A Cortical Immune Network Map Identifies a Subset of Human Microglia Involved in Tau Pathology
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genome-Wide DNA Methylation Analysis of KRAS Mutant Cell Lines Ben Yi Tew1,5, Joel K
www.nature.com/scientificreports OPEN Genome-wide DNA methylation analysis of KRAS mutant cell lines Ben Yi Tew1,5, Joel K. Durand2,5, Kirsten L. Bryant2, Tikvah K. Hayes2, Sen Peng3, Nhan L. Tran4, Gerald C. Gooden1, David N. Buckley1, Channing J. Der2, Albert S. Baldwin2 ✉ & Bodour Salhia1 ✉ Oncogenic RAS mutations are associated with DNA methylation changes that alter gene expression to drive cancer. Recent studies suggest that DNA methylation changes may be stochastic in nature, while other groups propose distinct signaling pathways responsible for aberrant methylation. Better understanding of DNA methylation events associated with oncogenic KRAS expression could enhance therapeutic approaches. Here we analyzed the basal CpG methylation of 11 KRAS-mutant and dependent pancreatic cancer cell lines and observed strikingly similar methylation patterns. KRAS knockdown resulted in unique methylation changes with limited overlap between each cell line. In KRAS-mutant Pa16C pancreatic cancer cells, while KRAS knockdown resulted in over 8,000 diferentially methylated (DM) CpGs, treatment with the ERK1/2-selective inhibitor SCH772984 showed less than 40 DM CpGs, suggesting that ERK is not a broadly active driver of KRAS-associated DNA methylation. KRAS G12V overexpression in an isogenic lung model reveals >50,600 DM CpGs compared to non-transformed controls. In lung and pancreatic cells, gene ontology analyses of DM promoters show an enrichment for genes involved in diferentiation and development. Taken all together, KRAS-mediated DNA methylation are stochastic and independent of canonical downstream efector signaling. These epigenetically altered genes associated with KRAS expression could represent potential therapeutic targets in KRAS-driven cancer. Activating KRAS mutations can be found in nearly 25 percent of all cancers1. -

Análise Integrativa De Perfis Transcricionais De Pacientes Com
UNIVERSIDADE DE SÃO PAULO FACULDADE DE MEDICINA DE RIBEIRÃO PRETO PROGRAMA DE PÓS-GRADUAÇÃO EM GENÉTICA ADRIANE FEIJÓ EVANGELISTA Análise integrativa de perfis transcricionais de pacientes com diabetes mellitus tipo 1, tipo 2 e gestacional, comparando-os com manifestações demográficas, clínicas, laboratoriais, fisiopatológicas e terapêuticas Ribeirão Preto – 2012 ADRIANE FEIJÓ EVANGELISTA Análise integrativa de perfis transcricionais de pacientes com diabetes mellitus tipo 1, tipo 2 e gestacional, comparando-os com manifestações demográficas, clínicas, laboratoriais, fisiopatológicas e terapêuticas Tese apresentada à Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo para obtenção do título de Doutor em Ciências. Área de Concentração: Genética Orientador: Prof. Dr. Eduardo Antonio Donadi Co-orientador: Prof. Dr. Geraldo A. S. Passos Ribeirão Preto – 2012 AUTORIZO A REPRODUÇÃO E DIVULGAÇÃO TOTAL OU PARCIAL DESTE TRABALHO, POR QUALQUER MEIO CONVENCIONAL OU ELETRÔNICO, PARA FINS DE ESTUDO E PESQUISA, DESDE QUE CITADA A FONTE. FICHA CATALOGRÁFICA Evangelista, Adriane Feijó Análise integrativa de perfis transcricionais de pacientes com diabetes mellitus tipo 1, tipo 2 e gestacional, comparando-os com manifestações demográficas, clínicas, laboratoriais, fisiopatológicas e terapêuticas. Ribeirão Preto, 2012 192p. Tese de Doutorado apresentada à Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo. Área de Concentração: Genética. Orientador: Donadi, Eduardo Antonio Co-orientador: Passos, Geraldo A. 1. Expressão gênica – microarrays 2. Análise bioinformática por module maps 3. Diabetes mellitus tipo 1 4. Diabetes mellitus tipo 2 5. Diabetes mellitus gestacional FOLHA DE APROVAÇÃO ADRIANE FEIJÓ EVANGELISTA Análise integrativa de perfis transcricionais de pacientes com diabetes mellitus tipo 1, tipo 2 e gestacional, comparando-os com manifestações demográficas, clínicas, laboratoriais, fisiopatológicas e terapêuticas. -

