Modern Physics
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Discurso 03.Pdf
1 2 BUSCANDO LA MATERIA OSCURA DEL UNIVERSO EN FORMA DE PARTICULAS ELEMENTALES DEBILIES Depósito Legal: M-31269-2003 Imprime: Ibergráficas, S.A. Lope de Rueda, 11-13. 28009 Madrid 3 4 PREFACIO BUSCANDO LA MATERIA OSCURA Es un honor enorme ser nombrado Académico de Honor en la DEL UNIVERSO Academia de Ciencias e Ingenierías de Lanzarote, que ha nacido en el contexto del Centro Científico-Cultural Blas Cabrera. En particular, quiero dar las gracias EN FORMA DE PARTICULAS al Profesores Francisco González de Posada y Dominga Trujillo Jacinto del ELEMENTALES DEBILIES* Castillo por sus esfuerzos , ya que gracias a ellos tenemos desde hace muchos años este Centro en Arrecife. Así no nos olvidamos de las importantes obras científicas de mi abuelo Blas Cabrera Felipe y de la tradición científica en Lanzarote, Canarias y España. También quiero dar las gracias a mi tío José Cabrera Ramírez por ser el único que mantiene contacto con los que estamos a mucha distancia física, pero que nos acordamos de nuestra familia y de nuestros antepasados. Discurso leído en el acto de su recepción como Académico de Honor por el Prof. Dr. D. Blas Cabrera Navarro el 7 de julio de 2003 Arrecife (Lanzarote), Centro Científico-cultural Blas Cabrera Fig 1: Sixth Solvay Conference in 1930 held in Munich. * Based on invited talk and honorary membership in the newly formed “Academia de Ciencias e Ingenierias de Lanzarote” of at opening of “VII Cursos Universitarios de Verano en Canarias, Lanzatorte 2003” held at the “Centro Cientifico-Cultural – Blas Cabrera” in Arrecife, Lanzarote in the Ustedes me perdonaran las faltas de gramática y de pronunciación, Canary Islands, Spain, July 7, 2003. -

George De Hevesy in America
Journal of Nuclear Medicine, published on July 13, 2019 as doi:10.2967/jnumed.119.233254 George de Hevesy in America George de Hevesy was a Hungarian radiochemist who was awarded the Nobel Prize in Chemistry in 1943 for the discovery of the radiotracer principle (1). As the radiotracer principle is the foundation of all diagnostic and therapeutic nuclear medicine procedures, Hevesy is widely considered the father of nuclear medicine (1). Although it is well-known that he spent time at a number of European institutions, it is not widely known that he also spent six weeks at Cornell University in Ithaca, NY, in the fall of 1930 as that year’s Baker Lecturer in the Department of Chemistry (2-6). “[T]he Baker Lecturer gave two formal presentations per week, to a large and diverse audience and provided an informal seminar weekly for students and faculty members interested in the subject. The lecturer had an office in Baker Laboratory and was available to faculty and students for further discussion.” (7) There is also evidence that, “…Hevesy visited Harvard [University, Cambridge, MA] as a Baker Lecturer at Cornell in 1930…” (8). Neither of the authors of this Note/Letter was aware of Hevesy’s association with Cornell University despite our longstanding ties to Cornell until one of us (WCK) noticed the association in Hevesy’s biographical page on the official Nobel website (6). WCK obtained both his undergraduate degree and medical degree from Cornell in Ithaca and New York City, respectively, and spent his career in nuclear medicine. JRO did his nuclear medicine training at Columbia University and has subsequently been a faculty member of Weill Cornell Medical College for the last eleven years (with a brief tenure at Memorial Sloan Kettering Cancer Center (affiliated with Cornell)), and is now the program director of the Nuclear Medicine residency and Chief of the Molecular Imaging and Therapeutics Section. -

