MARIA GOEPPERT MAYER June 28,1906-February 20,1972
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Manhattan Project and Its Legacy
Transforming the Relationship between Science and Society: The Manhattan Project and Its Legacy Report on the workshop funded by the National Science Foundation held on February 14 and 15, 2013 in Washington, DC Table of Contents Executive Summary iii Introduction 1 The Workshop 2 Two Motifs 4 Core Session Discussions 6 Scientific Responsibility 6 The Culture of Secrecy and the National Security State 9 The Decision to Drop the Bomb 13 Aftermath 15 Next Steps 18 Conclusion 21 Appendix: Participant List and Biographies 22 Copyright © 2013 by the Atomic Heritage Foundation. All rights reserved. No part of this book, either text or illustration, may be reproduced or transmit- ted in any form by any means, electronic or mechanical, including photocopying, reporting, or by any information storage or retrieval system without written persmission from the publisher. Report prepared by Carla Borden. Design and layout by Alexandra Levy. Executive Summary The story of the Manhattan Project—the effort to develop and build the first atomic bomb—is epic, and it continues to unfold. The decision by the United States to use the bomb against Japan in August 1945 to end World War II is still being mythologized, argued, dissected, and researched. The moral responsibility of scientists, then and now, also has remained a live issue. Secrecy and security practices deemed necessary for the Manhattan Project have spread through the govern- ment, sometimes conflicting with notions of democracy. From the Manhattan Project, the scientific enterprise has grown enormously, to include research into the human genome, for example, and what became the Internet. Nuclear power plants provide needed electricity yet are controversial for many people. -

Catholic Christian Christian
Religious Scientists (From the Vatican Observatory Website) https://www.vofoundation.org/faith-and-science/religious-scientists/ Many scientists are religious people—men and women of faith—believers in God. This section features some of the religious scientists who appear in different entries on these Faith and Science pages. Some of these scientists are well-known, others less so. Many are Catholic, many are not. Most are Christian, but some are not. Some of these scientists of faith have lived saintly lives. Many scientists who are faith-full tend to describe science as an effort to understand the works of God and thus to grow closer to God. Quite a few describe their work in science almost as a duty they have to seek to improve the lives of their fellow human beings through greater understanding of the world around them. But the people featured here are featured because they are scientists, not because they are saints (even when they are, in fact, saints). Scientists tend to be creative, independent-minded and confident of their ideas. We also maintain a longer listing of scientists of faith who may or may not be discussed on these Faith and Science pages—click here for that listing. Agnesi, Maria Gaetana (1718-1799) Catholic Christian A child prodigy who obtained education and acclaim for her abilities in math and physics, as well as support from Pope Benedict XIV, Agnesi would write an early calculus textbook. She later abandoned her work in mathematics and physics and chose a life of service to those in need. Click here for Vatican Observatory Faith and Science entries about Maria Gaetana Agnesi. -

Bringing out the Dead Alison Abbott Reviews the Story of How a DNA Forensics Team Cracked a Grisly Puzzle
BOOKS & ARTS COMMENT DADO RUVIC/REUTERS/CORBIS DADO A forensics specialist from the International Commission on Missing Persons examines human remains from a mass grave in Tomašica, Bosnia and Herzegovina. FORENSIC SCIENCE Bringing out the dead Alison Abbott reviews the story of how a DNA forensics team cracked a grisly puzzle. uring nine sweltering days in July Bosnia’s Million Bones tells the story of how locating, storing, pre- 1995, Bosnian Serb soldiers slaugh- innovative DNA forensic science solved the paring and analysing tered about 7,000 Muslim men and grisly conundrum of identifying each bone the million or more Dboys from Srebrenica in Bosnia. They took so that grieving families might find some bones. It was in large them to several different locations and shot closure. part possible because them, or blew them up with hand grenades. This is an important book: it illustrates the during those fate- They then scooped up the bodies with bull- unspeakable horrors of a complex war whose ful days in July 1995, dozers and heavy earth-moving equipment, causes have always been hard for outsiders to aerial reconnais- and dumped them into mass graves. comprehend. The author, a British journalist, sance missions by the Bosnia’s Million It was the single most inhuman massacre has the advantage of on-the-ground knowl- Bones: Solving the United States and the of the Bosnian war, which erupted after the edge of the war and of the International World’s Greatest North Atlantic Treaty break-up of Yugoslavia and lasted from 1992 Commission on Missing Persons (ICMP), an Forensic Puzzle Organization had to 1995, leaving some 100,000 dead. -

