 Eastern Europe .................................................................................................................................... 9 2.2 The World (including Europe) ........................................................................................................ 11 3. What is the Control Strategy? Is There an Effective Package of Control Methods Assembled into a “Control Strategy” for Most Epidemiological Settings?................................................................. 13 3.1 Is there a pharmaceutical ‘gap’? .................................................................................................... -

Dedication to Kyoichi A. Watanabe
Heterocycl. Commun. 2015; 21(5): 245–248 Guest Editorial Dedication to Kyoichi A. Watanabe DOI 10.1515/hc-2015-0206 synthesize a 2-fluoro-2-deoxy-D-arabinose derivative from D-glucose [9]. This issue of Heterocyclic Communications is dedicated The above arabinose was key in synthesis of F- ara- to Kyoichi A. ‘Kyo’ Watanabe (1935–2015), formerly of the nucleosides that have anti-viral activity [10–13], anti-cancer Memorial Sloan Kettering Cancer Center (MSKCC), Codon activity [14–16] and are commercially available today. Impor- Pharmaceuticals and Pharmasset, Inc. Kyo earned his PhD tant examples of these nucleosides include 1-(2′-deoxy-2′- in the Mizuno lab at Hokkaido University. He was a member fluoro-1-β-D-arabinofuranosyl)-5-methyluracil (FMAU) and of the American Chemical Society, the Polish Academy 1-(2-deoxy-2-fluoro-1-β-D-arabinofuranosyl)-5-iodouracil of Sciences and a founding member of the International (FIAU, fialuridine). FMAU advanced to clinical trials for Society of Nucleosides, Nucleotides and Nucleic Acids cancer [17] and a radiolabeled FMAU has been used as an (IS3NA). As the head of the Organic Chemistry Laboratory imaging agent [18]. FIAU reached clinical trials for hepatitis at MSKCC and Vice President for Research and Develop- B but was withdrawn due to safety concerns [19]. However, ment at Pharmasset he was an eminent organic chemist the most notable example is probably 2′-F-ara-C. Radiola- who trained and influenced the careers of numerous sci- beled versions are used in positron emission tomography entists. Kyo’s impact has been recognized in five interna- imaging and for in vivo prediction of clinical response to tional awards, the most notable and recent being the Marie gemcitabine [20].