Ascidian News #76 December 2015
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Colonial Tunicates: Species Guide
SPECIES IN DEPTH Colonial Tunicates Colonial Tunicates Tunicates are small marine filter feeder animals that have an inhalant siphon, which takes in water, and an exhalant siphon that expels water once it has trapped food particles. Tunicates get their name from the tough, nonliving tunic formed from a cellulose-like material of carbohydrates and proteins that surrounds their bodies. Their other name, sea squirts, comes from the fact that many species will shoot LambertGretchen water out of their bodies when disturbed. Massively lobate colony of Didemnum sp. A growing on a rope in Sausalito, in San Francisco Bay. A colony of tunicates is comprised of many tiny sea squirts called zooids. These INVASIVE SEA SQUIRTS individuals are arranged in groups called systems, which form interconnected Star sea squirts (Botryllus schlosseri) are so named because colonies. Systems of these filter feeders the systems arrange themselves in a star. Zooids are shaped share a common area for expelling water like ovals or teardrops and then group together in small instead of having individual excurrent circles of about 20 individuals. This species occurs in a wide siphons. Individuals and systems are all variety of colors: orange, yellow, red, white, purple, grayish encased in a matrix that is often clear and green, or black. The larvae each have eight papillae, or fleshy full of blood vessels. All ascidian tunicates projections that help them attach to a substrate. have a tadpole-like larva that swims for Chain sea squirts (Botryloides violaceus) have elongated, less than a day before attaching itself to circular systems. Each system can have dozens of zooids. -

Settlement Patterns in Ascidians Concerning Have Been
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Digital.CSIC Larval settlement behaviour in six gregarious ascidians in relation to adult 2 distribution 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 Marc Rius1,2,*, George M. Branch2, Charles L. Griffiths1,2, Xavier Turon3 18 19 20 1 Centre for Invasion Biology, Zoology Department, University of Cape Town, 21 Rondebosch 7701, South Africa 22 23 2 Marine Biology Research Centre, Zoology Department, University of Cape Town, 24 Rondebosch 7701, South Africa 25 26 3 Center for Advanced Studies of Blanes (CEAB, CSIC), Accés Cala St. Francesc 14, 27 17300 Blanes (Girona), Spain 28 29 30 31 32 33 34 * Corresponding author: Marc Rius 35 Centre for Invasion Biology, Zoology Department, University of Cape Town, 36 Rondebosch 7701, South Africa 37 E-mail: [email protected] 38 Telephone: +27 21 650 4939 39 Fax: +27 21 650 3301 40 41 Running head: Settlement patterns of gregarious ascidians 42 43 44 1 45 ABSTRACT 46 Settlement influences the distribution and abundance of many marine organisms, 47 although the relative roles of abiotic and biotic factors influencing settlement are poorly 48 understood. Species that aggregate often owe this to larval behaviour, and we ask 49 whether this predisposes ascidians to becoming invasive, by increasing their capacity to 50 maintain their populations. We explored the interactive effects of larval phototaxis and 51 geotaxis and conspecific adult extracts on settlement rates of a representative suite of 52 six species of ascidians that form aggregations in the field, including four aliens with 53 global distributions, and how they relate to adult habitat characteristics. -

