Glycerides (Mono And
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
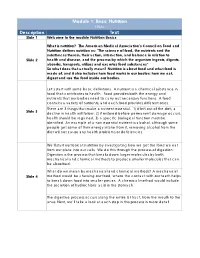
Module 1: Basic Nutrition FINAL Description Text Slide 1 Welcome to the Module Nutrition Basics
Module 1: Basic Nutrition FINAL Description Text Slide 1 Welcome to the module Nutrition Basics What is nutrition? The American Medical Association’s Council on Food and Nutrition defines nutrition as: "The science of food, the nutrients and the substances therein, their action, interaction, and balance in relation to Slide 2 health and disease, and the process by which the organism ingests, digests, absorbs, transports, utilizes and excretes food substances" So what does that actually mean? Nutrition is about food and what food is made of, and it also includes how food works in our bodies: how we eat, digest and use the food inside our bodies. Let’s start with some basic definitions. A nutrient is a chemical substance in food that contributes to health. Food provides both the energy and nutrients that our bodies need to carry out necessary functions. A food contains a variety of nutrients, and each food provides different ones. There are 3 things that make a nutrient essential. 1) if left out of the diet, a Slide 3 decline in health will follow, 2) if restored before permanent damage occurs, health should be regained, 3) a specific biological function must be identified. An example of a non essential nutrient is alcohol, although some people get some of their energy intake from it, removing alcohol from the diet will not cause any health problems or deficiencies. We‘ll start our look at nutrition by investigating how we get the food we eat from our plate into our cells. We do this through the process of digestion. -

II. Phosphoglyceride Fatty Acids
Pediat. Res. 8: 93-102 (1974) Arachidonic acid ethanolamine phosphoglycerides brain lipids choline phosphoglycerides Some Chemical Aspects of Human Brain Development. II. Phosphoglyceride Fatty Acids MANUELAMARTINEZ, CARMEN CONDE, AND ANGELBALLABRIGA[~~~ Autonomous University, School of Medicine, Children's Hospital of the "Seguridad Social," Barcelona, Spain Extract The fatty acids of total phosphoglycerides (TPG), ethanolamine phosphoglycerides (EPG), and choline phosphoglycerides (CPG) were obtained by mild alkaline trans- methylation from lipid extracts of whole cerebrum and then analyzed by gas chroma- tography. A complete brain hemisphere from each of the 34 newborn infants reported previously was homogenized and its lipids extracted according to the procedure speci- fied in that report. As the gestational age of the children went up, a statistically significant increase of the n-3/n-6 ratio and, especially, of the 22:4(n-6)/22:5(n-6) index was observed. Other ratios, such as the n-6/n-9 and the 18:0/18: 1(n-9), were also studied in the fatty acid patterns of EPG and CPG. Both of them showed sig- nificant increases with the gestational age of the infants. The [22 :4(n-6) + 22 :5(n-6)]/ 20:4(n-6) index, an indicator of the elongation process of arachidonic acid, on the other hand, did not show appreciable changes with maturation in TPG during this period of life. When ethanolamine and choline phosphoglycerides were analyzed separately, however, the elongation of arachidonic acid did rise with the gestational age in the former whereas it decreased in the latter. As intrauterine maturation of the human brain progresses, changes in the polyun- saturated fatty acid patterns, contrary to those observed in undernourished animals, take place. -

Understanding the Basics of Efas
[Nutritional Lipids] Vol. 14 No 9 September 2009 Understanding the Basics of EFAs By Robin Koon, Contributing Editor While knowledge of essential fatty acids (EFAs) is growing among marketers and consumers, reviewing the comprehensive lipid category provides a point of references to delve further into the structure and function of these beneficial fats. Dietary lipids are an essential constituent of the human diet along with carbohydrates, proteins and fiber. Fats are a source of energy, supplying about 9 calories per gram, and make up approximately 40 percent of the body’s normal caloric intake. Lipids are a large and diverse group of naturally occurring organic compounds related by their solubility in non-polar organic solvents (e.g., ether, chloroform, acetone, benzene, etc.) and insolubility in water. In the human diet, triglycerides are the most abundantly consumed form of edible dietary lipids and normally constitute approximately 95 percent of total fat ingested. The remaining 5 percent is in the form of cerebrosides, phospholipids, fatty acids, fatty alcohols, cholesterol, sterols, terpenes, etc. The average human being consumes between 50 and 200 g/d of fat. Triglycerides (TG) are comprised predominantly of fatty acids (present in the form of esters of glycerol), although they are technically a combination of glycerol and three fatty acids, sometimes referred to as triacylglycerol. Glycerol is a three-carbon chain alcohol molecule with chemical formula of C3H803. When combined with one, two or three fatty acids, it forms a monoglyceride, diglyceride or triglyceride, respectively. Triglycerides are generally not well-absorbed in the gut and are enzymatically hydrolyzed, which splits the TG into its components by removing the fatty acids from their glycerol base, so all of these components can be easily absorbed. -

