Primary Immature Teratoma of the Thigh Fig
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genetic Markers in Lung Cancer Diagnosis: a Review
International Journal of Molecular Sciences Review Genetic Markers in Lung Cancer Diagnosis: A Review Katarzyna Wadowska 1 , Iwona Bil-Lula 1 , Łukasz Trembecki 2,3 and Mariola Sliwi´ ´nska-Mosso´n 1,* 1 Department of Medical Laboratory Diagnostics, Division of Clinical Chemistry and Laboratory Haematology, Wroclaw Medical University, 50-556 Wroclaw, Poland; [email protected] (K.W.); [email protected] (I.B.-L.) 2 Department of Radiation Oncology, Lower Silesian Oncology Center, 53-413 Wroclaw, Poland; [email protected] 3 Department of Oncology, Faculty of Medicine, Wroclaw Medical University, 53-413 Wroclaw, Poland * Correspondence: [email protected]; Tel.: +48-71-784-06-30 Received: 1 June 2020; Accepted: 25 June 2020; Published: 27 June 2020 Abstract: Lung cancer is the most often diagnosed cancer in the world and the most frequent cause of cancer death. The prognosis for lung cancer is relatively poor and 75% of patients are diagnosed at its advanced stage. The currently used diagnostic tools are not sensitive enough and do not enable diagnosis at the early stage of the disease. Therefore, searching for new methods of early and accurate diagnosis of lung cancer is crucial for its effective treatment. Lung cancer is the result of multistage carcinogenesis with gradually increasing genetic and epigenetic changes. Screening for the characteristic genetic markers could enable the diagnosis of lung cancer at its early stage. The aim of this review was the summarization of both the preclinical and clinical approaches in the genetic diagnostics of lung cancer. The advancement of molecular strategies and analytic platforms makes it possible to analyze the genome changes leading to cancer development—i.e., the potential biomarkers of lung cancer. -

Ovarian Cancer and Cervical Cancer
What Every Woman Should Know About Gynecologic Cancer R. Kevin Reynolds, MD The George W. Morley Professor & Chief, Division of Gyn Oncology University of Michigan Ann Arbor, MI What is gynecologic cancer? Cancer is a disease where cells grow and spread without control. Gynecologic cancers begin in the female reproductive organs. The most common gynecologic cancers are endometrial cancer, ovarian cancer and cervical cancer. Less common gynecologic cancers involve vulva, Fallopian tube, uterine wall (sarcoma), vagina, and placenta (pregnancy tissue: molar pregnancy). Ovary Uterus Endometrium Cervix Vagina Vulva What causes endometrial cancer? Endometrial cancer is the most common gynecologic cancer: one out of every 40 women will develop endometrial cancer. It is caused by too much estrogen, a hormone normally present in women. The most common cause of the excess estrogen is being overweight: fat cells actually produce estrogen. Another cause of excess estrogen is medication such as tamoxifen (often prescribed for breast cancer treatment) or some forms of prescribed estrogen hormone therapy (unopposed estrogen). How is endometrial cancer detected? Almost all endometrial cancer is detected when a woman notices vaginal bleeding after her menopause or irregular bleeding before her menopause. If bleeding occurs, a woman should contact her doctor so that appropriate testing can be performed. This usually includes an endometrial biopsy, a brief, slightly crampy test, performed in the office. Fortunately, most endometrial cancers are detected before spread to other parts of the body occurs Is endometrial cancer treatable? Yes! Most women with endometrial cancer will undergo surgery including hysterectomy (removal of the uterus) in addition to removal of ovaries and lymph nodes. -

