Drug Interactions Between Hormonal Contraceptives and Antiretrovirals
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Everything You Need to Know About the Contraceptive Implant FDA Approval Means US Women Now Have a Progestin-Only Implant Among Their Birth Control Options
OBGM_0906_Darney.final 8/18/06 11:36 AM Page 50 OBGMANAGEMENT Everything you need to know about the contraceptive implant FDA approval means US women now have a progestin-only implant among their birth control options n July 17, the US Food and Drug or even monthly action on the part of the Administration (FDA) approved user, it is well-suited for: Philip D. Darney, MD, MSc what may be the most effective • Women who wish to or need Professor and Chief, Department O of Obstetrics, Gynecology, and hormonal contraceptive ever developed, a to avoid estrogen Reproductive Sciences, University single-rod implant that goes by the trade • Teens who find adherence to a of California-San Francisco, name Implanon. The implant contains 68 contraceptive regimen difficult San Francisco General Hospital ® Dowden Health Media mg of etonogestrel (ENG), the active • Healthy adult women who desire metabolite of desogestrel, in a membrane long-term protection ofCopyright ethylene vinylFor acetate. personal In clinical usetrials only• Women who are breastfeeding involving 20,648 cycles of exposure, only 6 pregnancies occurred, for a cumulative 1 Pearl Index of 0.38 per 100 woman-years. ❚ IN THIS ARTICLE This article reviews: Pros and cons • the 2 contraceptive mechanisms Advantages include: ❙ How the • indications and patient selection • Cost. A study2 of 15 contraceptive progestin-only • pharmacology, safety, adverse effects methods found the implant cost-effec- implant prevents • patient satisfaction and discontinua- tive compared to short-acting methods, tion rates provided it was used long-term. As of fertilization • insertion and removal press time, the manufacturer had not Page 52 • key points of patient counseling released the price. -

Contraception: Choosing the Right Method for You
Contraception: Choosing the Right Method for You Megan Sax, MD and James L. Whiteside, MD What’s available? Choosing a method of contraception can be overwhelming. The most commonly used methods of reversible contraception in the United States are: hormonal methods the intrauterine device (IUD) the implant barrier methods (e.g. male condom)1 Friends, family, and the Internet are full of stories of failed contraception or bad reactions and these stories can have a big influence that doesn’t always line up with the facts.2 However, knowing these facts is critical to figuring out what type will work best for you. Highly Effective Contraception The best place to start in choosing your contraception is to determine when, if ever, you are planning on starting a family. If you do not wish to become pregnant in the next year, a Long-Acting Reversible Contraceptive (LARC) device may be a good option. LARCs include an implant placed under the skin of the upper, inner arm (brand name Nexplanon) and the IUD. IUDs are placed inside the uterus (see Figure 1). They use copper (brand name ParaGard) or hormones (brand names: Mirena, Lilleta, Skyla, Kyleena) to stop a pregnancy from happening. LARCs are the most effective reversible form of contraception. Less than 1% of users experience unintended pregnancy during the first year of use.3 Currently, the implant Nexplanon is effective for 3 years. The hormonal IUD may be used for 3 to 5 years, depending on the brand. The copper IUD works for 10 years. These devices are inserted and removed by a medical care provider. -