Fats and Fatty Acid in Human Nutrition
ISSN 0254-4725 91 FAO Fats and fatty acids FOOD AND NUTRITION PAPER in human nutrition Report of an expert consultation 91 Fats and fatty acids in human nutrition − Report of an expert consultation Knowledge of the role of fatty acids in determining health and nutritional well-being has expanded dramatically in the past 15 years. In November 2008, an international consultation of experts was convened to consider recent scientific developments, particularly with respect to the role of fatty acids in neonatal and infant growth and development, health maintenance, the prevention of cardiovascular disease, diabetes, cancers and age-related functional decline. This report will be a useful reference for nutrition scientists, medical researchers, designers of public health interventions and food producers. ISBN 978-92-5-106733-8 ISSN 0254-4725 9 7 8 9 2 5 1 0 6 7 3 3 8 Food and Agriculture I1953E/1/11.10 Organization of FAO the United Nations FAO Fats and fatty acids FOOD AND NUTRITION in human nutrition PAPER Report of an expert consultation 91 10 − 14 November 2008 Geneva FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 2010 The designations employed and the presentation of material in this information product do not imply the expression of any opinion whatsoever on the part of the Food and Agriculture Organization of the United Nations (FAO) concerning the legal or development status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. The mention of specific companies or products of manufacturers, whether or not these have been patented, does not imply that these have been endorsed or recommended by FAO in preference to others of a similar nature that are not mentioned. -

Medium-Chain Fatty Acids and Monoglycerides As Feed Additives for Pig Production: Towards Gut Health Improvement and Feed Pathogen Mitigation Joshua A
Jackman et al. Journal of Animal Science and Biotechnology (2020) 11:44 https://doi.org/10.1186/s40104-020-00446-1 REVIEW Open Access Medium-chain fatty acids and monoglycerides as feed additives for pig production: towards gut health improvement and feed pathogen mitigation Joshua A. Jackman1*, R. Dean Boyd2,3 and Charles C. Elrod4,5* Abstract Ongoing challenges in the swine industry, such as reduced access to antibiotics and virus outbreaks (e.g., porcine epidemic diarrhea virus, African swine fever virus), have prompted calls for innovative feed additives to support pig production. Medium-chain fatty acids (MCFAs) and monoglycerides have emerged as a potential option due to key molecular features and versatile functions, including inhibitory activity against viral and bacterial pathogens. In this review, we summarize recent studies examining the potential of MCFAs and monoglycerides as feed additives to improve pig gut health and to mitigate feed pathogens. The molecular properties and biological functions of MCFAs and monoglycerides are first introduced along with an overview of intervention needs at different stages of pig production. The latest progress in testing MCFAs and monoglycerides as feed additives in pig diets is then presented, and their effects on a wide range of production issues, such as growth performance, pathogenic infections, and gut health, are covered. The utilization of MCFAs and monoglycerides together with other feed additives such as organic acids and probiotics is also described, along with advances in molecular encapsulation and delivery strategies. Finally, we discuss how MCFAs and monoglycerides demonstrate potential for feed pathogen mitigation to curb disease transmission. Looking forward, we envision that MCFAs and monoglycerides may become an important class of feed additives in pig production for gut health improvement and feed pathogen mitigation. -