Ciona Intestinalis and Comparative Analysis with Ciona Savignyi
Downloaded from genome.cshlp.org on September 24, 2021 - Published by Cold Spring Harbor Laboratory Press Methods Diploid genome reconstruction of Ciona intestinalis and comparative analysis with Ciona savignyi Jong Hyun Kim,1,4 Michael S. Waterman,2,3 and Lei M. Li2,3 1Department of Computer Science, Yonsei University, Seoul, 120-749, Republic of Korea; 2Molecular and Computational Biology Program, Department of Biological Sciences, University of Southern California, Los Angeles, California 90089, USA; 3Department of Mathematics, University of Southern California, Los Angeles, California 90089, USA One of the main goals in genome sequencing projects is to determine a haploid consensus sequence even when clone libraries are constructed from homologous chromosomes. However, it has been noticed that haplotypes can be inferred from genome assemblies by investigating phase conservation in sequenced reads. In this study, we seek to infer haplotypes, a diploid consensus sequence, from the genome assembly of an organism, Ciona intestinalis. The Ciona intestinalis genome is an ideal resource from which haplotypes can be inferred because of the high polymorphism rate (1.2%). The haplotype estimation scheme consists of polymorphism detection and phase estimation. The core step of our method is a Gibbs sampling procedure. The mate-pair information from two-end sequenced clone inserts is exploited to provide long-range continuity. We estimate the polymorphism rate of Ciona intestinalis to be 1.2% and 1.5%, according to two different polymorphism counting schemes. The distribution of heterozygosity number is well fit by a compound Poisson distribution. The N50 length of haplotype segments is 37.9 kb in our assembly, while the N50 scaffold length of the Ciona intestinalis assembly is 190 kb. -
Appendicularia of CTAW
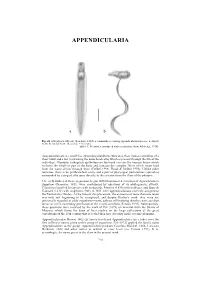
APPENDICULARIA APPENDICULARIA a b Fig. 26. Oikopleura albicans (Leuckart, 1854), a commonly occurring appendicularian species: a, dorsal view; b, lateral view. (Scale bar = 0.1 mm). [after T. Prentiss, reproduced with permission, from Alldredge 1976] Appendicularians are small free swimming planktonic tunicates, their bodies consisting of a short trunk and a tail (containing the notochord cells) which is present through the life of the individual. Glandular (oikoplast) epithelium on the trunk secretes the mucous house which encloses the whole or part of the body and contains the complex filters which strain food from the water driven through them (Deibel 1998; Flood & Deibel 1998). Unlike other tunicates, there is no peribranchial cavity and a pair of pharyngeal perforations (spiracles) surrounded by a ring of cilia open directly to the exterior from the floor of the pharynx. The early studies of these organisms, begun with Chamisso's description of Appendicularia flagellum Chamisso, 1821, were confounded by questions of its phylogenetic affinity. Chamisso classified his species with medusoids, Mertens (1830) with molluscs, and Quoy & Gaimard (1833) with zoophytes. Only in 1851 were appendicularians correctly assigned to the Tunicata by Huxley. At the time of this placement, the existence of more than one taxon was only just beginning to be recognised, and despite Huxley's work, they were not universally regarded as adult organisms—some authors still insisting that they were ascidian larvae or a free swimming generation of the sessile ascidians (Fenaux 1993). Subsequently, these questions were resolved by the work of Fol (1872) on material from the Straits of Messina, which forms the basis of later studies on the large collections of the great expeditions of the 19th century that revealed their true diversity in the oceanic plankton. -

Ascidiacea, Phlebobranchia, Corellidae) in the Southern Hemisphere with Description of a New Species
Zootaxa 3702 (2): 135–149 ISSN 1175-5326 (print edition) www.mapress.com/zootaxa/ Article ZOOTAXA Copyright © 2013 Magnolia Press ISSN 1175-5334 (online edition) http://dx.doi.org/10.11646/zootaxa.3702.2.3 http://zoobank.org/urn:lsid:zoobank.org:pub:E972F88B-7981-4F38-803D-8F4F92FE6A37 The genus Corella (Ascidiacea, Phlebobranchia, Corellidae) in the Southern Hemisphere with description of a new species FRANÇOISE MONNIOT Muséum national d’Histoire naturelle, 57 rue Cuvier Fr 75231 Paris cedex 05, France.E-mail : [email protected] Abstract In the Southern Hemisphere the species attributed to Corella eumyota, Traustedt, 1882 are likely more varied than previously expected. This ascidian species was described from specimens collected at Valparaiso (Chile). Until now it was considered as a widely distributed species in the southern hemisphere. New collections from Chile and the Antarctic area have allowed to separate two species and re-establish Corella antarctica Sluiter, 1905 as a valid species (Alurralde 2013).A morphological re- examination of many specimens from the MNHN collections and especially recent surveys as CEAMARC and REVOLTA confirms that Antarctic specimens from the Antarctic Peninsula and Terre Adélie obviously differ from sub-Antarctic material more varied than previously estimated. On the other hand, C. eumyota invasive in Europe (Lambert 2004) has been shown to be the same as specimens from Chile, New Zealand and other sub-Antarctic regions. The present morphological study compares Corella from different regions and describes a new species Corella brewinae n. sp that is found living mixed with C. eumyota populations. Key words: Ascidians, Corellidae, Antarctic, Sub-Antarctic, new species Introduction The genus Corella was created by Hancock (1870) for Ascidia parallelogramma Müller, 1776. -