Role of RUNX1 in Aberrant Retinal Angiogenesis Jonathan D
Page 1 of 25 Diabetes Identification of RUNX1 as a mediator of aberrant retinal angiogenesis Short Title: Role of RUNX1 in aberrant retinal angiogenesis Jonathan D. Lam,†1 Daniel J. Oh,†1 Lindsay L. Wong,1 Dhanesh Amarnani,1 Cindy Park- Windhol,1 Angie V. Sanchez,1 Jonathan Cardona-Velez,1,2 Declan McGuone,3 Anat O. Stemmer- Rachamimov,3 Dean Eliott,4 Diane R. Bielenberg,5 Tave van Zyl,4 Lishuang Shen,1 Xiaowu Gai,6 Patricia A. D’Amore*,1,7 Leo A. Kim*,1,4 Joseph F. Arboleda-Velasquez*1 Author affiliations: 1Schepens Eye Research Institute/Massachusetts Eye and Ear, Department of Ophthalmology, Harvard Medical School, 20 Staniford St., Boston, MA 02114 2Universidad Pontificia Bolivariana, Medellin, Colombia, #68- a, Cq. 1 #68305, Medellín, Antioquia, Colombia 3C.S. Kubik Laboratory for Neuropathology, Massachusetts General Hospital, 55 Fruit St., Boston, MA 02114 4Retina Service, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, 243 Charles St., Boston, MA 02114 5Vascular Biology Program, Boston Children’s Hospital, Department of Surgery, Harvard Medical School, 300 Longwood Ave., Boston, MA 02115 6Center for Personalized Medicine, Children’s Hospital Los Angeles, Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027, USA 7Department of Pathology, Harvard Medical School, 25 Shattuck St., Boston, MA 02115 Corresponding authors: Joseph F. Arboleda-Velasquez: [email protected] Ph: (617) 912-2517 Leo Kim: [email protected] Ph: (617) 912-2562 Patricia D’Amore: [email protected] Ph: (617) 912-2559 Fax: (617) 912-0128 20 Staniford St. Boston MA, 02114 † These authors contributed equally to this manuscript Word Count: 1905 Tables and Figures: 4 Diabetes Publish Ahead of Print, published online April 11, 2017 Diabetes Page 2 of 25 Abstract Proliferative diabetic retinopathy (PDR) is a common cause of blindness in the developed world’s working adult population, and affects those with type 1 and type 2 diabetes mellitus. -

Identification of Potential Key Genes and Pathway Linked with Sporadic Creutzfeldt-Jakob Disease Based on Integrated Bioinformatics Analyses
medRxiv preprint doi: https://doi.org/10.1101/2020.12.21.20248688; this version posted December 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. Identification of potential key genes and pathway linked with sporadic Creutzfeldt-Jakob disease based on integrated bioinformatics analyses Basavaraj Vastrad1, Chanabasayya Vastrad*2 , Iranna Kotturshetti 1. Department of Biochemistry, Basaveshwar College of Pharmacy, Gadag, Karnataka 582103, India. 2. Biostatistics and Bioinformatics, Chanabasava Nilaya, Bharthinagar, Dharwad 580001, Karanataka, India. 3. Department of Ayurveda, Rajiv Gandhi Education Society`s Ayurvedic Medical College, Ron, Karnataka 562209, India. * Chanabasayya Vastrad [email protected] Ph: +919480073398 Chanabasava Nilaya, Bharthinagar, Dharwad 580001 , Karanataka, India NOTE: This preprint reports new research that has not been certified by peer review and should not be used to guide clinical practice. medRxiv preprint doi: https://doi.org/10.1101/2020.12.21.20248688; this version posted December 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. Abstract Sporadic Creutzfeldt-Jakob disease (sCJD) is neurodegenerative disease also called prion disease linked with poor prognosis. The aim of the current study was to illuminate the underlying molecular mechanisms of sCJD. The mRNA microarray dataset GSE124571 was downloaded from the Gene Expression Omnibus database. Differentially expressed genes (DEGs) were screened. -
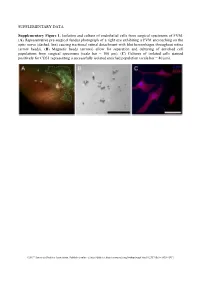
Supplementary Figures and Tables
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

