Biological Functions of Cytokeratin 18 in Cancer
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genetic Background Effects of Keratin 8 and 18 in a DDC-Induced Hepatotoxicity and Mallory-Denk Body Formation Mouse Model
Laboratory Investigation (2012) 92, 857–867 & 2012 USCAP, Inc All rights reserved 0023-6837/12 $32.00 Genetic background effects of keratin 8 and 18 in a DDC-induced hepatotoxicity and Mallory-Denk body formation mouse model Johannes Haybaeck1, Cornelia Stumptner1, Andrea Thueringer1, Thomas Kolbe2, Thomas M Magin3, Michael Hesse4, Peter Fickert5, Oleksiy Tsybrovskyy1, Heimo Mu¨ller1, Michael Trauner5,6, Kurt Zatloukal1 and Helmut Denk1 Keratin 8 (K8) and keratin 18 (K18) form the major hepatocyte cytoskeleton. We investigated the impact of genetic loss of either K8 or K18 on liver homeostasis under toxic stress with the hypothesis that K8 and K18 exert different functions. krt8À/À and krt18À/À mice crossed into the same 129-ola genetic background were treated by acute and chronic ad- ministration of 3,5-diethoxy-carbonyl-1,4-dihydrocollidine (DDC). In acutely DDC-intoxicated mice, macrovesicular steatosis was more pronounced in krt8À/À and krt18À/À compared with wild-type (wt) animals. Mallory-Denk bodies (MDBs) appeared in krt18À/À mice already at an early stage of intoxication in contrast to krt8À/À mice that did not display MDB formation when fed with DDC. Keratin-deficient mice displayed significantly lower numbers of apoptotic hepatocytes than wt animals. krt8À/À, krt18À/À and control mice displayed comparable cell proliferation rates. Chronically DDC-intoxicated krt18À/À and wt mice showed a similarly increased degree of steatohepatitis with hepatocyte ballooning and MDB formation. In krt8À/À mice, steatosis was less, ballooning, and MDBs were absent. krt18À/À mice developed MDBs whereas krt8À/À mice on the same genetic background did not, highlighting the significance of different structural properties of keratins. -
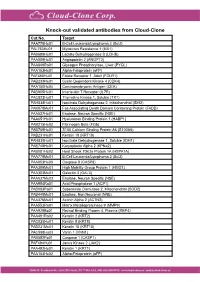
Knock-Out Validated Antibodies from Cloud-Clone Cat.No
Knock-out validated antibodies from Cloud-Clone Cat.No. Target PAA778Hu01 B-Cell Leukemia/Lymphoma 2 (Bcl2) PAL763Hu01 Myxovirus Resistance 1 (MX1) PAB698Hu01 Lactate Dehydrogenase B (LDHB) PAA009Hu01 Angiopoietin 2 (ANGPT2) PAA849Ra01 Glycogen Phosphorylase, Liver (PYGL) PAA153Hu01 Alpha-Fetoprotein (aFP) PAF460Hu01 Folate Receptor 1, Adult (FOLR1) PAB233Hu01 Cyclin Dependent Kinase 4 (CDK4) PAA150Hu04 Carcinoembryonic Antigen (CEA) PAB905Hu01 Interleukin 7 Receptor (IL7R) PAC823Hu01 Thymidine Kinase 1, Soluble (TK1) PAH838Hu01 Isocitrate Dehydrogenase 2, mitochondrial (IDH2) PAK078Mu01 Fas Associating Death Domain Containing Protein (FADD) PAA537Hu01 Enolase, Neuron Specific (NSE) PAA651Hu01 Hyaluronan Binding Protein 1 (HABP1) PAB215Hu02 Fibrinogen Beta (FGb) PAB769Hu01 S100 Calcium Binding Protein A6 (S100A6) PAB231Hu01 Keratin 18 (KRT18) PAH839Hu01 Isocitrate Dehydrogenase 1, Soluble (IDH1) PAE748Hu01 Karyopherin Alpha 2 (KPNa2) PAB081Hu02 Heat Shock 70kDa Protein 1A (HSPA1A) PAA778Mu01 B-Cell Leukemia/Lymphoma 2 (Bcl2) PAA853Hu03 Caspase 8 (CASP8) PAA399Mu01 High Mobility Group Protein 1 (HMG1) PAA303Mu01 Galectin 3 (GAL3) PAA537Mu02 Enolase, Neuron Specific (NSE) PAA994Ra01 Acid Phosphatase 1 (ACP1) PAB083Ra01 Superoxide Dismutase 2, Mitochondrial (SOD2) PAB449Mu01 Enolase, Non Neuronal (NNE) PAA376Mu01 Actinin Alpha 2 (ACTN2) PAA553Ra01 Matrix Metalloproteinase 9 (MMP9) PAA929Bo01 Retinol Binding Protein 4, Plasma (RBP4) PAA491Ra02 Keratin 2 (KRT2) PAC025Hu01 Keratin 8 (KRT8) PAB231Mu01 Keratin 18 (KRT18) PAC598Hu03 Vanin 1 (VNN1) -