Luis Alvarez: the Ideas Man
CERN Courier March 2012 Commemoration Luis Alvarez: the ideas man The years from the early 1950s to the late 1980s came alive again during a symposium to commemorate the birth of one of the great scientists and inventors of the 20th century. Luis Alvarez – one of the greatest experimental physicists of the 20th century – combined the interests of a scientist, an inventor, a detective and an explorer. He left his mark on areas that ranged from radar through to cosmic rays, nuclear physics, particle accel- erators, detectors and large-scale data analysis, as well as particles and astrophysics. On 19 November, some 200 people gathered at Berkeley to commemorate the 100th anniversary of his birth. Alumni of the Alvarez group – among them physicists, engineers, programmers and bubble-chamber film scanners – were joined by his collaborators, family, present-day students and admirers, as well as scientists whose professional lineage traces back to him. Hosted by the Lawrence Berkeley National Laboratory (LBNL) and the University of California at Berkeley, the symposium reviewed his long career and lasting legacy. A recurring theme of the symposium was, as one speaker put it, a “Shakespeare-type dilemma”: how could one person have accom- plished all of that in one lifetime? Beyond his own initiatives, Alvarez created a culture around him that inspired others to, as George Smoot put it, “think big,” as well as to “think broadly and then deep” and to take risks. Combined with Alvarez’s strong scientific standards and great care in execut- ing them, these principles led directly to the awarding of two Nobel Luis Alvarez celebrating the announcement of his 1968 Nobel prizes in physics to scientists at Berkeley – George Smoot in 2006 prize. -

