Ligand-Gated Ion Channels' British Journal of Pharmacology, Vol
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Release Β ATP-Induced IL-1 and Canonical Nicotinic Agonists Inhibit Phosphocholine-Modified Macromolecules
Phosphocholine-Modified Macromolecules and Canonical Nicotinic Agonists Inhibit ATP-Induced IL-1β Release This information is current as Andreas Hecker, Mira Küllmar, Sigrid Wilker, Katrin of September 24, 2021. Richter, Anna Zakrzewicz, Srebrena Atanasova, Verena Mathes, Thomas Timm, Sabrina Lerner, Jochen Klein, Andreas Kaufmann, Stefan Bauer, Winfried Padberg, Wolfgang Kummer, Sabina Janciauskiene, Martin Fronius, Elke K. H. Schweda, Günter Lochnit and Veronika Grau Downloaded from J Immunol 2015; 195:2325-2334; Prepublished online 22 July 2015; doi: 10.4049/jimmunol.1400974 http://www.jimmunol.org/content/195/5/2325 http://www.jimmunol.org/ Supplementary http://www.jimmunol.org/content/suppl/2015/07/22/jimmunol.140097 Material 4.DCSupplemental References This article cites 42 articles, 11 of which you can access for free at: http://www.jimmunol.org/content/195/5/2325.full#ref-list-1 by guest on September 24, 2021 Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2015 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. -
ANNNNNNNNNNNNNNNNNNNN 100A 006 Left Eye Input Right Eye Input
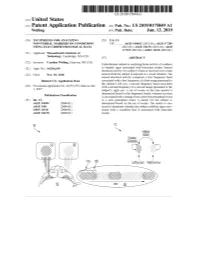
US 20190175049A1 ( 19) United States (12 ) Patent Application Publication (10 ) Pub. No. : US 2019 /0175049 A1 Welling ( 43 ) Pub . Date : Jun . 13 , 2019 ( 54 ) TECHNIQUES FOR ANALYZING (52 ) U . S . CI. NON -VERBAL MARKERS OF CONDITIONS CPC . .. A61B 5 /04842 (2013 . 01 ) ; A61B 5 / 7289 USING ELECTROPHYSIOLOGICAL DATA (2013 . 01) ; A61B 5 /0478 ( 2013 .01 ) ; A61B 5 /7225 ( 2013. 01 ) ; G06N 20 / 10 (2019 .01 ) (71 ) Applicant: Massachusetts Institute of Technology , Cambridge , MA (US ) ( 57 ) ABSTRACT (72 ) Inventor : Caroline Welling, Hanover, NH (US ) Embodiments related to analyzing brain activity of a subject to identify signs associated with binocular rivalry . Sensed ( 21 ) Appl. No. : 16 / 206, 639 electrical activity of a subject' s brain is received over a time period while the subject is exposed to a visual stimulus. The ( 22 ) Filed : Nov. 30 , 2018 sensed electrical activity comprises a first frequency band Related U . S . Application Data associated with a first frequency of a first image presented to the subject ' s left eye , a second frequency band associated (60 ) Provisional application No .62 / 593 , 535, filed on Dec . with a second frequency of a second image presented to the 1 , 2017 subject ' s right eye . A set of events in the time period is determined based on the frequency bands, wherein an event Publication Classification is associated with a change from a previous perceptual event (51 ) Int. Ci. to a new perceptual event. A metric for the subject is A61B 5 /0484 ( 2006 .01 ) determined based on the set of events . The metric is ana A61B 5 /00 ( 2006 .01 ) lyzed to determine whether the subject exhibits signs asso GO6N 20 / 10 (2006 .01 ) ciated with a condition that is associated with binocular A61B 5 /0478 ( 2006 .01 ) rivalry . -