Motility-Related Actinin Alpha-4 Is Associated with Advanced And
Laboratory Investigation (2008) 88, 602–614 & 2008 USCAP, Inc All rights reserved 0023-6837/08 $30.00 Motility-related actinin alpha-4 is associated with advanced and metastatic ovarian carcinoma Maria V Barbolina1, Brian P Adley2, David L Kelly3, Angela J Fought4, Denise M Scholtens4, Lonnie D Shea1 and M Sharon Stack5 Advanced and metastatic ovarian cancer is a leading cause of death from gynecologic malignancies. A more detailed understanding of the factors controlling invasion and metastasis may lead to novel anti-metastatic therapies. To model cellular interactions that occur during intraperitoneal metastasis, comparative cDNA microarray analysis and confirmatory real-time reverse transcription PCR (RT-PCR) were employed to uncover changes in gene expression that may occur in late stage ovarian cancer in response to microenvironmental cues, particularly native three-dimensional collagen I. Gene expression in human ovarian carcinoma tissues was evaluated on the RNA and protein level using real-time RT-PCR and immunohistochemistry. Cell invasion and migration were evaluated in a collagen invasion assay and a scratch wound assay. Three-dimensional collagen I culture led to differential expression of several genes. The role of actinin alpha-4 (ACTN4), a cytoskeleton-associated protein implicated in the regulation of cell motility, was examined in detail. ACTN4 RNA and protein expression were associated with advanced and metastatic human ovarian carcinoma. This report demonstrates that a cytoskeletal-associated protein ACTN4 is upregulated by three-dimensional collagen culture conditions, leading to increased invasion and motility of ovarian cancer cells. Expression of ACTN4 in human ovarian tumors was found to be associated with advanced-stage disease and peritoneal metastases. -

Human Induced Pluripotent Stem Cell–Derived Podocytes Mature Into Vascularized Glomeruli Upon Experimental Transplantation
BASIC RESEARCH www.jasn.org Human Induced Pluripotent Stem Cell–Derived Podocytes Mature into Vascularized Glomeruli upon Experimental Transplantation † Sazia Sharmin,* Atsuhiro Taguchi,* Yusuke Kaku,* Yasuhiro Yoshimura,* Tomoko Ohmori,* ‡ † ‡ Tetsushi Sakuma, Masashi Mukoyama, Takashi Yamamoto, Hidetake Kurihara,§ and | Ryuichi Nishinakamura* *Department of Kidney Development, Institute of Molecular Embryology and Genetics, and †Department of Nephrology, Faculty of Life Sciences, Kumamoto University, Kumamoto, Japan; ‡Department of Mathematical and Life Sciences, Graduate School of Science, Hiroshima University, Hiroshima, Japan; §Division of Anatomy, Juntendo University School of Medicine, Tokyo, Japan; and |Japan Science and Technology Agency, CREST, Kumamoto, Japan ABSTRACT Glomerular podocytes express proteins, such as nephrin, that constitute the slit diaphragm, thereby contributing to the filtration process in the kidney. Glomerular development has been analyzed mainly in mice, whereas analysis of human kidney development has been minimal because of limited access to embryonic kidneys. We previously reported the induction of three-dimensional primordial glomeruli from human induced pluripotent stem (iPS) cells. Here, using transcription activator–like effector nuclease-mediated homologous recombination, we generated human iPS cell lines that express green fluorescent protein (GFP) in the NPHS1 locus, which encodes nephrin, and we show that GFP expression facilitated accurate visualization of nephrin-positive podocyte formation in -

Supplementary Material Contents
Supplementary Material Contents Immune modulating proteins identified from exosomal samples.....................................................................2 Figure S1: Overlap between exosomal and soluble proteomes.................................................................................... 4 Bacterial strains:..............................................................................................................................................4 Figure S2: Variability between subjects of effects of exosomes on BL21-lux growth.................................................... 5 Figure S3: Early effects of exosomes on growth of BL21 E. coli .................................................................................... 5 Figure S4: Exosomal Lysis............................................................................................................................................ 6 Figure S5: Effect of pH on exosomal action.................................................................................................................. 7 Figure S6: Effect of exosomes on growth of UPEC (pH = 6.5) suspended in exosome-depleted urine supernatant ....... 8 Effective exosomal concentration....................................................................................................................8 Figure S7: Sample constitution for luminometry experiments..................................................................................... 8 Figure S8: Determining effective concentration ......................................................................................................... -