Information to Users
INFORMATION TO USERS The most advanced technology has been used to photograph and reproduce this manuscript from the microfilm master. UMI films the text directly from the original or copy submitted. Thus, some thesis and dissertation copies are in typewriter face, while others may be from any type of computer printer. The quality of this reproduction is dependent upon the quality of the copy submitted. Broken or indistinct print, colored or poor quality illustrations and photographs, print bleedthrough, substandard margins, and improper alignment can adversely affect reproduction. In the unlikely event that the author did not send UMI a complete manuscript and there are missing pages, these will be noted. Also, if unauthorized copyright material had to be removed, a note will indicate the deletion. Oversize materials (e.g., maps, drawings, charts) are reproduced by sectioning the original, beginning at the upper left-hand corner and continuing from left to right in equal sections with small overlaps. Each original is also photographed in one exposure and is included in reduced form at the back of the book. Photographs included in the original manuscript have been reproduced xerographically in this copy. Higher quality 6" x 9" black and white photographic prints are available for any photographs or illustrations appearing in this copy for an additional charge. Contact UMI directly to order. University Microfilms International A Bell & Howell Information Company 300 North Zeeb Road, Ann Arbor, Ml 48106-1346 USA 313/761-4700 800/521-0600 Order Number 00S1079 The survival ofStaphylococcus aureus in abscesses from streptozotocin-induced diabetic mice Harvey, Kevin Michael, Ph.D. -

Deacidification of High-Acid Rice Bran Oil by Reesterification With
Deacidification of High-Acid Rice Bran Oil by Reesterification with Monoglyceride B.K. De and D.K. Bhattacharyya* Department of Chemical Technology, University of Calcutta, Oil Technology Division, Calcutta 700 009, West Bengal, India ABSTRACT: Autocatalytic esterification of free fatty acids (FFA) better process for extracting FFA along with color and peroxide in rice bran oil (RBO) containing high FFA (9.5 to 35.0% w/w) bodies. Bhattacharyya et al. (5) took advantage of this process was examined at a high temperature (210°C) and under low pres- in refining high FFA (20.5 to 56.0%) RBO and showed that a sure (10 mm Hg). The study was conducted to determine the ef- good quality edible-grade RBO could be obtained by this fectiveness of monoglyceride in esterifying the FFA of RBO. The process. study showed that monoglycerides can reduce the FFA level of Chemical or biochemical reesterification of FFA is another degummed, dewaxed, and bleached RBO to an acceptable level approach for the deacidification of high-FFA RBO. Enzymatic (0.5 ± 0.10 to 3.5 ± 0.19% w/w) depending on the FFA content of the crude oil. This allows RBO to be alkali refined, bleached, deacidification (6–10) of vegetable oils by microbial lipases and deodorized or simply deodorized after monoglyceride treat- (i.e., biorefining) has recently received much attention. Lipases ment to obtain a good quality oil. The color of the refined oil is can esterify the FFA to hydroxyl groups containing compounds dependent upon the color of the crude oil used. already present in the oil or to the hydroxyl groups of added Paper no. -

Lymphatic Absorption of Docosahexaenoic Acid Given As Monoglyceride, Diglyceride, Triglyceride, and Ethyl Ester in Rats
JNutrSci Vitaminol, 48, 30-35, 2002 Lymphatic Absorption of Docosahexaenoic Acid Given as Monoglyceride, Diglyceride, Triglyceride, and Ethyl Ester in Rats Fumiaki BANN01,*,Shinji DOIsAKI2,Nobutoshi SHIMIZU2 and Kenshiro FUJIMOTO1 1 Graduate School of Agricultural Science , Tohoku University, 1-1 Tsutsumidori-Amamiyamachi,Aoba, Sendai 981-8555, Japan 2 Central Research Laboratory , Nippon Suisan Kaisha, Ltd., 559-6 Kitano-machi, Hachioji, Tokyo192-0906, Japan (Received March 9, 2001) Summary Lymphatic absorption of docosahexaenoic acids (DHA) given as monoglyc eride (MG), consisting of 1(or 3)-species (91.4%), 2-species (4.2%) and diglyceride (DG)con sisting of 1,3-species (70.8%), 1(or 3),2-species (28.6%), were investigated in comparison with that of triglyceride (TG) and ethyl ester (EE). Rats were infused with a lipid emulsion containing 200mg of DHA-MG, DG, TG, or EE via a gastric cannula. Lymph was collected through the thoracic lymph duct at 2h intervals for 10h and at a single collection from 10 to 30h. Physiological saline containing glucose was infused (2mL/h) throughout the lymph collection. The overall recovery of DHA at 30h after its infusion was significantly higher in the rank order DHA-MG>DG>TG=EE. Moreover, time-dependent changes in re covery rates from 2 to 10h of DHA given as MG were significantly higher than those of the corresponding DG, TG, and EE. These results indicate that DHA-MG and DG are absorbed and transported more effectively than TG and EE forms under restricted water supply, even if they mainly consist of 1(or 3)-species. -