A Brief History of Nuclear Astrophysics
A BRIEF HISTORY OF NUCLEAR ASTROPHYSICS PART I THE ENERGY OF THE SUN AND STARS Nikos Prantzos Institut d’Astrophysique de Paris Stellar Origin of Energy the Elements Nuclear Astrophysics Astronomy Nuclear Physics Thermodynamics: the energy of the Sun and the age of the Earth 1847 : Robert Julius von Mayer Sun heated by fall of meteors 1854 : Hermann von Helmholtz Gravitational energy of Kant’s contracting protosolar nebula of gas and dust turns into kinetic energy Timescale ~ EGrav/LSun ~ 30 My 1850s : William Thompson (Lord Kelvin) Sun heated at formation from meteorite fall, now « an incadescent liquid mass » cooling Age 10 – 100 My 1859: Charles Darwin Origin of species : Rate of erosion of the Weald valley is 1 inch/century or 22 miles wild (X 1100 feet high) in 300 My Such large Earth ages also required by geologists, like Charles Lyell A gaseous, contracting and heating Sun 푀⊙ Mean solar density : ~1.35 g/cc Sun liquid Incompressible = 4 3 푅 3 ⊙ 1870s: J. Homer Lane ; 1880s :August Ritter : Sun gaseous Compressible As it shrinks, it releases gravitational energy AND it gets hotter Earth Mayer – Kelvin - Helmholtz Helmholtz - Ritter A gaseous, contracting and heating Sun 푀⊙ Mean solar density : ~1.35 g/cc Sun liquid Incompressible = 4 3 푅 3 ⊙ 1870s: J. Homer Lane ; 1880s :August Ritter : Sun gaseous Compressible As it shrinks, it releases gravitational energy AND it gets hotter Earth Mayer – Kelvin - Helmholtz Helmholtz - Ritter A gaseous, contracting and heating Sun 푀⊙ Mean solar density : ~1.35 g/cc Sun liquid Incompressible = 4 3 푅 3 ⊙ 1870s: J. -

Pauling-Linus.Pdf
NATIONAL ACADEMY OF SCIENCES L I N U S C A R L P A U L I N G 1901—1994 A Biographical Memoir by J A C K D. D UNITZ Any opinions expressed in this memoir are those of the author(s) and do not necessarily reflect the views of the National Academy of Sciences. Biographical Memoir COPYRIGHT 1997 NATIONAL ACADEMIES PRESS WASHINGTON D.C. LINUS CARL PAULING February 28, 1901–August 19, 1994 BY JACK D. DUNITZ INUS CARL PAULING was born in Portland, Oregon, on LFebruary 28, 1901, and died at his ranch at Big Sur, California, on August 19, 1994. In 1922 he married Ava Helen Miller (died 1981), who bore him four children: Linus Carl, Peter Jeffress, Linda Helen (Kamb), and Edward Crellin. Pauling is widely considered the greatest chemist of this century. Most scientists create a niche for themselves, an area where they feel secure, but Pauling had an enormously wide range of scientific interests: quantum mechanics, crys- tallography, mineralogy, structural chemistry, anesthesia, immunology, medicine, evolution. In all these fields and especially in the border regions between them, he saw where the problems lay, and, backed by his speedy assimilation of the essential facts and by his prodigious memory, he made distinctive and decisive contributions. He is best known, perhaps, for his insights into chemical bonding, for the discovery of the principal elements of protein secondary structure, the alpha-helix and the beta-sheet, and for the first identification of a molecular disease (sickle-cell ane- mia), but there are a multitude of other important contri- This biographical memoir was prepared for publication by both The Royal Society of London and the National Academy of Sciences of the United States of America. -

Europe's Biggest General Science Conference Concludes Successfully
SCIENCEScience PagesPAGES Special Report - ESOF 2016 Europe’s biggest generalSpecial Report science conference concludes successfully ESOF 2016, Europe's biggest general science conference concludes successfully in Manchester,in Manchester,UK UK Theme: Science as Revolution Theme: Science as Revolution - Veena Patwardhan rom 23rd to 27th July, 2016, FManchester flaunted its City of Science status as the host city of the seventh edition of EuroScience Open Forum (ESOF 2016). A bi- ennial event held in a different European city every two years, this time it was Manchester's turn to host this globally reputed science conference. Around 4500 delegates – scien- tists, innovators, academics, young researchers, journalists, policy makers, industry representatives and others – converged on the world's first industrial city to dis- cover and have discussions about the latest advancements in scien- rd th Manchester Central, venue of ESOF 2016 tific and technological researchFrom 23 to 27 July, 2016, Manchester flaunted its City of Science status as the host city of the across Europe and beyond. The seventhmain theme edition this of EuroScienceyear Laureates Open Forumand distinguished (ESOF 2016). Ascientists biennial inevent the held packed in a different was 'Science as Revolution', indicatingEuropean that city the every focus two of years,Exchange this time Hall it wasof Manchester Manchester's Central, turn to hostthe venuethis globally of the reputed the conference would be on how sciencescience andconference. technology conference. could transform life on the planet, revolutionise econo- The proceedings began with a string quartet render- mies, and help in overcoming challenges faced by global ing a piece of specially composed music. -