MOCHI Enables Discovery of Heterogeneous Interactome Modules in 3D Nucleome
Downloaded from genome.cshlp.org on October 4, 2021 - Published by Cold Spring Harbor Laboratory Press MOCHI enables discovery of heterogeneous interactome modules in 3D nucleome Dechao Tian1,# , Ruochi Zhang1,# , Yang Zhang1, Xiaopeng Zhu1, and Jian Ma1,* 1Computational Biology Department, School of Computer Science, Carnegie Mellon University, Pittsburgh, PA 15213, USA #These two authors contributed equally *Correspondence: [email protected] Contact To whom correspondence should be addressed: Jian Ma School of Computer Science Carnegie Mellon University 7705 Gates-Hillman Complex 5000 Forbes Avenue Pittsburgh, PA 15213 Phone: +1 (412) 268-2776 Email: [email protected] 1 Downloaded from genome.cshlp.org on October 4, 2021 - Published by Cold Spring Harbor Laboratory Press Abstract The composition of the cell nucleus is highly heterogeneous, with different constituents forming complex interactomes. However, the global patterns of these interwoven heterogeneous interactomes remain poorly understood. Here we focus on two different interactomes, chromatin interaction network and gene regulatory network, as a proof-of-principle, to identify heterogeneous interactome modules (HIMs), each of which represents a cluster of gene loci that are in spatial contact more frequently than expected and that are regulated by the same group of transcription factors. HIM integrates transcription factor binding and 3D genome structure to reflect “transcriptional niche” in the nucleus. We develop a new algorithm MOCHI to facilitate the discovery of HIMs based on network motif clustering in heterogeneous interactomes. By applying MOCHI to five different cell types, we found that HIMs have strong spatial preference within the nucleus and exhibit distinct functional properties. Through integrative analysis, this work demonstrates the utility of MOCHI to identify HIMs, which may provide new perspectives on the interplay between transcriptional regulation and 3D genome organization. -

A Tool to Identify Coordinately Expressed Genes
The CO-Regulation Database (CORD): A Tool to Identify Coordinately Expressed Genes John P. Fahrenbach1*, Jorge Andrade2, Elizabeth M. McNally1,3 1 Department of Medicine, The University of Chicago, Chicago, Illinois, United States of America, 2 Center for Research Informatics, The University of Chicago, Chicago, Illinois, United States of America, 3 Department of Human Genetics, The University of Chicago, Chicago, Illinois, United States of America Abstract Background: Meta-analysis of gene expression array databases has the potential to reveal information about gene function. The identification of gene-gene interactions may be inferred from gene expression information but such meta-analysis is often limited to a single microarray platform. To address this limitation, we developed a gene-centered approach to analyze differential expression across thousands of gene expression experiments and created the CO-Regulation Database (CORD) to determine which genes are correlated with a queried gene. Results: Using the GEO and ArrayExpress database, we analyzed over 120,000 group by group experiments from gene microarrays to determine the correlating genes for over 30,000 different genes or hypothesized genes. CORD output data is presented for sample queries with focus on genes with well-known interaction networks including p16 (CDKN2A), vimentin (VIM), MyoD (MYOD1). CDKN2A, VIM, and MYOD1 all displayed gene correlations consistent with known interacting genes. Conclusions: We developed a facile, web-enabled program to determine gene-gene correlations across different gene expression microarray platforms. Using well-characterized genes, we illustrate how CORD’s identification of co-expressed genes contributes to a better understanding a gene’s potential function. The website is found at http://cord-db.org. -