Long Active Injectable Formulations Containing Triacetin
Europaisches Patentamt J European Patent Office OT) Publication number: 0 413 538 A1 Office europeen des brevets EUROPEAN PATENT APPLICATION © Application number: 90308881.3 © int. CIA A61K 31/71, A61K 9/00, A61K 47/14 © Date of filing: 13.08.90 © Priority: 14.08.89 US 393356 © Applicant: MERCK & CO. INC. 126, East Lincoln Avenue P.O. Box 2000 © Date of publication of application: Rahway New Jersey 07065-0900(US) 20.02.91 Bulletin 91/08 © Inventor: Williams, James B. © Designated Contracting States: 4 Coventry Drive CH DE FR GB IT LI NL Freehold, NJ 07728(US) © Representative: Hesketh, Alan, Dr. et al European Patent Department Merck & Co., Inc. Terlings Park Eastwick Road Harlow Essex, CM20 2QR(GB) © Long active injectable formulations containing triacetin. © There is disclosed an injectable formulation containing triacetin and an avermectin compound which has been discovered to provide an unexpectedly long duration of activity. The formulation contains the avermectin active ingredient and at least 50% triacetin and effective levels of the avermectin compounds have been maintained for up to 42 days. Prior injectable formulations provided only 14 days duration of activity. 00 CO If) CO 5 o Q_ LU Xerox Copy Centre EP 0 413 538 A1 BACKGROUND OF THE INVENTION The avermectin series of compounds are potent anthelmintic and antiparasitic agents against internal and external parasites. The natural product avermectins are disclosed in US 4,310,519 to Albers Schonberg et al ., and the 22,23-dihydro avermectin compounds are disclosed in Chabala et al, US 4,199,569. Triacetin (Triacetyl glycerine) is a known formulating agent which is generally recognized as safe by the 5 US Food and Drug Administration. -

Production of Glyceryl Monostearate by Immobilized Candida Antarctica B Lipase in Organic Media
Journal of Applied Biotechnology & Bioengineering Research Article Open Access Production of glyceryl monostearate by immobilized candida antarctica B lipase in organic media Abstract Volume 2 Issue 3 - 2017 Chemical methods of synthesizing monoglycerides (MGs) produce mixture of Kavadia MR, Yadav MG, Odaneth AA, Arvind acylglycerols making the synthesis of MGs of a single fatty acid type not possible through this route. Lipases due to their selectivity and specificity offer a useful way M Lali DBT-ICT-Centre for Energy Biosciences, India of synthesizing highly purified single fatty acid containing monoglycerides. Glyceryl monostearate (GMS), a nonionic amphiphilic monoglyceride of glycerol and stearic Correspondence: Annmmma A Odaneth, Assistant Professor acid is widely used as emulsifier in food, cosmetic, pharmaceutical and textile in Biochemistry, DBT-ICT Centre for Energy Biosciences, industry. In the present work, lipase mediated synthesis of GMS was explored using Institute of Chemical Technology, Nathalal Parikh Marg, Novozym 435 and indigenously immobilized Candida antarctica B lipase. Direct Mumbai-400 019, Mahrashtra, India, Fax +91-22-24145614, Tel esterification of glycerol and stearic acid was used as strategy and various parameters +91-22-33612312, Email [email protected] that influence synthesis of glyceryl monostearate such as molar ratio of substrates, enzyme load, reaction time and solvent polarity were systematically studied using Received: October 27, 2016 | Published: February 28, 2017 Novozym 435 lipase. Esterification efficiency of indigenously immobilized Candida antarctica B lipase and Novozym 435 was compared for synthesis of GMS and indigenously immobilized Candida antarctica B lipase gave conversion competitive to Novozym 435. Indigenously immobilized Candida antarctica B lipase in the presence of homogenous substrate solution using tert-butyl alcohol as the reaction medium, resulted in exclusive production of monoglyceride without formation of diglyceride. -