Twenty Five Hundred Years of Small Science What’S Next?
Twenty Five Hundred Years of Small Science What’s Next? Lloyd Whitman Assistant Director for Nanotechnology White House Office of Science and Technology Policy Workshop on Integrated Nanosystems for Atomically Precise Manufacturing Berkeley, CA, August 5, 2015 Democritus (ca. 460 – 370 BC) Everything is composed of “atoms” Atomos (ἄτομος): that which can not be cut www.phil-fak.uni- duesseldorf.de/philo/galerie/antike/ demokrit.html Quantum Mechanics (1920s) Max Planck 1918* Albert Einstein 1921 Niels Bohr 1922 Louis de Broglie 1929 Max Born 1954 Paul Dirac 1933 On the Theory of Quanta Louis-Victor de Broglie Werner Heisenberg 1932 Wolfgang Pauli 1945 Erwin Schrödinger 1933 *Nobel Prizes in Physics https://tel.archives-ouvertes.fr/tel- 00006807 Ernst Ruska (1906 – 1988) Electron Microscopy Magnifying higher than the light microscope - 1933 Nobel Prize in Physics 1986 www.nobelprize.org/nobel_prizes/physics/laureates /1986/ruska-lecture.pdf Richard Feynman (1918-1988) There's Plenty of Room at the Bottom, An Invitation to Enter a New Field of Physics What would happen if we could arrange the atoms one by one the way we want them…? December 29, 1959 richard-feynman.net Heinrich Rohrer (1933 – 2013) Gerd Binnig Atomic resolution Scanning Tunneling Microscopy - 1981 1983 I could not stop looking at the images. It was like entering a new world. Gerd Binnig, Nobel lecture Binnig, et al., PRL 50, 120 (1983) Nobel Prize in Physics 1986 C60: Buckminsterfullerene Kroto, Heath, O‘Brien, Curl and September 1985 Smalley - 1985 …a remarkably stable cluster consisting of 60 carbon atoms…a truncated icosahedron. Nature 318, 162 (1985) http://www.acs.org/content/acs/en/education/whatis chemistry/landmarks/fullerenes.html Nobel Prize in Chemistry 1996 Curl, Kroto, and Smalley Positioning Single Atoms with a Scanning Tunnelling Microscope Eigler and Schweizer - 1990 …fabricate rudimentary structures of our own design, atom by atom. -
![I. I. Rabi Papers [Finding Aid]. Library of Congress. [PDF Rendered Tue Apr](https://docslib.b-cdn.net/cover/8589/i-i-rabi-papers-finding-aid-library-of-congress-pdf-rendered-tue-apr-428589.webp)
I. I. Rabi Papers [Finding Aid]. Library of Congress. [PDF Rendered Tue Apr
I. I. Rabi Papers A Finding Aid to the Collection in the Library of Congress Manuscript Division, Library of Congress Washington, D.C. 1992 Revised 2010 March Contact information: http://hdl.loc.gov/loc.mss/mss.contact Additional search options available at: http://hdl.loc.gov/loc.mss/eadmss.ms998009 LC Online Catalog record: http://lccn.loc.gov/mm89076467 Prepared by Joseph Sullivan with the assistance of Kathleen A. Kelly and John R. Monagle Collection Summary Title: I. I. Rabi Papers Span Dates: 1899-1989 Bulk Dates: (bulk 1945-1968) ID No.: MSS76467 Creator: Rabi, I. I. (Isador Isaac), 1898- Extent: 41,500 items ; 105 cartons plus 1 oversize plus 4 classified ; 42 linear feet Language: Collection material in English Location: Manuscript Division, Library of Congress, Washington, D.C. Summary: Physicist and educator. The collection documents Rabi's research in physics, particularly in the fields of radar and nuclear energy, leading to the development of lasers, atomic clocks, and magnetic resonance imaging (MRI) and to his 1944 Nobel Prize in physics; his work as a consultant to the atomic bomb project at Los Alamos Scientific Laboratory and as an advisor on science policy to the United States government, the United Nations, and the North Atlantic Treaty Organization during and after World War II; and his studies, research, and professorships in physics chiefly at Columbia University and also at Massachusetts Institute of Technology. Selected Search Terms The following terms have been used to index the description of this collection in the Library's online catalog. They are grouped by name of person or organization, by subject or location, and by occupation and listed alphabetically therein. -

Radiocarbon After Four Decades RADIOCARBON R.E
Radiocarbon After Four Decades RADIOCARBON R.E. Taylor A. Long R.S. Kra Editors Radiocarbon After Four Decades An Interdisciplinary Perspective With 148 Illustrations Springer Science+Business Media, LLC R.E. Thylor Austin Long Department of Anthropology Department of Geosciences Institute of Geophysics and Planetary Physics The University of Arizona University of California, Riverside Thscon, AZ 85721 USA Riverside, CA 92521-0418 USA Renee S. Kra Department of Geosciences The University of Arizona Thscon, AZ 85721 USA Library of Congress Cataloging-in-Publication Data Radiocarbon after four decades: an interdisciplinary perspective / [editors], R.E. Taylor, Austin Long, Renee S. Kra. p. cm. Includes bibliographical references and index. ISBN 978-1-4757-4251-0 ISBN 978-1-4757-4249-7 (eBook) DOI 1O.l007/978-1-4757-4249-7 1. Radiocarbon dating. I. Taylor, R. E. (Royal Ervin), 1938- II. Long, Austin. III. Kra, Renee S. QC798.D3R3 1992 546'.6815884-dc20 91-44448 Printed on acid-free paper. © 1992 Springer Science+BusinessMedia New York Originally published by Springer-Verlag New York, Inc. in 1992 Softcover reprint of the hardcover 1st edition 1992 All rights reserved. This work may not be translated or copied in whole or in part without the written per mission of the publisher,Springer Science+Bnsiness Media, LLC, except for brief excerpts in connection with reviews or scholarly analysis. Use in connection with any form of information storage and retrieval, electronic adaptation, computer software, or by similar or dissimilar methodology now known or hereaf ter developed is forbidden. The use of general descriptive names, trade names, trademarks, etc., in this publication, even if the former are not especially identified, is not to be taken as a sign that such names, as understood by the Trade Marks and Merchandise Marks Act, may accordingly be used freely by anyone. -