Rabbit Anti-CHRNA10/FITC Conjugated Antibody-SL12111R-FITC
SunLong Biotech Co.,LTD Tel: 0086-571- 56623320 Fax:0086-571- 56623318 E-mail:[email protected] www.sunlongbiotech.com Rabbit Anti-CHRNA10/FITC Conjugated antibody SL12111R-FITC Product Name: Anti-CHRNA10/FITC Chinese Name: FITC标记的烟碱型乙酰胆碱受体α10/AChRα10抗体 Acetylcholine receptor, neuronal nicotinic, alpha-10 subunit; ACH10_HUMAN; Alpha 10 nAChR; Cholinergic receptor nicotinic alpha 10; Cholinergic receptor, neuronal nicotinic, alpha polypeptide 10; Cholinergic receptor, nicotinic, alpha polypeptide 10; Alias: CHRNA 10; CHRNA10; NACHR alpha 10; NACHR alpha-10; NACHRA10; Neuronal acetylcholine receptor protein subunit alpha 10; Neuronal acetylcholine receptor subunit alpha-10; Nicotinic acetylcholine receptor subunit alpha 10; Nicotinic acetylcholine receptor subunit alpha-10. Organism Species: Rabbit Clonality: Polyclonal React Species: Human,Mouse,Rat,Dog,Pig,Cow,Horse,Rabbit,Sheep, ICC=1:50-200IF=1:50-200 Applications: not yet tested in other applications. optimal dilutions/concentrations should be determined by the end user. Molecular weight: 47kDa Cellular localization: The cell membrane Form: Lyophilizedwww.sunlongbiotech.com or Liquid Concentration: 1mg/ml immunogen: KLH conjugated synthetic peptide derived from human CHRNA10 Lsotype: IgG Purification: affinity purified by Protein A Storage Buffer: 0.01M TBS(pH7.4) with 1% BSA, 0.03% Proclin300 and 50% Glycerol. Store at -20 °C for one year. Avoid repeated freeze/thaw cycles. The lyophilized antibody is stable at room temperature for at least one month and for greater than a year Storage: when kept at -20°C. When reconstituted in sterile pH 7.4 0.01M PBS or diluent of antibody the antibody is stable for at least two weeks at 2-4 °C. background: Product Detail: Members of the ligand-gated ion channel receptor family are characterized by their fast transmitting response to neurotransmitters. -

Effect of Repeated Gaboxadol Administration on Night Sleep and Next-Day Performance in Healthy Elderly Subjects
Neuropsychopharmacology (2005) 30, 833–841 & 2005 Nature Publishing Group All rights reserved 0893-133X/05 $30.00 www.neuropsychopharmacology.org Effect of Repeated Gaboxadol Administration on Night Sleep and Next-Day Performance in Healthy Elderly Subjects 1 2 ,3 1 Stefan Mathias , Josef Zihl , Axel Steiger* and Marike Lancel 1Section of Sleep Pharmacology, Max-Planck-Institute of Psychiatry, Munich, Germany; 2Section of Neuropsychology, Max-Planck-Institute of Psychiatry, Munich, Germany; 3Department of Psychiatry, Max-Planck-Institute of Psychiatry, Munich, Germany Aging is associated with dramatic reductions in sleep continuity and sleep intensity. Since gaboxadol, a selective GABAA receptor agonist, has been demonstrated to improve sleep consolidation and promote deep sleep, it may be an effective hypnotic, particularly for elderly patients with insomnia. In the present study, we investigated the effects of subchronic gaboxadol administration on nocturnal sleep and its residual effects during the next days in elderly subjects. This was a randomized, double-blind, placebo-controlled, balanced crossover study in 10 healthy elderly subjects without sleep complaints. The subjects were administered either placebo or 15 mg gaboxadol hydrochloride at bedtime on three consecutive nights. Sleep was recorded during each night from 2300 to 0700 h and tests assessing attention (target detection, stroop test) and memory function (visual form recognition, immediate word recall, digit span) were applied at 0900, 1400, and 1700 h during the following days. Compared with placebo, gaboxadol significantly shortened subjective sleep onset latency and increased self-rated sleep intensity and quality. Polysomnographic recordings showed that it significantly decreased the number of awakenings, the amount of intermittent wakefulness, and stage 1, and increased slow wave sleep and stage 2. -

GABA Receptors
D Reviews • BIOTREND Reviews • BIOTREND Reviews • BIOTREND Reviews • BIOTREND Reviews Review No.7 / 1-2011 GABA receptors Wolfgang Froestl , CNS & Chemistry Expert, AC Immune SA, PSE Building B - EPFL, CH-1015 Lausanne, Phone: +41 21 693 91 43, FAX: +41 21 693 91 20, E-mail: [email protected] GABA Activation of the GABA A receptor leads to an influx of chloride GABA ( -aminobutyric acid; Figure 1) is the most important and ions and to a hyperpolarization of the membrane. 16 subunits with γ most abundant inhibitory neurotransmitter in the mammalian molecular weights between 50 and 65 kD have been identified brain 1,2 , where it was first discovered in 1950 3-5 . It is a small achiral so far, 6 subunits, 3 subunits, 3 subunits, and the , , α β γ δ ε θ molecule with molecular weight of 103 g/mol and high water solu - and subunits 8,9 . π bility. At 25°C one gram of water can dissolve 1.3 grams of GABA. 2 Such a hydrophilic molecule (log P = -2.13, PSA = 63.3 Å ) cannot In the meantime all GABA A receptor binding sites have been eluci - cross the blood brain barrier. It is produced in the brain by decarb- dated in great detail. The GABA site is located at the interface oxylation of L-glutamic acid by the enzyme glutamic acid decarb- between and subunits. Benzodiazepines interact with subunit α β oxylase (GAD, EC 4.1.1.15). It is a neutral amino acid with pK = combinations ( ) ( ) , which is the most abundant combi - 1 α1 2 β2 2 γ2 4.23 and pK = 10.43. -