Utilization of Glycerol-C'4 for Intestinal Glyceride Esterifi- Cation: Studies in a Patient with Chyluria * PETER R
Journal of Clinical Investigation Vol. 43, No. 3, 1964 Utilization of Glycerol-C'4 for Intestinal Glyceride Esterifi- cation: Studies in a Patient with Chyluria * PETER R. HOLT (Fromz the Gastroinitestinial Research Laboratory anid Medical Service, St. Luike's Hospital, New York, N. Y.) Ingested long chain triglycerides are hydrolyzed quantitative estimation of the incorporation of the to partial glycerides and free fatty acids in the in- exogenous labeled glycerol into intestinal lymiplh testinal lumen, but the extent of intraluminal hy- lipids. drolysis is still in dispute (2). After digestion Materials and Methods and absorption into the intestinal mucosa, re-esteri- fication of the fatty acids is necessary before in- Cliniical features. The subject, a 63-year-old Puerto corporation into chylomicra, which pass into the Rican male, was admitted to St. Luke's Hospital with a 30-year history of chyluria. Complete clinical data on intestinal lymphatics. Favarger and Gerlach this patient have been published (10). Lymphography originally considered that mucosal esterification of revealed tortuous dilated intra-abdominal lymphatics fatty acids occurred by a mechanism that was a with communications from the cisterna chyli and para- reversal of lipolysis (3). Subsequent studies in- aortic lymph glands to the left kidney and bladder (10). dicated that a more complex system of synthesis Cystoscopy showed a fistula in the region of the bladder neck. was involved (4). In addition, glycerol, liberated Before these studies the patient had been maintained onl within the intestinal lumen by hydrolysis, was not formula feedings, and while ingesting only long chain believed to be a substrate for glyceride-glycerol fats (C16 to C18 triglycerides), excreted in the urine fat synthesis, for when C14-labeled glycerol and free amounting to 3.5 to 14% (mean, 8.5%) of the daily fat fatty acids or triglycerides were fed to rats, little intake. -

A Novel Omega‐3 Glyceride Mixture Enhances Enrichment Of
A novel omega‐3 glyceride mixture enhances enrichment of eicosapentaenoic acid and docosahexaenoic acid after single dosing in healthy older adults: results from a double‐ blind crossover trial Helena L. Fisk1,2,*, Grete Mørk Kindberg3, Svein Olaf Hustvedt3, and Philip C Calder1,2 1School of Human Development and Health, Faculty of Medicine, University of Southampton, Southampton SO16 6YD, United Kingdom; 2NIHR Southampton Biomedical Research Centre, University Hospital Southampton NHS Foundation Trust and University of Southampton, Southampton SO16 6YD, United Kingdom; 3BASF Norge AS, 0283 Oslo, Norway. *Author for correspondence: School of Human Development and Health, Faculty of Medicine, University of Southampton, IDS Building, MP887 Southampton General Hospital, Tremona Road, Southampton SO16 6YD, United Kingdom Running heading: Enrichment of EPA and DHA after single dosing List of abbreviations: ω‐3, omega‐3; ALA, alpha‐linoleic acid; BMI, body mass index; Cmax, maximal concentration; DHA, docosahexaenoic acid; DPA, docosapentaenoic acid; EE, ethyl‐ester(s); EPA, eicosapentaenoic acid; FA, fatty acid(s); FAME, fatty acid methyl esters; FFA, free fatty acid(s); Glyceride formulation, mix of mono, di and triglycerides; HDL, high density lipoprotein; HOMA2‐IR, homeostatic model assessment of insulin resistance; hsCRP, high sensitivity C‐reactive protein; iAUC, incremental area under the curve; ω‐3 LCPUFA, omega‐3 long chain polyunsaturated fatty acid(s); SMEDS, self‐micro‐emulsifying delivery system; TG, triglyceride(s); Tmax,, time at which maximal concentration occurs. 1 1 Abstract 2 A glyceride mix of mono, di, and triglycerides increases solubilisation and enhances emulsification of 3 omega‐3 (ω‐3) fatty acid (FA) containing lipids in the stomach. This allows for better access of 4 digestive enzymes, pivotal for the release of bioactive ω‐3 FA.