Biological Functions of Cytokeratin 18 in Cancer
Published OnlineFirst March 27, 2012; DOI: 10.1158/1541-7786.MCR-11-0222 Molecular Cancer Review Research Biological Functions of Cytokeratin 18 in Cancer Yu-Rong Weng1,2, Yun Cui1,2, and Jing-Yuan Fang1,2,3 Abstract The structural proteins cytokeratin 18 (CK18) and its coexpressed complementary partner CK8 are expressed in a variety of adult epithelial organs and may play a role in carcinogenesis. In this study, we focused on the biological functions of CK18, which is thought to modulate intracellular signaling and operates in conjunction with various related proteins. CK18 may affect carcinogenesis through several signaling pathways, including the phosphoinosi- tide 3-kinase (PI3K)/Akt, Wnt, and extracellular signal-regulated kinase (ERK) mitogen-activated protein kinase (MAPK) signaling pathways. CK18 acts as an identical target of Akt in the PI3K/Akt pathway and of ERK1/2 in the ERK MAPK pathway, and regulation of CK18 by Wnt is involved in Akt activation. Finally, we discuss the importance of gaining a more complete understanding of the expression of CK18 during carcinogenesis, and suggest potential clinical applications of that understanding. Mol Cancer Res; 10(4); 1–9. Ó2012 AACR. Introduction epithelial organs, such as the liver, lung, kidney, pancreas, The intermediate filaments consist of a large number of gastrointestinal tract, and mammary gland, and are also nuclear and cytoplasmic proteins that are expressed in a expressed by cancers that arise from these tissues (7). In the tissue- and differentiation-dependent manner. The compo- absence of CK8, the CK18 protein is degraded and keratin fi intermediate filaments are not formed (8). -

Keratin Gene Expression in Non-Epithelial Tissues Detection with Polymerase Chain Reaction
American Journal ofPathology, Vol. 142, No. 4, April 1993 Copyright C American Societyfor Investigative Pathology Keratin Gene Expression in Non-Epithelial Tissues Detection with Polymerase Chain Reaction S. Thomas Traweek, Jane Liu, and ceUs, or any of the seven normal bone marrows Hector Battifora examined. Dilutional experiments showedPCR to From the Sylvia Cowan Laboratory ofSurgical Pathology, be highly sensitive in the detection of keratin 19 Division ofPathology, City ofHope National Medical gene expression, capable of registering one Center, Duarte, California MCF-7 ceU in 106 HL-60 ceUs. These studies show that variable levels of keratin 8 and 18 gene ex- pression may be detected by PCR in a wide varlety ofnon-epithelial tissues, supportlingprevious im- Keratinfilament are characteristicaly present in munohistochemical and phylogenetic studies. epithelial ceUs and tumors, but have also been de- However, keratin 19 gene expression appears to tected in many normal and neoplastic non- be more restricted and was not evident in any he- epithelial ceU types using immunohistochemical matopoietic ceUs devoid of contaminating stro- techniques. To investigate the validity of this mal elements. these flndings suggest a role for seemingly aberrant protein expression, we ap- PCR in the detection ofepithelialmicrometastasis plied the highly sensitivepolymerase chain reac- in certain sites, particularly bone marrow. (Am tion (PCR) technique to study keratin gene ex- JPathol 1993, 142:1111-1118) pression in a variety of non-epithelial tissues. Total RNA was extracted from nine samples of leiomyosarcoma, four non-Hodgkin's lymphoma, Intermediate filaments (IF) are the principal compo- seven normal bone marrows, normal lymph node, nents of the cytoskeleton in mammalian cells. -