Treatment Protocol Copyright © 2018 Kostoff Et Al
Prevention and reversal of Alzheimer's disease: treatment protocol Copyright © 2018 Kostoff et al PREVENTION AND REVERSAL OF ALZHEIMER'S DISEASE: TREATMENT PROTOCOL by Ronald N. Kostoffa, Alan L. Porterb, Henry. A. Buchtelc (a) Research Affiliate, School of Public Policy, Georgia Institute of Technology, USA (b) Professor Emeritus, School of Public Policy, Georgia Institute of Technology, USA (c) Associate Professor, Department of Psychiatry, University of Michigan, USA KEYWORDS Alzheimer's Disease; Dementia; Text Mining; Literature-Based Discovery; Information Technology; Treatments Prevention and reversal of Alzheimer's disease: treatment protocol Copyright © 2018 Kostoff et al CITATION TO MONOGRAPH Kostoff RN, Porter AL, Buchtel HA. Prevention and reversal of Alzheimer's disease: treatment protocol. Georgia Institute of Technology. 2018. PDF. https://smartech.gatech.edu/handle/1853/59311 COPYRIGHT AND CREATIVE COMMONS LICENSE COPYRIGHT Copyright © 2018 by Ronald N. Kostoff, Alan L. Porter, Henry A. Buchtel Printed in the United States of America; First Printing, 2018 CREATIVE COMMONS LICENSE This work can be copied and redistributed in any medium or format provided that credit is given to the original author. For more details on the CC BY license, see: http://creativecommons.org/licenses/by/4.0/ This work is licensed under a Creative Commons Attribution 4.0 International License<http://creativecommons.org/licenses/by/4.0/>. DISCLAIMERS The views in this monograph are solely those of the authors, and do not represent the views of the Georgia Institute of Technology or the University of Michigan. This monograph is not intended as a substitute for the medical advice of physicians. The reader should regularly consult a physician in matters relating to his/her health and particularly with respect to any symptoms that may require diagnosis or medical attention. -

Mixed Antagonistic Effects of the Ginkgolides at Recombinant Human R1 GABAC Receptors
Neuropharmacology 63 (2012) 1127e1139 Contents lists available at SciVerse ScienceDirect Neuropharmacology journal homepage: www.elsevier.com/locate/neuropharm Mixed antagonistic effects of the ginkgolides at recombinant human r1 GABAC receptors Shelley H. Huang a, Trevor M. Lewis b, Sarah C.R. Lummis c, Andrew J. Thompson c, Mary Chebib d, Graham A.R. Johnston a, Rujee K. Duke a,* a Discipline of Pharmacology, School of Medical Sciences, Faculty of Medicine, University of Sydney, Australia b School of Medical Sciences, University of New South Wales, Australia c Department of Biochemistry, University of Cambridge, Cambridge, United Kingdom d Faculty of Pharmacy, University of Sydney, Australia article info abstract Article history: The diterpene lactones of Ginkgo biloba, ginkgolides A, B and C are antagonists at a range of Cys-loop Received 11 July 2011 receptors. This study examined the effects of the ginkgolides at recombinant human r1 GABAC recep- Received in revised form tors expressed in Xenopus oocytes using two-electrode voltage clamp. The ginkgolides were moderately 18 June 2012 potent antagonists with IC sinthemM range. At 10 mM, 30 mM and 100 mM, the ginkgolides caused Accepted 24 June 2012 50 rightward shifts of GABA doseeresponse curves and reduced maximal GABA responses, characteristic of noncompetitive antagonists, while the potencies showed a clear dependence on GABA concentration, Keywords: indicating apparent competitive antagonism. This suggests that the ginkgolides exert a mixed-type Ginkgolide Bilobalide antagonism at the r1 GABAC receptors. The ginkgolides did not exhibit any obvious use-dependent Mixed-antagonism inhibition. Fitting of the data to a number of kinetic schemes suggests an allosteric inhibition as Use-dependent a possible mechanism of action of the ginkgolides which accounts for their inhibition of the responses GABAr receptor without channel block or use-dependent inhibition. -