Pharmacokinetic Interactions Between Herbal Medicines and Drugs: Their Mechanisms and Clinical Relevance
life Review Pharmacokinetic Interactions between Herbal Medicines and Drugs: Their Mechanisms and Clinical Relevance Laura Rombolà 1 , Damiana Scuteri 1,2 , Straface Marilisa 1, Chizuko Watanabe 3, Luigi Antonio Morrone 1, Giacinto Bagetta 1,2,* and Maria Tiziana Corasaniti 4 1 Preclinical and Translational Pharmacology, Department of Pharmacy, Health and Nutritional Sciences, Section of Preclinical and Translational Pharmacology, University of Calabria, 87036 Rende, Italy; [email protected] (L.R.); [email protected] (D.S.); [email protected] (S.M.); [email protected] (L.A.M.) 2 Pharmacotechnology Documentation and Transfer Unit, Preclinical and Translational Pharmacology, Department of Pharmacy, Health and Nutritional Sciences, University of Calabria, 87036 Rende, Italy 3 Department of Physiology and Anatomy, Tohoku Pharmaceutical University, 981-8558 Sendai, Japan; [email protected] 4 School of Hospital Pharmacy, University “Magna Graecia” of Catanzaro and Department of Health Sciences, University “Magna Graecia” of Catanzaro, 88100 Catanzaro, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-0984-493462 Received: 28 May 2020; Accepted: 30 June 2020; Published: 4 July 2020 Abstract: The therapeutic efficacy of a drug or its unexpected unwanted side effects may depend on the concurrent use of a medicinal plant. In particular, constituents in the medicinal plant extracts may influence drug bioavailability, metabolism and half-life, leading to drug toxicity or failure to obtain a therapeutic response. This narrative review focuses on clinical studies improving knowledge on the ability of selected herbal medicines to influence the pharmacokinetics of co-administered drugs. Moreover, in vitro studies are useful to anticipate potential herbal medicine-drug interactions. -
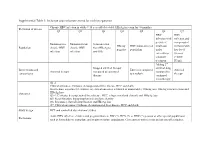
Inclusion and Exclusion Criteria for Each Key Question
Supplemental Table 1: Inclusion and exclusion criteria for each key question Chronic HBV infection in adults ≥ 18 year old (detectable HBsAg in serum for >6 months) Definition of disease Q1 Q2 Q3 Q4 Q5 Q6 Q7 HBV HBV infection with infection and persistent compensated Immunoactive Immunotolerant Seroconverted HBeAg HBV mono-infected viral load cirrhosis with Population chronic HBV chronic HBV from HBeAg to negative population under low level infection infection anti-HBe entecavir or viremia tenofovir (<2000 treatment IU/ml) Adding 2nd Stopped antiviral therapy antiviral drug Interventions and Entecavir compared Antiviral Antiviral therapy compared to continued compared to comparisons to tenofovir therapy therapy continued monotherapy Q1-2: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Intermediate outcomes (if evidence on clinical outcomes is limited or unavailable): HBsAg loss, HBeAg seroconversion and Outcomes HBeAg loss Q3-4: Cirrhosis, decompensated liver disease, HCC, relapse (viral and clinical) and HBsAg loss Q5: Renal function, hypophosphatemia and bone density Q6: Resistance, flare/decompensation and HBeAg loss Q7: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Study design RCT and controlled observational studies Acute HBV infection, children and pregnant women, HIV (+), HCV (+) or HDV (+) persons or other special populations Exclusions such as hemodialysis, transplant, and treatment failure populations. Co treatment with steroids and uncontrolled studies. Supplemental Table 2: Detailed Search Strategy: Ovid Database(s): Embase 1988 to 2014 Week 37, Ovid MEDLINE(R) In-Process & Other Non- Indexed Citations and Ovid MEDLINE(R) 1946 to Present, EBM Reviews - Cochrane Central Register of Controlled Trials August 2014, EBM Reviews - Cochrane Database of Systematic Reviews 2005 to July 2014 Search Strategy: # Searches Results 1 exp Hepatitis B/dt 26410 ("hepatitis B" or "serum hepatitis" or "hippie hepatitis" or "injection hepatitis" or 2 178548 "hepatitis type B").mp. -

Indinavir Sulfate Capsule Merck & Co., Inc
CRIXIVAN - indinavir sulfate capsule Merck & Co., Inc. ---------- CRIXIVAN® (INDINAVIR SULFATE) CAPSULES DESCRIPTION CRIXIVAN1 (indinavir sulfate) is an inhibitor of the human immunodeficiency virus (HIV) protease. CRIXIVAN Capsules are formulated as a sulfate salt and are available for oral administration in strengths of 100, 200, 333, and 400 mg of indinavir (corresponding to 125, 250, 416.3, and 500 mg indinavir sulfate, respectively). Each capsule also contains the inactive ingredients anhydrous lactose and magnesium stearate. The capsule shell has the following inactive ingredients and dyes: gelatin, titanium dioxide, silicon dioxide and sodium lauryl sulfate. The chemical name for indinavir sulfate is [1(1S,2R),5(S)]-2,3,5-trideoxy-N-(2,3-dihydro-2-hydroxy-1H-inden-1-yl)-5-[2-[[(1,1 dimethylethyl)amino]carbonyl]-4-(3-pyridinylmethyl)-1-piperazinyl]-2-(phenylmethyl)-D-erythro-pentonamide sulfate (1:1) salt. Indinavir sulfate has the following structural formula: Indinavir sulfate is a white to off-white, hygroscopic, crystalline powder with the molecular formula C36H47N5O4• H2SO4 and a molecular weight of 711.88. It is very soluble in water and in methanol. 1 Registered trademark of MERCK & CO., Inc. COPYRIGHT © 1996, 1997, 1998, 1999, 2004 MERCK & CO., Inc. All rights reserved MICROBIOLOGY Mechanism of Action HIV-1 protease is an enzyme required for the proteolytic cleavage of the viral polyprotein precursors into the individual functional proteins found in infectious HIV-1. Indinavir binds to the protease active site and inhibits the activity of the enzyme. This inhibition prevents cleavage of the viral polyproteins resulting in the formation of immature non-infectious viral particles. -