Characterization and Expression Profile of CMTM3/CKLFSF3
Journal of Biochemistry and Molecular Biology, Vol. 39, No. 5, September 2006, pp. 537-545 Characterization and Expression Profile of CMTM3/CKLFSF3 Ji Zhong, Yu Wang, Xiaoyan Qiu, Xiaoning Mo, Yanan Liu, Ting Li, Quansheng Song, Dalong Ma and Wenling Han* Peking University Center for Human Disease Genomics, 38 Xueyuan Road, Beijing 100083, China Received 22 February 2006, Accepted 10 May 2006 CMTM/CKLFSF is a novel family of proteins linking Introduction chemokines and TM4SF. In humans, these proteins are encoded by nine genes, CKLF and CMTM1-8/CKLFSF1-8. CKLFSF is a novel family of proteins linking chemokines and Here we report the characteristics and expression profile Transmembrane 4 Super Family (TM4SF), which are encoded of CMTM3/CKLFSF3. Human CMTM3/CKLFSF3 has a by nine genes, CKLF and CKLFSF1-8, in humans. CKLF, high sequence identity among various species and similar CKLFSF1, and CKLFSF2 are active during evolution, and characteristics as its mouse and rat homologues. Established CKLFSF2 has two orthologues in rat and mouse designated by results both of RT-PCR and Quantitative Real-time Cklfsf2a and Cklfsf2b. CKLFSF3-8 are evolutionarily conserved. PCR, the gene is highly transcribed in testis, leukocytes CKLFSF members form two gene clusters: CKLF and and spleen. For further verification, we generated a CKLFSF1-4 at chromosome 16q22.1, and CKLFSF6-8 at polyclonal antibody against human CMTM3/CKLFSF3 chromosome 3p23. Most CKLFSFs have different spliced and found that the protein is highly expressed in the testis forms, and the protein product of at least one spliced form of and some cells of PBMCs. -

GSKB: a Gene Set Database for Pathway Analysis in Mouse
bioRxiv preprint doi: https://doi.org/10.1101/082511; this version posted October 24, 2016. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. GSKB: A gene set database for pathway analysis in mouse Liming Lai1, Jason Hennessey1, Valerie Bares1, Eun Woo Son1, Yuguang Ban1, Wei Wang1, Jianli Qi1, Gaixin Jiang1, Arthur Liberzon2, and Steven Xijin Ge1* 1Department of Mathematics and Statistics, South Dakota State University, Brookings, SD 57007, USA 2Broad Institute of MIT and Harvard, 415 Main Street, Cambridge, MA 02142, USA * Corresponding author: Tel: +1-605-688-5845; Email: [email protected] ABSTRACT: Interpretation of high-throughput genomics data based on biological pathways constitutes a constant challenge, partly because of the lack of supporting pathway database. In this study, we created a functional genomics knowledgebase in mouse, which includes 33,261 pathways and gene sets compiled from 40 sources such as Gene Ontology, KEGG, GeneSetDB, PANTHER, microRNA and transcription factor target genes, etc. In addition, we also manually collected and curated 8,747 lists of differentially expressed genes from 2,526 published gene expression studies to enable the detection of similarity to previously reported gene expression signatures. These two types of data constitute a Gene Set Knowledgebase (GSKB), which can be readily used by various pathway analysis software such as gene set enrichment analysis (GSEA). Using our knowledgebase, we were able to detect the correct microRNA (miR-29) pathway that was suppressed using antisense oligonucleotides and confirmed its role in inhibiting fibrogenesis, which might involve upregulation of transcription factor SMAD3. -

High-Risk Oral Leukoplakia Is Associated with Aberrant Promoter
Abe et al. BMC Cancer (2016) 16:350 DOI 10.1186/s12885-016-2371-5 RESEARCHARTICLE Open Access High-risk oral leukoplakia is associated with aberrant promoter methylation of multiple genes Masanobu Abe1,2*, Satoshi Yamashita3, Yoshiyuki Mori4, Takahiro Abe1, Hideto Saijo1, Kazuto Hoshi1, Toshikazu Ushijima3 and Tsuyoshi Takato1 Abstract Background: Early detection of oral squamous cell carcinomas (OSCCs) is urgently needed to improve the prognosis and quality of life (QOL) of patients. Oral leukoplakias (OLs), known as the most common premalignant lesions in the oral cavity, often precede OSCCs. Especially, OLs with dysplasia are known to have a high risk of malignant transformation. Here, we searched for the promoter methylation characteristic of high-risk OLs. Methods: To identify methylation-silenced genes, a combined analysis of methylated DNA immunoprecipitation (MeDIP) − CpG island (CGI) microarray analysis and expression microarray analysis after treatment with a demethylating agent was performed in two OSCC cell lines (Ca9–22 and HSC-2). The methylation statuses of each gene were examined by methylation-specific PCR. Results: A total of 52 genes were identified as candidates for methylation-silenced genes in Ca9-22 or HSC-2. The promoter regions of 13 genes among the 15 genes randomly selected for further analysis were confirmed to be methylated in one or more of five cell lines. In OSCC tissues (n = 26), 8 of the 13 genes, TSPYL5, EGFLAM, CLDN11, NKX2-3, RBP4, CMTM3, TRPC4, and MAP6, were methylated. In OL tissues (n = 24), seven of the eight genes, except for EGFLAM, were found to be methylated in their promoter regions. -