Gaboxadol Normalizes Behavioral Abnormalities in a Mouse Model of Fragile X Syndrome
ORIGINAL RESEARCH published: 25 June 2019 doi: 10.3389/fnbeh.2019.00141 Gaboxadol Normalizes Behavioral Abnormalities in a Mouse Model of Fragile X Syndrome Patricia Cogram 1,2,3,4, Robert M. J. Deacon 1,2,3,4, Jennifer L. Warner-Schmidt 5, Melanie J. von Schimmelmann 6, Brett S. Abrahams 6,7 and Matthew J. During 6,8* 1FRAXA-DVI, FRAXA Research Foundation, Boston, MA, United States, 2Centre for Systems Biotechnology, Biomedicine Division, Fraunhofer-Gesellschaft, Santiago, Chile, 3GEN.DDI Limited, London, United Kingdom, 4Institute of Ecology and Biodiversity (IEB), University of Chile, Santiago, Chile, 5NeuroJenic Consulting, LLC, Garden City, NY, United States, 6Ovid Therapeutics, New York, NY, United States, 7Department of Genetics and Neuroscience, Albert Einstein College of Medicine, Bronx, NY, United States, 8Department of Neurological Surgery and Molecular Virology, Immunology and Medical Genetics, Ohio State University College of Medicine, Columbus, OH, United States Fragile X syndrome (FXS) is the most common inherited form of intellectual disability and autism. FXS is also accompanied by attention problems, hyperactivity, anxiety, aggression, poor sleep, repetitive behaviors, and self-injury. Recent work supports the role of g-aminobutyric-acid (GABA), the primary inhibitory neurotransmitter in the brain, in mediating symptoms of FXS. Deficits in GABA machinery have been observed in a mouse model of FXS, including a loss of tonic inhibition in the amygdala, which is Edited by: Martine Ammassari-Teule, mediated by extrasynaptic GABAA receptors. Humans with FXS also show reduced Italian National Research Council GABAA receptor availability. Here, we sought to evaluate the potential of gaboxadol (CNR), Italy (also called OV101 and THIP), a selective and potent agonist for delta-subunit-containing Reviewed by: extrasynaptic GABA receptors (dSEGA), as a therapeutic agent for FXS by assessing Giulia Poggi, A University of Zurich, Switzerland its ability to normalize aberrant behaviors in a relatively uncharacterized mouse model Valerie J. -

Transcriptomic Analysis of Native Versus Cultured Human and Mouse Dorsal Root Ganglia Focused on Pharmacological Targets Short
bioRxiv preprint doi: https://doi.org/10.1101/766865; this version posted September 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. Transcriptomic analysis of native versus cultured human and mouse dorsal root ganglia focused on pharmacological targets Short title: Comparative transcriptomics of acutely dissected versus cultured DRGs Andi Wangzhou1, Lisa A. McIlvried2, Candler Paige1, Paulino Barragan-Iglesias1, Carolyn A. Guzman1, Gregory Dussor1, Pradipta R. Ray1,#, Robert W. Gereau IV2, # and Theodore J. Price1, # 1The University of Texas at Dallas, School of Behavioral and Brain Sciences and Center for Advanced Pain Studies, 800 W Campbell Rd. Richardson, TX, 75080, USA 2Washington University Pain Center and Department of Anesthesiology, Washington University School of Medicine # corresponding authors [email protected], [email protected] and [email protected] Funding: NIH grants T32DA007261 (LM); NS065926 and NS102161 (TJP); NS106953 and NS042595 (RWG). The authors declare no conflicts of interest Author Contributions Conceived of the Project: PRR, RWG IV and TJP Performed Experiments: AW, LAM, CP, PB-I Supervised Experiments: GD, RWG IV, TJP Analyzed Data: AW, LAM, CP, CAG, PRR Supervised Bioinformatics Analysis: PRR Drew Figures: AW, PRR Wrote and Edited Manuscript: AW, LAM, CP, GD, PRR, RWG IV, TJP All authors approved the final version of the manuscript. 1 bioRxiv preprint doi: https://doi.org/10.1101/766865; this version posted September 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. -