Enrico Fermi
Fermi, Enrico Inventors and Inventions Enrico Fermi Italian American physicist Fermi helped develop Fermi-Dirac statistics, which liceo (secondary school) and, on the advice of Amidei, elucidate the group behavior of elementary particles. joined the Scuola Normale Superiore at Pisa. This elite He also developed the theory of beta decay and college, attached to the University of Pisa, admitted only discovered neutron-induced artificial radioactivity. forty of Italy’s top students, who were given free board Finally, he succeeded in producing the first sustained and lodging. Fermi performed exceedingly well in the nuclear chain reaction, which led to the discovery highly competitive entrance exam. He completed his of nuclear energy and the development of the university education after only four years of research and atomic bomb. studies, receiving his Ph.D. in physics from the Univer- sity of Pisa and his undergraduate diploma from the Born: September 29, 1901; Rome, Italy Scuola Normale Superiore in July, 1922. He became Died: November 28, 1954; Chicago, Illinois an expert theoretical physicist and a talented exper- Primary field: Physics imentalist. This rare combination provided a solid foun- Primary inventions: Controlled nuclear chain dation for all his subsequent inventions. reaction; Fermi-Dirac statistics; theory of beta decay Life’s Work After postdoctoral work at the University of Göttingen, Early Life in Germany (1922-1923), and the University of Leiden, Enrico Fermi (ehn-REE-koh FUR-mee) was the third in the Netherlands (fall, 1924), Fermi took an interim po- child of Alberto Fermi and Ida de Gattis. Enrico was very sition at the University of Florence in December, 1924. -

Date: To: September 22, 1 997 Mr Ian Johnston©
22-SEP-1997 16:36 NOBELSTIFTELSEN 4& 8 6603847 SID 01 NOBELSTIFTELSEN The Nobel Foundation TELEFAX Date: September 22, 1 997 To: Mr Ian Johnston© Company: Executive Office of the Secretary-General Fax no: 0091-2129633511 From: The Nobel Foundation Total number of pages: olO MESSAGE DearMrJohnstone, With reference to your fax and to our telephone conversation, I am enclosing the address list of all Nobel Prize laureates. Yours sincerely, Ingr BergstrSm Mailing address: Bos StU S-102 45 Stockholm. Sweden Strat itddrtSMi Suircfatan 14 Teleptelrtts: (-MB S) 663 » 20 Fsuc (*-«>!) «W Jg 47 22-SEP-1997 16:36 NOBELSTIFTELSEN 46 B S603847 SID 02 22-SEP-1997 16:35 NOBELSTIFTELSEN 46 8 6603847 SID 03 Professor Willis E, Lamb Jr Prof. Aleksandre M. Prokhorov Dr. Leo EsaJki 848 North Norris Avenue Russian Academy of Sciences University of Tsukuba TUCSON, AZ 857 19 Leninskii Prospect 14 Tsukuba USA MSOCOWV71 Ibaraki Ru s s I a 305 Japan 59* c>io Dr. Tsung Dao Lee Professor Hans A. Bethe Professor Antony Hewlsh Department of Physics Cornell University Cavendish Laboratory Columbia University ITHACA, NY 14853 University of Cambridge 538 West I20th Street USA CAMBRIDGE CB3 OHE NEW YORK, NY 10027 England USA S96 014 S ' Dr. Chen Ning Yang Professor Murray Gell-Mann ^ Professor Aage Bohr The Institute for Department of Physics Niels Bohr Institutet Theoretical Physics California Institute of Technology Blegdamsvej 17 State University of New York PASADENA, CA91125 DK-2100 KOPENHAMN 0 STONY BROOK, NY 11794 USA D anni ark USA 595 600 613 Professor Owen Chamberlain Professor Louis Neel ' Professor Ben Mottelson 6068 Margarldo Drive Membre de rinstitute Nordita OAKLAND, CA 946 IS 15 Rue Marcel-Allegot Blegdamsvej 17 USA F-92190 MEUDON-BELLEVUE DK-2100 KOPENHAMN 0 Frankrike D an m ar k 599 615 Professor Donald A. -