Brains of Primitive Chordates 439
Brains of Primitive Chordates 439 Brains of Primitive Chordates J C Glover, University of Oslo, Oslo, Norway although providing a more direct link to the evolu- B Fritzsch, Creighton University, Omaha, NE, USA tionary clock, is nevertheless hampered by differing ã 2009 Elsevier Ltd. All rights reserved. rates of evolution, both among species and among genes, and a still largely deficient fossil record. Until recently, it was widely accepted, both on morpholog- ical and molecular grounds, that cephalochordates Introduction and craniates were sister taxons, with urochordates Craniates (which include the sister taxa vertebrata being more distant craniate relatives and with hemi- and hyperotreti, or hagfishes) represent the most chordates being more closely related to echinoderms complex organisms in the chordate phylum, particu- (Figure 1(a)). The molecular data only weakly sup- larly with respect to the organization and function of ported a coherent chordate taxon, however, indicat- the central nervous system. How brain complexity ing that apparent morphological similarities among has arisen during evolution is one of the most chordates are imposed on deep divisions among the fascinating questions facing modern science, and it extant deuterostome taxa. Recent analysis of a sub- speaks directly to the more philosophical question stantially larger number of genes has reversed the of what makes us human. Considerable interest has positions of cephalochordates and urochordates, pro- therefore been directed toward understanding the moting the latter to the most closely related craniate genetic and developmental underpinnings of nervous relatives (Figure 1(b)). system organization in our more ‘primitive’ chordate relatives, in the search for the origins of the vertebrate Comparative Appearance of Brains, brain in a common chordate ancestor. -

Phlebobranchia of CTAW
PHLEBOBRANCHIA PHLEBOBRANCHIA The suborder Phlebobranchia (order Enterogona) is characterised by having unpaired gonads present only on the same side of the body as the gut. As in Stolidobranchia, the body is not divided into different sections (such as thorax, abdomen and posterior abdomen) as the gut is folded up in the parietal body wall outside the pharynx and the large branchial sac occupies the whole length of the body. Usually the branchial sac (which is flat, without folds) has internal longitudinal vessels (although only vestiges remain in Agneziidae). Epicardial sacs do not persist in adults as they do in Aplousobranchia, although excretory vesicles (nephrocytes) embedded in the body wall over the gut are known to originate from the embryonic epicardium in Ascidiidae and Corellidae. Most phlebobranchs are solitary. However, Plurellidae Kott, 1973 includes both solitary and colonial forms, and Perophoridae Giard, 1872 are all colonial. Replication in Perophoridae is from ectodermal epithelium (rather than endodermal or mesodermal tissue the mesodermal tissue of the vascular stolon (rather than the endodermal tissue as in most as in Aplousobranchia). The process of replication has not been investigated in Plurellidae. Phlebobranch taxa occurring in Australia are documented in Kott (1985). Family level taxa are characterised principally by the size and form of the branchial sac including the number of branchial vessels and form of the stigmata; the form, size and position of the gonads; and the habit (colonial or solitary) of the taxon. Berrill (1950) has discussed problems in assessing the phylogeny of Perophoridae. References Berrill, N.J. (1950). The Tunicata. Ray Soc. Publs 133: 1–354 Giard, A.M. -

Settlement Patterns in Ascidians Concerning Have Been Patchily
Larval settlement behaviour in six gregarious ascidians in relation to adult 2 distribution 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 Marc Rius1,2,*, George M. Branch2, Charles L. Griffiths1,2, Xavier Turon3 18 19 20 1 Centre for Invasion Biology, Zoology Department, University of Cape Town, 21 Rondebosch 7701, South Africa 22 23 2 Marine Biology Research Centre, Zoology Department, University of Cape Town, 24 Rondebosch 7701, South Africa 25 26 3 Center for Advanced Studies of Blanes (CEAB, CSIC), Accés Cala St. Francesc 14, 27 17300 Blanes (Girona), Spain 28 29 30 31 32 33 34 * Corresponding author: Marc Rius 35 Centre for Invasion Biology, Zoology Department, University of Cape Town, 36 Rondebosch 7701, South Africa 37 E-mail: [email protected] 38 Telephone: +27 21 650 4939 39 Fax: +27 21 650 3301 40 41 Running head: Settlement patterns of gregarious ascidians 42 43 44 1 45 ABSTRACT 46 Settlement influences the distribution and abundance of many marine organisms, 47 although the relative roles of abiotic and biotic factors influencing settlement are poorly 48 understood. Species that aggregate often owe this to larval behaviour, and we ask 49 whether this predisposes ascidians to becoming invasive, by increasing their capacity to 50 maintain their populations. We explored the interactive effects of larval phototaxis and 51 geotaxis and conspecific adult extracts on settlement rates of a representative suite of 52 six species of ascidians that form aggregations in the field, including four aliens with 53 global distributions, and how they relate to adult habitat characteristics. -