The Nobel Laureate George De Hevesy (1885-1966) - Universal Genius and Father of Nuclear Medicine Niese S* Am Silberblick 9, 01723 Wilsdruff, Germany
Open Access SAJ Biotechnology LETTER ISSN: 2375-6713 The Nobel Laureate George de Hevesy (1885-1966) - Universal Genius and Father of Nuclear Medicine Niese S* Am Silberblick 9, 01723 Wilsdruff, Germany *Corresponding author: Niese S, Am Silberblick 9, 01723 Wilsdruff, Germany, Tel: +49 35209 22849, E-mail: [email protected] Citation: Niese S, The Nobel Laureate George de Hevesy (1885-1966) - Universal Genius and Father of Nuclear Medicine. SAJ Biotechnol 5: 102 Article history: Received: 20 March 2018, Accepted: 29 March 2018, Published: 03 April 2018 Abstract The scientific work of the universal genius the Nobel Laureate George de Hevesy who has discovered and developed news in physics, chemistry, geology, biology and medicine is described. Special attention is given to his work in life science which he had done in the second half of his scientific career and was the base of the development of nuclear medicine. Keywords: George de Hevesy; Radionuclides; Nuclear Medicine Introduction George de Hevesy has founded Radioanalytical Chemistry and Nuclear Medicine, discovered the element hafnium and first separated stable isotopes. He was an inventor in many disciplines and his interest was not only focused on the development and refinement of methods, but also on the structure of matter and its changes: atoms, molecules, cells, organs, plants, animals, men and cosmic objects. He was working under complicated political situation in Europe in the 20th century. During his stay in Germany, Austria, Hungary, Switzerland, Denmark, and Sweden he wrote a lot papers in German. In 1962 he edited a large part of his articles in a collection where German papers are translated in English [1]. -

April 17-19, 2018 the 2018 Franklin Institute Laureates the 2018 Franklin Institute AWARDS CONVOCATION APRIL 17–19, 2018
april 17-19, 2018 The 2018 Franklin Institute Laureates The 2018 Franklin Institute AWARDS CONVOCATION APRIL 17–19, 2018 Welcome to The Franklin Institute Awards, the a range of disciplines. The week culminates in a grand United States’ oldest comprehensive science and medaling ceremony, befitting the distinction of this technology awards program. Each year, the Institute historic awards program. celebrates extraordinary people who are shaping our In this convocation book, you will find a schedule of world through their groundbreaking achievements these events and biographies of our 2018 laureates. in science, engineering, and business. They stand as We invite you to read about each one and to attend modern-day exemplars of our namesake, Benjamin the events to learn even more. Unless noted otherwise, Franklin, whose impact as a statesman, scientist, all events are free, open to the public, and located in inventor, and humanitarian remains unmatched Philadelphia, Pennsylvania. in American history. Along with our laureates, we celebrate his legacy, which has fueled the Institute’s We hope this year’s remarkable class of laureates mission since its inception in 1824. sparks your curiosity as much as they have ours. We look forward to seeing you during The Franklin From sparking a gene editing revolution to saving Institute Awards Week. a technology giant, from making strides toward a unified theory to discovering the flow in everything, from finding clues to climate change deep in our forests to seeing the future in a terahertz wave, and from enabling us to unplug to connecting us with the III world, this year’s Franklin Institute laureates personify the trailblazing spirit so crucial to our future with its many challenges and opportunities. -
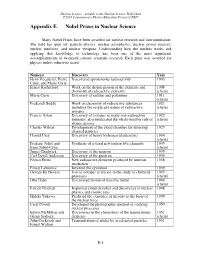
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

ARIE SKLODOWSKA CURIE Opened up the Science of Radioactivity
ARIE SKLODOWSKA CURIE opened up the science of radioactivity. She is best known as the discoverer of the radioactive elements polonium and radium and as the first person to win two Nobel prizes. For scientists and the public, her radium was a key to a basic change in our understanding of matter and energy. Her work not only influenced the development of fundamental science but also ushered in a new era in medical research and treatment. This file contains most of the text of the Web exhibit “Marie Curie and the Science of Radioactivity” at http://www.aip.org/history/curie/contents.htm. You must visit the Web exhibit to explore hyperlinks within the exhibit and to other exhibits. Material in this document is copyright © American Institute of Physics and Naomi Pasachoff and is based on the book Marie Curie and the Science of Radioactivity by Naomi Pasachoff, Oxford University Press, copyright © 1996 by Naomi Pasachoff. Site created 2000, revised May 2005 http://www.aip.org/history/curie/contents.htm Page 1 of 79 Table of Contents Polish Girlhood (1867-1891) 3 Nation and Family 3 The Floating University 6 The Governess 6 The Periodic Table of Elements 10 Dmitri Ivanovich Mendeleev (1834-1907) 10 Elements and Their Properties 10 Classifying the Elements 12 A Student in Paris (1891-1897) 13 Years of Study 13 Love and Marriage 15 Working Wife and Mother 18 Work and Family 20 Pierre Curie (1859-1906) 21 Radioactivity: The Unstable Nucleus and its Uses 23 Uses of Radioactivity 25 Radium and Radioactivity 26 On a New, Strongly Radio-active Substance -