Association Between Keratin and Vimentin Expression, Malignant Phenotype, and Survival in Postmenopausal Breast Cancer Patients1
2698 Vol. 5, 2698–2703, October 1999 Clinical Cancer Research Advances in Brief Association between Keratin and Vimentin Expression, Malignant Phenotype, and Survival in Postmenopausal Breast Cancer Patients1 Patricia A. Thomas,2 Dawn A. Kirschmann, tumors. These latter two groups demonstrated similar James R. Cerhan, Robert Folberg, Kaplan-Meier survival curves; the former group (keratin Elisabeth A. Seftor, Thomas A. Sellers, and and vimentin in approximately similar amounts) demon- 3 strated a poorer survival, with a hazard ratio of 2.1 (95% Mary J. C. Hendrix confidence interval, 0.5–9.6). These data suggest that rela- University of Iowa Cancer Center [P. A. T., D. A. K., J. R. C., R. F., tive keratin and vimentin IF expression is more indicative of E. A. S., M. J. C. H.], Departments of Pathology [P. A. T., R. F.], prognosis and tumor phenotype than either IF marker de- Ophthalmology [R. F.], Anatomy and Cell Biology [D. A. K., E. A. S., M. J. C. H.], Preventive Medicine and Environmental Health tected independently. [J. R. C.], The University of Iowa, Iowa City, Iowa 52242-1109 and Health Sciences Research [T. A. S.], Mayo Clinic, Rochester, Introduction Minnesota 55905 IF4 proteins are best known for their cell type specificity and their static structural role as components of the vertebrate Abstract cell cytoskeleton. There is growing evidence for IF involvement Pathology observational reports and experimental data in a variety of dynamic cellular functions, including intercellular suggest that keratin and vimentin intermediate filament (IF) and cell-to-extracellular matrix signal transduction, cellular mo- coexpression in breast cancer confers a more aggressive tility, and tumor cell invasiveness (1–5). -

Id-Gestational Lethality in Mice Lacking Eratin 8
Downloaded from genesdev.cshlp.org on October 5, 2021 - Published by Cold Spring Harbor Laboratory Press id-gestational lethality in mice lacking eratin 8 H. Baribault, 1'4 J. Price, 2 K. Miyai, 3 and R.G. Oshima ~ ILa Jolla Cancer Research Foundation, La Jolla, California 92037 USA; 2Department of Immunology, Scripps Clinic Research Institute, La Jolla, California 92037 USA; 3Department of Pathology, University of California, San Diego, La Jolla, California 92093 USA. Keratin 8 (mK8) and its partner keratin 18 (mK18) are the first intermediate filament proteins expressed during mouse embryogenesis. They are found in most extraembryonic and embryonic simple epithelia, including trophectoderm, visceral yolk sac, gastrointestinal tract, lungs, mammary glands, and uterus. We report that a targeted null mutation in the inK8 gene causes mid-gestational lethality. Mutant embryos are growth retarded and suffer from internal bleeding, with an abnormal accumulation of erythrocytes in fetal livers. The mK8- phenotype has 94% penetrance, with a few mice surviving into adulthood. We suggest that mK8/mK18 filaments are important for the integrity of the fetal liver, like specialized human epidermal keratins for the integrity of the epidermis. This phenotype in mice differs from the reported function of simple epithelium keratins in Xenopus at the gastrulation stage. In mice, mK8 fulfills a vital function at 12 days postcoitum. [Key Words: Keratin 8; gene targeting; simple epithelium; fetal liver; intermediate filaments] Received March 11, 1993; revised version accepted April 22, 1993. The two types of keratins constitute the largest family of molysis bullosa simplex (Bonifas et al. 1991; Coulombe intermediate filament (IF) proteins. -

Novel Protein Pathways in Development and Progression of Pulmonary Sarcoidosis Maneesh Bhargava1*, K
www.nature.com/scientificreports OPEN Novel protein pathways in development and progression of pulmonary sarcoidosis Maneesh Bhargava1*, K. J. Viken1, B. Barkes2, T. J. Grifn3, M. Gillespie2, P. D. Jagtap3, R. Sajulga3, E. J. Peterson4, H. E. Dincer1, L. Li2, C. I. Restrepo2, B. P. O’Connor5, T. E. Fingerlin5, D. M. Perlman1 & L. A. Maier2 Pulmonary involvement occurs in up to 95% of sarcoidosis cases. In this pilot study, we examine lung compartment-specifc protein expression to identify pathways linked to development and progression of pulmonary sarcoidosis. We characterized bronchoalveolar lavage (BAL) cells and fuid (BALF) proteins in recently diagnosed sarcoidosis cases. We identifed 4,306 proteins in BAL cells, of which 272 proteins were diferentially expressed in sarcoidosis compared to controls. These proteins map to novel pathways such as integrin-linked kinase and IL-8 signaling and previously implicated pathways in sarcoidosis, including phagosome maturation, clathrin-mediated endocytic signaling and redox balance. In the BALF, the diferentially expressed proteins map to several pathways identifed in the BAL cells. The diferentially expressed BALF proteins also map to aryl hydrocarbon signaling, communication between innate and adaptive immune response, integrin, PTEN and phospholipase C signaling, serotonin and tryptophan metabolism, autophagy, and B cell receptor signaling. Additional pathways that were diferent between progressive and non-progressive sarcoidosis in the BALF included CD28 signaling and PFKFB4 signaling. Our studies demonstrate the power of contemporary proteomics to reveal novel mechanisms operational in sarcoidosis. Application of our workfows in well-phenotyped large cohorts maybe benefcial to identify biomarkers for diagnosis and prognosis and therapeutically tenable molecular mechanisms. -