Contraception Pearls for Practice
Contraception Pearls for Practice Academic Detailing Service Planning committee Content Experts Clinical reviewer Gillian Graves MD FRCS(C), Professor, Department of Obstetrics and Gynecology, Faculty of Medicine, Dalhousie University Drug evaluation pharmacist Pam McLean-Veysey BScPharm, Drug Evaluation Unit, Nova Scotia Health Family Physician Advisory Panel Bernie Buffett MD, Neils Harbour, Nova Scotia Ken Cameron BSc MD CCFP, Dartmouth, Nova Scotia Norah Mogan MD CCFP, Liverpool, Nova Scotia Dalhousie CPD Bronwen Jones MD CCFP – Family Physician, Director Evidence-based Programs in CPD, Associate Professor, Faculty of Medicine, Dalhousie University Michael Allen MD MSc – Family Physician, Professor, Post-retirement Appointment, Consultant Michael Fleming MD CCFP FCFP – Family Physician, Director Family Physician Programs in CPD Academic Detailers Isobel Fleming BScPharm ACPR, Director of Academic Detailing Service Lillian Berry BScPharm Julia Green-Clements BScPharm Kelley LeBlanc BScPharm Gabrielle Richard-McGibney BScPharm, BCPS, PharmD Cathy Ross RN BScNursing Thanks to Katie McLean, Librarian Educator, NSHA Central Zone for her help with literature searching. Cover artwork generated with Tagxedo.com Disclosure statements The Academic Detailing Service is operated by Dalhousie Continuing Professional Development, Faculty of Medicine and funded by the Nova Scotia Department of Health and Wellness. Dalhousie University Office of Continuing Professional Development has full control over content. Dr Bronwen Jones receives funding for her Academic Detailing work from the Nova Scotia Department of Health and Wellness. Dr Michael Allen has received funding from the Nova Scotia Department of Health and Wellness for research projects and to develop CME programs. Dr Gillian Graves has received funding for presentations from Actavis (Fibristal®) and is on the board of AbbVie (for Lupron®). -

Pharmacokinetic Interactions of Drugs with St John's Wort
http://www.paper.edu.cn Pharmacokinetic interactions of Journal of Psychopharmacology 18(2) (2004) 262–276 © 2004 British Association drugs with St John’s wort for Psychopharmacology ISSN 0269-8811 SAGE Publications Ltd, London, Thousand Oaks, CA and New Delhi 10.1177/0269881104042632 Shufeng Zhou Department of Pharmacy, Faculty of Science, National University of Singapore, Singapore. Eli Chan Department of Pharmacy, Faculty of Science, National University of Singapore, Singapore. Shen-Quan Pan Department of Biological Sciences, Faculty of Science, National University of Singapore, Singapore. Min Huang Institute of Clinical Pharmacology, School of Pharmaceutical Sciences, Sun Yat-Sen University, Guangzhou 510089, PR China. Edmund Jon Deoon Lee Department of Pharmacology, Faculty of Medicine, National University of Singapore, Singapore. Abstract There is a worldwide increasing use of herbs which are often cancer patients receiving irinotecan treatment. St John’s wort did not administered in combination with therapeutic drugs, raising the alter the pharmacokinetics of tolbutamide, but increased the incidence potential for herb–drug interactions. St John’s wort (Hypericum of hypoglycaemia. Several cases have been reported that St John’s wort perforatum) is one of the most commonly used herbal antidepressants. A decreased cyclosporine blood concentration leading to organ rejection. literature search was performed using Medline (via Pubmed), Biological St John’s wort caused breakthrough bleeding and unplanned pregnancies Abstracts, Cochrane Library, AMED, PsycINFO and Embase (all from their when used concomitantly with oral contraceptives. It also caused inception to September 2003) to identify known drug interaction with serotonin syndrome when coadministered with selective serotonin- St John’s wort. The available data indicate that St John’s wort is a reuptake inhibitors (e.g. -