2014 Chemistry Newsletter
FALL 2014 Welcome from the Head Construction Begins on Greetings from the Department of Chemistry! This has been Science Learning Center another successful year for UGA Chemistry, and I am pleased to report more good news about our department, faculty and students. University enrollment continues to grow each year – this fall, the university enrolled 35,197 students. The increase in student numbers, particularly in the rapidly growing engineering program, has created significant extra demand for Chemistry courses. This growth in instructional demand will help us to make a case for the continued growth of our faculty numbers. As I have mentioned in the past, faculty recruiting is one of the most significant and satisfying parts of my job. Jon Amster In the last year, we were able to recruit a new organic faculty member. Prof. Eric Ferreira comes to us from Colorado State University, where he established a successful and well-funded research program in synthetic organic chemistry. Eric and four Rendering of the future Science Learning Center of his graduate students moved to Athens over the summer. While his laboratory renovations are taking place, his students are hard at work in temporary space, and his program has hit he long-awaited Science Learning Center (SLC) the ground running. You can read more about Eric and his research activities inside this has finally become a reality, with groundbreaking issue of the newsletter. We have also just completed the recruitment of an organic lecturer, ceremonies attended by Governor Nathan Deal Doug Jackson. Doug is a product of our department, where he is completing his Ph.D. -

A Brief History of Mangnetism
A Brief history of mangnetism Navinder Singh∗ and Arun M. Jayannavar∗∗ ∗Physical Research Laboratory, Ahmedabad, India, Pin: 380009. ∗∗ IOP, Bhubaneswar, India. ∗† Abstract In this article an overview of the historical development of the key ideas in the field of magnetism is presented. The presentation is semi-technical in nature.Starting by noting down important contribution of Greeks, William Gilbert, Coulomb, Poisson, Oersted, Ampere, Faraday, Maxwell, and Pierre Curie, we review early 20th century investigations by Paul Langevin and Pierre Weiss. The Langevin theory of paramagnetism and the Weiss theory of ferromagnetism were partly successful and real understanding of magnetism came with the advent of quantum mechanics. Van Vleck was the pioneer in applying quantum mechanics to the problem of magnetism and we discuss his main contributions: (1) his detailed quantum statistical mechanical study of magnetism of real gases; (2) his pointing out the importance of the crystal fields or ligand fields in the magnetic behavior of iron group salts (the ligand field theory); and (3) his many contributions to the elucidation of exchange interactions in d electron metals. Next, the pioneering contributions (but lesser known) of Dorfman are discussed. Then, in chronological order, the key contributions of Pauli, Heisenberg, and Landau are presented. Finally, we discuss a modern topic of quantum spin liquids. 1 Prologue In this presentation, the development of the conceptual structure of the field is highlighted, and the historical context is somewhat limited in scope. In that way, the presentation is not historically very rigorous. However, it may be useful for gaining a ”bird’s-eye view” of the vast field of magnetism. -

1972 Frã‰Dã‰Ric and Irãˆne Joliot-Curie
DISTINGUISHED NUCLEAR PIONEERS—1972 FRÉDÉRIC AND IRÈNE JOLIOT-CURIE The choice of Frédéricand IrèneJoliot-Curie as there under the guidance of his principal teacher, the Distinguished Nuclear Pioneers for 1972 by the So great physicist Paul Langevin, who was the first to ciety of Nuclear Medicine is one of the best that recognize Joliot's exceptional gifts and who had a could be made because their discovery of artificial decisive influence on him, not only in the field of radioactivity was not only a great step forward in science but also in leading him toward socialism and the development of nuclear physics but has also led pacifism. Joliot graduated first in his class, and after directly to the possibility of obtaining radioactive 15 months of military service, he might have started isotopes of practically all the chemical elements— a brilliant career as an engineer in industry. But fol those radioisotopes which are now so widely used lowing the advice of Paul Langevin he became a in medical research or practice, and without which personal assistant to Marie Curie who paid him on nuclear medicine could not exist. a grant from the Rockefeller Foundation. FrédéricJoliot was born in Paris on March 19, Thus Frederic Joliot started his scientific career 1900. His father, Henri Joliot, having participated in in the spring of 1925 at the Institut du Radium of the uprising of the Commune of Paris in 1871, had the University of Paris under the guidance of Marie been obliged to spend several years in Belgium.