Control of the Physical and Antimicrobial Skin Barrier by an IL-31–IL-1 Signaling Network
The Journal of Immunology Control of the Physical and Antimicrobial Skin Barrier by an IL-31–IL-1 Signaling Network Kai H. Ha¨nel,*,†,1,2 Carolina M. Pfaff,*,†,1 Christian Cornelissen,*,†,3 Philipp M. Amann,*,4 Yvonne Marquardt,* Katharina Czaja,* Arianna Kim,‡ Bernhard Luscher,€ †,5 and Jens M. Baron*,5 Atopic dermatitis, a chronic inflammatory skin disease with increasing prevalence, is closely associated with skin barrier defects. A cy- tokine related to disease severity and inhibition of keratinocyte differentiation is IL-31. To identify its molecular targets, IL-31–dependent gene expression was determined in three-dimensional organotypic skin models. IL-31–regulated genes are involved in the formation of an intact physical skin barrier. Many of these genes were poorly induced during differentiation as a consequence of IL-31 treatment, resulting in increased penetrability to allergens and irritants. Furthermore, studies employing cell-sorted skin equivalents in SCID/NOD mice demonstrated enhanced transepidermal water loss following s.c. administration of IL-31. We identified the IL-1 cytokine network as a downstream effector of IL-31 signaling. Anakinra, an IL-1R antagonist, blocked the IL-31 effects on skin differentiation. In addition to the effects on the physical barrier, IL-31 stimulated the expression of antimicrobial peptides, thereby inhibiting bacterial growth on the three-dimensional organotypic skin models. This was evident already at low doses of IL-31, insufficient to interfere with the physical barrier. Together, these findings demonstrate that IL-31 affects keratinocyte differentiation in multiple ways and that the IL-1 cytokine network is a major downstream effector of IL-31 signaling in deregulating the physical skin barrier. -
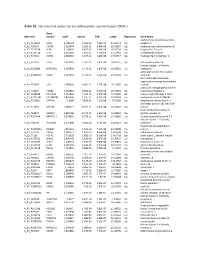
Table S1. the Statistical Metrics for Key Differentially Expressed Genes (Degs)
Table S1. The statistical metrics for key differentially expressed genes (DEGs) Gene Agilent Id Symbol logFC pValue FDR tvalue Regulation Gene Name oxidized low density lipoprotein A_24_P124624 OLR1 2.458429 1.19E-13 7.25E-10 24.04241 Up receptor 1 A_23_P90273 CHST8 2.622464 3.85E-12 6.96E-09 19.05867 Up carbohydrate sulfotransferase 8 A_23_P217528 KLF8 2.109007 4.85E-12 7.64E-09 18.76234 Up Kruppel like factor 8 A_23_P114740 CFH 2.651636 1.85E-11 1.79E-08 17.13652 Up complement factor H A_23_P34031 XAGE2 2.000935 2.04E-11 1.81E-08 17.02457 Up X antigen family member 2 A_23_P27332 TCF4 1.613097 2.32E-11 1.87E-08 16.87275 Up transcription factor 4 histone cluster 1 H1 family A_23_P250385 HIST1H1B 2.298658 2.47E-11 1.87E-08 16.80362 Up member b abnormal spindle microtubule A_33_P3288159 ASPM 2.162032 2.79E-11 2.01E-08 16.66292 Up assembly H19, imprinted maternally expressed transcript (non-protein A_24_P52697 H19 1.499364 4.09E-11 2.76E-08 16.23387 Up coding) potassium voltage-gated channel A_24_P31627 KCNB1 2.289689 6.65E-11 3.97E-08 15.70253 Up subfamily B member 1 A_23_P214168 COL12A1 2.155835 7.59E-11 4.15E-08 15.56005 Up collagen type XII alpha 1 chain A_33_P3271341 LOC388282 2.859496 7.61E-11 4.15E-08 15.55704 Up uncharacterized LOC388282 A_32_P150891 DIAPH3 2.2068 7.83E-11 4.22E-08 15.5268 Up diaphanous related formin 3 zinc finger protein 185 with LIM A_23_P11025 ZNF185 1.385721 8.74E-11 4.59E-08 15.41041 Up domain heat shock protein family B A_23_P96872 HSPB11 1.887166 8.94E-11 4.64E-08 15.38599 Up (small) member 11 A_23_P107454