Copper Intrauterine Device (IUD)
Copper Intrauterine Device (IUD) What is it? An IUD is a small, flexible device which is inserted into the uterus by a health professional to prevent pregnancy. The copper IUD is a plastic frame with copper bands and/or wire. It has fine threads which extend through the cervix into the vagina. Once inserted, IUDs are not usually noticeable to you or your partner/s. How does it work? The copper IUD prevents pregnancy by: Quick Facts • affecting sperm movement so they cannot move through the uterus • affecting egg movement, and in the rare instance of an egg being A long-acting reversible fertilised, preventing the egg from attaching to the lining of the uterus. contraception (LARC). One of the most effective How effective is it? methods of contraception The copper IUD is more than 99% effective at preventing pregnancy and can last for 5-10 years (depending on the type). Copper IUDs inserted in available in Australia. people over 40 can be left until after menopause. Method Copper IUDs can also be used as a very effective form of emergency Non hormonal contraception, up to five days after unprotected sex or up to day 12 of your menstrual cycle. Contact the Sexual Health Helpline for more Effectiveness information. More than 99% Who can use it? Return to Fertility A health professional will take a detailed medical history and pelvic No delay examination to ensure that an IUD is suitable for you. Availability Copper IUDs are suitable for those who: Simple insertion and • are looking for very effective and reliable long-term contraception removal by a health • cannot tolerate hormones due to side effects or other medical conditions professional. -

CYP3A4 Mediated Pharmacokinetics Drug Interaction Potential of Maha
www.nature.com/scientificreports OPEN CYP3A4 mediated pharmacokinetics drug interaction potential of Maha‑Yogaraj Gugglu and E, Z guggulsterone Sarvesh Sabarathinam1, Satish Kumar Rajappan Chandra2 & Vijayakumar Thangavel Mahalingam1* Maha yogaraja guggulu (MYG) is a classical herbomineral polyherbal formulation being widely used since centuries. The aim of this study was to investigate the efect of MYG formulation and its major constituents E & Z guggulsterone on CYP3A4 mediated metabolism. In vitro inhibition of MYG and Guggulsterone isomers on CYP3A4 was evaluated by high throughput fuorometric assay. Eighteen Adult male Sprague–Dawley rats (200 ± 25 g body weight) were randomly divided into three groups. Group A, Group B and Group C were treated with placebo, MYG and Standard E & Z guggulsterone for 14 days respectively by oral route. On 15th day, midazolam (5 mg/kg) was administered orally to all rats in each group. Blood samples (0.3 mL) were collected from the retro orbital vein at 0.25, 0.5, 0.75, 1, 2, 4, 6, 12 and 24 h of each rat were collected. The fndings from the in vitro & in vivo study proposed that the MYG tablets and its guggulsterone isomers have drug interaction potential when consumed along with conventional drugs which are CYP3A4 substrates. In vivo pharmacokinetic drug interaction study of midazolam pointed out that the MYG tablets and guggulsterone isomers showed an inhibitory activity towards CYP3A4 which may have leads to clinically signifcant interactions. Te use of alternative medicine such as herbal medicines, phytonutrients, ayurvedic products and nutraceuticals used widely by the majority of the patients for their primary healthcare needs. -

Pp375-430-Annex 1.Qxd
ANNEX 1 CHEMICAL AND PHYSICAL DATA ON COMPOUNDS USED IN COMBINED ESTROGEN–PROGESTOGEN CONTRACEPTIVES AND HORMONAL MENOPAUSAL THERAPY Annex 1 describes the chemical and physical data, technical products, trends in produc- tion by region and uses of estrogens and progestogens in combined estrogen–progestogen contraceptives and hormonal menopausal therapy. Estrogens and progestogens are listed separately in alphabetical order. Trade names for these compounds alone and in combination are given in Annexes 2–4. Sales are listed according to the regions designated by WHO. These are: Africa: Algeria, Angola, Benin, Botswana, Burkina Faso, Burundi, Cameroon, Cape Verde, Central African Republic, Chad, Comoros, Congo, Côte d'Ivoire, Democratic Republic of the Congo, Equatorial Guinea, Eritrea, Ethiopia, Gabon, Gambia, Ghana, Guinea, Guinea-Bissau, Kenya, Lesotho, Liberia, Madagascar, Malawi, Mali, Mauritania, Mauritius, Mozambique, Namibia, Niger, Nigeria, Rwanda, Sao Tome and Principe, Senegal, Seychelles, Sierra Leone, South Africa, Swaziland, Togo, Uganda, United Republic of Tanzania, Zambia and Zimbabwe America (North): Canada, Central America (Antigua and Barbuda, Bahamas, Barbados, Belize, Costa Rica, Cuba, Dominica, El Salvador, Grenada, Guatemala, Haiti, Honduras, Jamaica, Mexico, Nicaragua, Panama, Puerto Rico, Saint Kitts and Nevis, Saint Lucia, Saint Vincent and the Grenadines, Suriname, Trinidad and Tobago), United States of America America (South): Argentina, Bolivia, Brazil, Chile, Colombia, Dominican Republic, Ecuador, Guyana, Paraguay, -