Cionin, a Vertebrate Cholecystokinin/Gastrin
www.nature.com/scientificreports OPEN Cionin, a vertebrate cholecystokinin/gastrin homolog, induces ovulation in the ascidian Ciona intestinalis type A Tomohiro Osugi, Natsuko Miyasaka, Akira Shiraishi, Shin Matsubara & Honoo Satake* Cionin is a homolog of vertebrate cholecystokinin/gastrin that has been identifed in the ascidian Ciona intestinalis type A. The phylogenetic position of ascidians as the closest living relatives of vertebrates suggests that cionin can provide clues to the evolution of endocrine/neuroendocrine systems throughout chordates. Here, we show the biological role of cionin in the regulation of ovulation. In situ hybridization demonstrated that the mRNA of the cionin receptor, Cior2, was expressed specifcally in the inner follicular cells of pre-ovulatory follicles in the Ciona ovary. Cionin was found to signifcantly stimulate ovulation after 24-h incubation. Transcriptome and subsequent Real-time PCR analyses confrmed that the expression levels of receptor tyrosine kinase (RTK) signaling genes and a matrix metalloproteinase (MMP) gene were signifcantly elevated in the cionin-treated follicles. Of particular interest is that an RTK inhibitor and MMP inhibitor markedly suppressed the stimulatory efect of cionin on ovulation. Furthermore, inhibition of RTK signaling reduced the MMP gene expression in the cionin-treated follicles. These results provide evidence that cionin induces ovulation by stimulating MMP gene expression via the RTK signaling pathway. This is the frst report on the endogenous roles of cionin and the induction of ovulation by cholecystokinin/gastrin family peptides in an organism. Ascidians are the closest living relatives of vertebrates in the Chordata superphylum, and thus they provide important insights into the evolution of peptidergic systems in chordates. -

From the National Park La Restinga, Isla Margarita, Venezuela
Biota Neotrop., vol. 10, no. 1 Inventory of ascidians (Tunicata, Ascidiacea) from the National Park La Restinga, Isla Margarita, Venezuela Rosana Moreira Rocha1,11, Edlin Guerra-Castro2, Carlos Lira3, Sheila Marquez Pauls4, Ivan Hernández5, Adriana Pérez3, Adriana Sardi6, Jeannette Pérez6, César Herrera6, Ana Karinna Carbonini7, Virginia Caraballo3, Dioceline Salazar8, Maria Cristina Diaz9 & Juan José Cruz-Motta6,10 1 Departamento de Zoologia, Universidade Federal do Paraná – UFPR, CP 19020, CEP 82531-980 Curitiba, PR, Brasil 2Centro de Ecología, Instituto Venezolano de Investigaciones Científicas, CP 21827, Caracas 1020-A, Venezuela, e-mail: [email protected] 3Laboratorio de Zoología, Universidad de Oriente, Núcleo de Nueva Esparta, Escuela de Ciencias Aplicadas del Mar, CP 658, Porlamar 6301, Isla Margarita, Venezuela, e-mail: [email protected], [email protected], [email protected] 4Instituto de Zoologia Tropical, Escuela de Biologia, Universidad Central de Venezuela, CP 47058, Caracas 1041, Venezuela, e-mail: [email protected] 5Departamento de Ciencias, Universidad de Oriente, Núcleo de Nueva Esparta, Guatamara, Isla de Margarita, Venezuela, e-mail: [email protected] 6Laboratorio de Ecología Experimental, Universidad Simón Bolívar, CP 89000, Sartenejas, Caracas 1080, Venezuela, e-mail: [email protected], [email protected], [email protected] 7Laboratorio de Biología Marina, Universidad Simón Bolívar, CP 89000, Sartenejas, Caracas 1080, Venezuela, e-mail: [email protected] 8Departamento de Biología, Escuela de Ciencias, Universidad de Oriente, Núcleo de Sucre, CP 245, CEP 6101,Cumaná, Venezuela, e-mail: [email protected] 9Museo Marino de Margarita, Bulevar El Paseo, Boca del Río, Margarita, Edo. Nueva Esparta, Venezuela, e-mail: [email protected] 10Departamento de Estudios Ambientales, Universidad Simón Bolívar, CP 89000, Sartenejas, Caracas 1080, Venezuela, e-mail: [email protected] 11Corresponding author: Rosana Moreira Rocha, e-mail: [email protected] ROCHA, R.M., GUERRA-CASTRO, E., LIRA, C., PAUL, S.M., HERNÁNDEZ. -