Supplemental Data
Supplementary Table 1. Gene sets from Figure 6. Lists of genes from each individual gene set defined in Figure 6, including the fold-change in expression of each gene in treatment group pair-wise comparisons. ENSEMBL: Ensembl gene identifier; Symbol: official gene symbol; logFC: log fold change; p value: significance of fold-change in a pair-wise comparison, P<0.05 cut-off; FDR: false discovery rate, expected proportion of false positives among the differentially expressed genes in a pair-wise comparison (FDR<0.25 cut-off). Sup. Table 1 SET I CP versus Sal CP versus CP+DCA DCA versus Sal ENSEMBL Symbol logFC PValue FDR logFC PValue FDR logFC PValue FDR Desc ENSMUSG00000020326 Ccng1 2.64 0.00 0.00 -0.06 0.13 0.96 0.40 0.00 0.23 cyclin G1 [Source:MGI Symbol;Acc:MGI:102890] ENSMUSG00000031886 Ces2e 3.97 0.00 0.00 -0.24 0.02 0.28 0.01 1.00 1.00 carboxylesterase 2E [Source:MGI Symbol;Acc:MGI:2443170] ENSMUSG00000041959 S100a10 2.31 0.00 0.00 -0.21 0.02 0.23 -0.11 0.53 1.00 S100 calcium binding protein A10 (calpactin) [Source:MGI Symbol;Acc:MGI:1339468] ENSMUSG00000092341 Malat1 1.09 0.00 0.00 -0.11 0.20 1.00 0.66 0.00 0.00 metastasis associated lung adenocarcinoma transcript 1 (non-coding RNA) [Source:MGI Symbol;Acc:MGI:1919539] ENSMUSG00000072949 Acot1 1.73 0.00 0.00 -0.22 0.01 0.12 -0.44 0.01 1.00 acyl-CoA thioesterase 1 [Source:MGI Symbol;Acc:MGI:1349396] ENSMUSG00000064339 mt-Rnr2 1.09 0.00 0.00 -0.08 0.17 1.00 0.67 0.00 0.07 mitochondrially encoded 16S rRNA [Source:MGI Symbol;Acc:MGI:102492] ENSMUSG00000025934 Gsta3 1.86 0.00 0.00 -0.28 -

Roles in Cancer Biological Processes and Potential Clinical Value
Cancer Biol Med 2020. doi: 10.20892/j.issn.2095-3941.2020.0032 REVIEW CMTM family proteins 1–8: roles in cancer biological processes and potential clinical value Jie Wu*, Lan Li*, Siyi Wu, Bin Xu Cancer Center, Renmin Hospital of Wuhan University, Wuhan 430060, China ABSTRACT The CKLF-like MARVEL transmembrane domain containing (CMTM) family of genes comprises CKLF and CMTM1–8 (previously called chemokine-like factor superfamily 1–8, CKLFSF1–8). The CMTM family proteins contain a structurally conserved MAL and related proteins for vesicle trafficking and membrane linking (MARVEL) domain. Dysregulated expression of multiple CMTM family members is a common feature in many human cancer types. CMTM proteins control critical biological processes in cancer development, including growth factor receptor activation and recycling, cell proliferation, apoptosis, metastasis, and immune evasion. Emerging in vivo and in vitro evidence indicates that the mechanisms of action of most CMTM proteins are complex and multifactorial. This review highlights new findings regarding the roles of CMTM1–8 in cancer, particularly in tumor growth, metastasis, and immune evasion. Additionally, the potential clinical value of CMTMs as novel drug targets or biomarkers is discussed. KEYWORDS CMTM family; cancer; cell cycle; EGFR; EMT; apoptosis; tumor immunity Introduction CMTM genes have been reported to be differentially expressed between tumor and normal tissue, thus suggesting Cancer is a highly heterogeneous group of diseases and one that CMTMs may actively regulate tumor development in of the leading causes of death worldwide. Each tumor type various cancer types4-10. The functions of CMTM family pro- exhibits distinct biological characteristics, clinical features, teins in tumor growth, metastasis, and antitumor immunity outcomes, and responses to therapies.