Plan C: Copper IUD As Emergency Contraception IMPLEMENTATION TOOLKIT for Administrators and Clinicians
Plan C: Copper IUD as Emergency Contraception IMPLEMENTATION TOOLKIT for Administrators and Clinicians March 2016 Developed by TABLE OF CONTENTS SECTION 1: OVERVIEW ● Introduction Page 1 ● Background Page 2 ● Who It’s For Page 3 ● How to Use It Page 4 ● Additional Considerations Page 5 SECTION 2: ADMINISTRATIVE ● Pre-Implementation Tools Page 6 1.1 Overview: Plan C 1.2 Checklist: Pre-Implementation 1.3 Staff Buy-in 1.4 Checklist: Policies and Procedures 1.5 Sample: Policies and Procedures 1.6 Marketing Plan C 1.7 Sample: Data Collection Tool SECTION 3: CLINICAL ● Implementation Tools Page 21 2.1 The Facts: The Copper-T as Plan C 2.2 Sample: EC Screening Questionnaire 2.3 Triage Scripts 2.4 Contraceptive Counseling 2.5 Eligibility Flowchart: Plan C 2.6 Checklist: Exam Room Preparation 2.7 Checklist: Client-Centered Approach 2.8 Fact Sheet: Copper IUD Aftercare 2.9 Side Effects Management: Steps in the Delivery of Care 2.10 Side Effects Management: Messages, Assessment & Treatment SECTION 4: ADDITIONAL RESOURCES ● Client Education Material: F.A.Q.’s Page 40 ● Client Education Material: EC Chart Page 42 SECTION 5: REFERENCES Page 44 OVERVIEW Introduction The New York State Center of Excellence for Family Planning and Reproductive Health Services (NYS COE) developed this toolkit to support agencies that receive Title X family planning funding through the New York State Department of Health (NYS DOH) Comprehensive Family Planning and Reproductive Health Care Services Program – as well as other sexual and reproductive health service providers – to implement Plan C: Copper IUD as Emergency Contraception (Plan C). -

F.8 Ethinylestradiol-Etonogestrel.Pdf
General Items 1. Summary statement of the proposal for inclusion, change or deletion. Here within, please find the evidence to support the inclusion Ethinylestradiol/Etonogestrel Vaginal Ring in the World Health Organization’s Essential Medicines List (EML). Unintended pregnancy is regarded as a serious public health issue both in developed and developing countries and has received growing research and policy attention during last few decades (1). It is a major global concern due to its association with adverse physical, mental, social and economic outcomes. Developing countries account for approximately 99% of the global maternal deaths in 2015, with sub-Saharan Africa alone accounting for roughly 66% (2). Even though the incidence of unintended pregnancy has declined globally in the past decade, the rate of unintended pregnancy remains high, particularly in developing regions. (3) Regarding the use of contraceptive vaginal rings, updated bibliography (4,5,6) states that contraceptive vaginal rings (CVR) offer an effective contraceptive option, expanding the available choices of hormonal contraception. Ethinylestradiol/Etonogestrel Vaginal Ring is a non-biodegradable, flexible, transparent with an outer diameter of 54 mm and a cross-sectional diameter of 4 mm. It contains 11.7 mg etonogestrel and 2.7 mg ethinyl estradiol. When placed in the vagina, each ring releases on average 0.120 mg/day of etonogestrel and 0.015 mg/day of ethinyl estradiol over a three-week period of use. Ethinylestradiol/Etonogestrel Vaginal Ring is intended for women of fertile age. The safety and efficacy have been established in women aged 18 to 40 years. The main advantages of CVRs are their effectiveness (similar or slightly better than the pill), ease of use without the need of remembering a daily routine, user ability to control initiation and discontinuation, nearly constant release rate allowing for lower doses, greater bioavailability and good cycle control with the combined ring, in comparison with oral contraceptives.