Marine Biology
Marine Biology Spatial and temporal dynamics of ascidian invasions in the continental United States and Alaska. --Manuscript Draft-- Manuscript Number: MABI-D-16-00297 Full Title: Spatial and temporal dynamics of ascidian invasions in the continental United States and Alaska. Article Type: S.I. : Invasive Species Keywords: ascidians, biofouling, biogeography, marine invasions, nonindigenous, non-native species, North America Corresponding Author: Christina Simkanin, Phd Smithsonian Environmental Research Center Edgewater, MD UNITED STATES Corresponding Author Secondary Information: Corresponding Author's Institution: Smithsonian Environmental Research Center Corresponding Author's Secondary Institution: First Author: Christina Simkanin, Phd First Author Secondary Information: Order of Authors: Christina Simkanin, Phd Paul W. Fofonoff Kristen Larson Gretchen Lambert Jennifer Dijkstra Gregory M. Ruiz Order of Authors Secondary Information: Funding Information: California Department of Fish and Wildlife Dr. Gregory M. Ruiz National Sea Grant Program Dr. Gregory M. Ruiz Prince William Sound Regional Citizens' Dr. Gregory M. Ruiz Advisory Council Smithsonian Institution Dr. Gregory M. Ruiz United States Coast Guard Dr. Gregory M. Ruiz United States Department of Defense Dr. Gregory M. Ruiz Legacy Program Abstract: SSpecies introductions have increased dramatically in number, rate, and magnitude of impact in recent decades. In marine systems, invertebrates are the largest and most diverse component of coastal invasions throughout the world. Ascidians are conspicuous and well-studied members of this group, however, much of what is known about their invasion history is limited to particular species or locations. Here, we provide a large-scale assessment of invasions, using an extensive literature review and standardized field surveys, to characterize the invasion dynamics of non-native ascidians in the continental United States and Alaska. -

Ciona Intestinalis
UC Berkeley UC Berkeley Previously Published Works Title Unraveling genomic regulatory networks in the simple chordate, Ciona intestinalis Permalink https://escholarship.org/uc/item/6531j4jb Journal Genome Research, 15(12) ISSN 1088-9051 Authors Shi, Weiyang Levine, Michael Davidson, Brad Publication Date 2005-12-01 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Perspective Unraveling genomic regulatory networks in the simple chordate, Ciona intestinalis Weiyang Shi, Michael Levine, and Brad Davidson1 Department of Molecular and Cell Biology, Division of Genetics and Development, Center for Integrative Genomics, University of California, Berkeley, California 94720, USA The draft genome of the primitive chordate, Ciona intestinalis, was published three years ago. Since then, significant progress has been made in utilizing Ciona’s genomic and morphological simplicity to better understand conserved chordate developmental processes. Extensive annotation and sequencing of staged EST libraries make the Ciona genome one of the best annotated among those that are publicly available. The formation of the Ciona tadpole depends on simple, well-defined cellular lineages, and it is possible to trace the lineages of key chordate tissues such as the notochord and neural tube to the fertilized egg. Electroporation methods permit the targeted expression of regulatory genes and signaling molecules in defined cell lineages, as well as the rapid identification of regulatory DNAs underlying cell-specific gene expression. The recent sequencing of a second Ciona genome (C. savignyi) permits the use of simple alignment algorithms for the identification of conserved noncoding sequences, including microRNA genes and enhancers. Detailed expression profiles are now available for almost every gene that encodes a regulatory protein or cell-signaling molecule.