Characterization of Zebrafish GABAA Receptor Subunits
www.nature.com/scientificreports OPEN Characterization of zebrafsh GABAA receptor subunits Kenichiro Sadamitsu, Leona Shigemitsu, Marina Suzuki, Daishi Ito, Makoto Kashima & Hiromi Hirata* γ-Aminobutyric acid (GABA), the major inhibitory neurotransmitter in the central nervous system, exerts its efect through the activation of GABA receptors. GABAA receptors are ligand-gated chloride channels composed of fve subunit proteins. Mammals have 19 diferent GABAA receptor subunits (α1–6, β1–3, γ1–3, δ, ε, π, θ, and ρ1–3), the physiological properties of which have been assayed by electrophysiology. However, the evolutionary conservation of the physiological characteristics of diverged GABAA receptor subunits remains unclear. Zebrafsh have 23 subunits (α1, α2a, α2b, α3–5, α6a, α6b, β1–4, γ1–3, δ, π, ζ, ρ1, ρ2a, ρ2b, ρ3a, and ρ3b), but the electrophysiological properties of these subunits have not been explored. In this study, we cloned the coding sequences for zebrafsh GABAA receptor subunits and investigated their expression patterns in larval zebrafsh by whole- mount in situ hybridization. We also performed electrophysiological recordings of GABA-evoked currents from Xenopus oocytes injected with one or multiple zebrafsh GABAA receptor subunit cRNAs and calculated the half-maximal efective concentrations (EC50s) for each. Our results revealed the spatial expressions and electrophysiological GABA sensitivities of zebrafsh GABAA receptors, suggesting that the properties of GABAA receptor subunits are conserved among vertebrates. γ-Aminobutyric acid (GABA), the major inhibitory neurotransmitter in the central nervous system of vertebrates, 1 controls the excitability of neural networks mainly through GABA A receptors . Te GABAA receptor mediates two types of inhibition, known as phasic and tonic inhibition2. -

Anti-CHRNA10 Antibody (ARG58421)
Product datasheet [email protected] ARG58421 Package: 100 μl anti-CHRNA10 antibody Store at: -20°C Summary Product Description Rabbit Polyclonal antibody recognizes CHRNA10 Tested Reactivity Hu Predict Reactivity Rat Tested Application IHC-P, WB Host Rabbit Clonality Polyclonal Isotype IgG Target Name CHRNA10 Antigen Species Human Immunogen KLH-conjugated synthetic peptide corresponding to aa. 179-206 (Center) of Human CHRNA10. Conjugation Un-conjugated Alternate Names NACHR alpha-10; Neuronal acetylcholine receptor subunit alpha-10; Nicotinic acetylcholine receptor subunit alpha-10 Application Instructions Application table Application Dilution IHC-P 1:10 - 1:50 WB 1:1000 Application Note * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. Positive Control NCI-H292 Calculated Mw 50 kDa Properties Form Liquid Purification Purification with Protein A and immunogen peptide. Buffer PBS and 0.09% (W/V) Sodium azide. Preservative 0.09% (W/V) Sodium azide. Storage instruction For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C or below. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. www.arigobio.com 1/2 Note For laboratory research only, not for drug, diagnostic or other use. Bioinformation Gene Symbol CHRNA10 Gene Full Name cholinergic receptor, nicotinic, alpha 10 (neuronal) Function Ionotropic receptor with a probable role in the modulation of auditory stimuli. Agonist binding may induce an extensive change in conformation that affects all subunits and leads to opening of an ion- conducting channel across the plasma membrane. -

Sex Differences in Glutamate Receptor Gene Expression in Major Depression and Suicide
Molecular Psychiatry (2015) 20, 1057–1068 © 2015 Macmillan Publishers Limited All rights reserved 1359-4184/15 www.nature.com/mp IMMEDIATE COMMUNICATION Sex differences in glutamate receptor gene expression in major depression and suicide AL Gray1, TM Hyde2,3, A Deep-Soboslay2, JE Kleinman2 and MS Sodhi1,4 Accumulating data indicate that the glutamate system is disrupted in major depressive disorder (MDD), and recent clinical research suggests that ketamine, an antagonist of the N-methyl-D-aspartate (NMDA) glutamate receptor (GluR), has rapid antidepressant efficacy. Here we report findings from gene expression studies of a large cohort of postmortem subjects, including subjects with MDD and controls. Our data reveal higher expression levels of the majority of glutamatergic genes tested in the dorsolateral prefrontal cortex (DLPFC) in MDD (F21,59 = 2.32, P = 0.006). Posthoc data indicate that these gene expression differences occurred mostly in the female subjects. Higher expression levels of GRIN1, GRIN2A-D, GRIA2-4, GRIK1-2, GRM1, GRM4, GRM5 and GRM7 were detected in the female patients with MDD. In contrast, GRM5 expression was lower in male MDD patients relative to male controls. When MDD suicides were compared with MDD non-suicides, GRIN2B, GRIK3 and GRM2 were expressed at higher levels in the suicides. Higher expression levels were detected for several additional genes, but these were not statistically significant after correction for multiple comparisons. In summary, our analyses indicate a generalized disruption of the regulation of the GluRs in the DLPFC of females with MDD, with more specific GluR alterations in the suicides and in the male groups.