Karl Herzfeld Retained Ties with His Family and with the German Physics Community by Occasional Visits to Germany
NATIONAL ACADEMY OF SCIENCES KARL FERDINAND HERZFELD 1892–1978 A Biographical Memoir by JOSEPH F. MULLIGAN Any opinions expressed in this memoir are those of the author and do not necessarily reflect the views of the National Academy of Sciences. Biographical Memoirs, VOLUME 80 PUBLISHED 2001 BY THE NATIONAL ACADEMY PRESS WASHINGTON, D.C. Courtesy of AIP Emilio Segrè Visual Archives, Physics Today Collection KARL FERDINAND HERZFELD February 24, 1892–June 3, 1978 BY JOSEPH F. MULLIGAN ARL F. HERZFELD, BORN in Vienna, Austria, studied at the Kuniversity there and at the universities in Zurich and Göttingen and took courses at the ETH (Technical Insti- tute) in Zurich before receiving his Ph.D. from the University of Vienna in 1914. In 1925, after four years in the Austro- Hungarian Army during World War I and five years as Privatdozent in Munich with Professors Arnold Sommerfeld and Kasimir Fajans, he was named extraordinary professor of theoretical physics at Munich University. A year later he accepted a visiting professorship in the United States at Johns Hopkins University in Baltimore, Maryland. This visiting position developed into a regular faculty appointment at Johns Hopkins, which he held until 1936. Herzfeld then moved to Catholic University of America in Washington, D.C., where he remained until his death in 1978. As physics chairman at Catholic University until 1961, Herzfeld built a small teaching-oriented department into a strong research department that achieved national renown for its programs in statistical mechanics, ultrasonics, and theoretical research on the structure of molecules, gases, liquids, and solids. During his career Herzfeld published about 140 research papers on physics and chemistry, wrote 3 4 BIOGRAPHICAL MEMOIRS two important books: Kinetische Theorie der Wärme (1925), and (with T. -
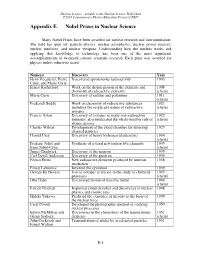
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

Chapter 2 Challenging the Boundaries Between Classical and Quantum Physics: the Case of Optical Dispersion Marta Jordi Taltavull
Chapter 2 Challenging the Boundaries between Classical and Quantum Physics: The Case of Optical Dispersion Marta Jordi Taltavull This paper describes one significant episode in the transition between classical and quantum theories. It analyzes the first theory of optical dispersion that ensued from the extension of Niels Bohr’s quantum model of the atom to other optical phenomena outside of spectroscopy. This theory was initially developed by Peter Debye in 1915 and then was endorsed and extended by Arnold Sommerfeld in 1915 and 1917. The most interesting aspect of the Debye-Sommerfeld theory for the present paper is that it clearly typifies important features of debates concerning the boundaries between classical and quantum physics, focusing on the period from 1913 to the early 1920s. Optical dispersion consisted of splitting white light into different colors be- cause of its change of velocity when passing through a transparent, prismatic medium. From the 1870s onward, it was well known that light was continuously dispersed across the entire spectrum, except at those specific frequencies, charac- teristic of the medium, at which light was completely absorbed. In other words, dispersion and absorption of light were complementary phenomena. From 1872, this behavior was explained using one enduring theoretical representation: the Mitschwingungen model. This model pictured the interaction between light and matter as a continuous process of interaction between waves and particles per- forming induced vibrations, called Mitschwingungen. In 1913, this model conflicted with certain aspects of Bohr’s quantum model of the atom. Contrary to the Mitschwingungen model, Bohr envisioned the ex- change of energy between light and matter as a discrete process, mediated by the emission or absorption of quanta of energy.