Invasive Cervical Cancer Audit; EU Guidelines for Quality Assurance
The 4th EFCS Annual Tutorial Ospedale Universitario di Cattinara, Strada di Fiume, Trieste Handouts for lectures and workshops – I I - Gynaecological cytopathology Mrs Rietje Salet‐van‐de Pol, Radboud University Nijmegen Medical Centre, Nijmegen, The Netherlands Gynecological cytology: technical aspects ............................................................................... 2 Non‐neoplastic gynecological cytology .................................................................................... 6 • Dr Giovanni Negri, General Hospital of Bolzano, Bozano SIL and cancer; ASC‐US, ASC‐H, diagnostic pitfalls and look‐alikes; glandular abnormalities 11 • Dr Amanda Herbert, Guy’s & St Thomas’ NHS Foundation Trust, London Invasive cervical cancer audit; EU guidelines for quality assurance ...................................... 17 1 Gynecological cytology: technical aspects Rietje Salet-van de Pol Important in specimen processing is to obtain as much as possible well preserved cells for microscopically evaluation. The quality of the smear depends on cell sampling, fixation and staining. For obtaining enough cervical material you are dependent on the cell sampler. For cervical cytology two types of specimen are available: conventional smears and liquid based cytology (LBC). Conventional, Thinprep and Surepath slides In conventional cytology the cell sampler makes the smear and is responsible for the fixation of the cells. Reasons for unsatisfactory conventional smears can be obscuring blood or inflammatory cells, thick smears with overlapping cells, poor preservation of the cells due to late fixation and low cellularity. In LBC the cell sampler immediately transferred the cellular material into a vial with fixative (fixating solution) which gives a better preservation of the cells. The laboratory is responsible for processing of the smear. LBC gives equally distribution of the cells in a thin cell layer of well preserved cells. The rate of unsatisfactory smears is lower. -
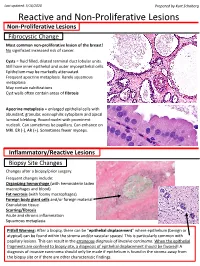
Reactive and Non-Proliferative Lesions
Last updated: 5/16/2020 Prepared by Kurt Schaberg Reactive and Non-Proliferative Lesions Non-Proliferative Lesions Fibrocystic Change Most common non-proliferative lesion of the breast! No significant increased risk of cancer. Cysts = fluid filled, dilated terminal duct lobular units. Still have inner epithelial and outer myoepithelial cells. Epithelium may be markedly attenuated. Frequent apocrine metaplasia. Rarely squamous metaplasia May contain calcifications Cyst walls often contain areas of fibrosis Apocrine metaplasia = enlarged epithelial cells with abundant, granular, eosinophilic cytoplasm and apical luminal blebbing. Round nuclei with prominent nucleoli. Can sometimes be papillary. Can enhance on MRI. ER (-), AR (+). Sometimes fewer myoeps. Inflammatory/Reactive Lesions Biopsy Site Changes Changes after a biopsy/prior surgery. Frequent changes include: Organizing hemorrhage (with hemosiderin laden macrophages and blood) Fat necrosis (with foamy macrophages) Foreign body giant cells and/or foreign material Granulation tissue Scarring/fibrosis Acute and chronic inflammation Squamous metaplasia Pitfall Warning: After a biopsy, there can be “epithelial displacement” where epithelium (benign or atypical) can be found within the stroma and/or vascular spaces! This is particularly common with papillary lesions. This can result in the erroneous diagnosis of invasive carcinoma. When the epithelial fragments are confined to biopsy site, a diagnosis of epithelial displacement should be favored! A diagnosis of invasive carcinoma should -

Squamous Cell Carcinoma Arising in an Ovarian Mature Cystic Teratoma
Case Report Obstet Gynecol Sci 2013;56(2):121-125 http://dx.doi.org/10.5468/OGS.2013.56.2.121 pISSN 2287-8572 · eISSN 2287-8580 Squamous cell carcinoma arising in an ovarian mature cystic teratoma complicating pregnancy Nae-Ri Yun1, Jung-Woo Park1, Min-Kyung Hyun1, Jee-Hyun Park1, Suk-Jin Choi2, Eunseop Song1 Departments of 1Obstetrics and Gynecology and 2Pathology, Inha University College of Medicine, Incheon, Korea Mature cystic teratomas of the ovary (MCT) are usually observed in women of reproductive age with the most dreadful complication being malignant transformation which occurs in approximately 1% to 3% of MCTs. In this case report, we present a patient with squamous cell carcinoma which developed from a MCT during pregnancy. The patient was treated conservatively without adjuvant chemotherapy and was followed without evidence of disease for more than 60 months using conventional tools as well as positron emission tomography-computed tomography following the initial surgery. We report this case along with the review of literature. Keywords: Dermoid cyst; Malignant transformation; Observation; Positron emission tomography-computed tomography Introduction An 18 cm solid and cystic left ovarian mass with a smooth surface and two small right ovarian cysts were detected re- The incidence of adnexal masses during pregnancy is 1% to sulting in a laparotomy at 13 weeks of gestation and left sal- 9% [1]. Mature cystic teratomas (MCT) are common during pingo-oophorectomy and right ovarian cystectomy (Fig. 1C). pregnancy with the most dreadful complication being ma- The report of the frozen section from both tissues revealed lignant transformation which occurs in approximately 1% to MCT. -

Pembrolizumab in Vaginal and Vulvar Squamous Cell Carcinoma: a Case Series from a Phase II Basket Trial Jefrey A
www.nature.com/scientificreports OPEN Pembrolizumab in vaginal and vulvar squamous cell carcinoma: a case series from a phase II basket trial Jefrey A. How 1, Amir A. Jazaeri 1, Pamela T. Soliman1, Nicole D. Fleming1, Jing Gong2, Sarina A. Piha‑Paul2, Filip Janku 2, Bettzy Stephen 2 & Aung Naing 2* Vaginal and vulvar squamous cell carcinoma (SCC) are rare tumors that can be challenging to treat in the recurrent or metastatic setting. We present a case series of patients with vaginal or vulvar SCC who were treated with single‑agent pembrolizumab as part of a phase II basket clinical trial to evaluate efcacy and safety. Two cases of recurrent and metastatic vaginal SCC, with multiple prior lines of systemic chemotherapy and radiation, received pembrolizumab. One patient had signifcant reduction (81%) in target tumor lesions prior to treatment discontinuation at cycle 10 following confrmed progression of disease with new metastatic lesions (stable disease by irRECIST criteria). In contrast, the other patient with vaginal SCC discontinued treatment after cycle 3 due to disease progression. Both patients had PD‑L1 positive vaginal tumors and tolerated treatment well. One case of recurrent vulvar SCC with multiple surgical resections and prior progression on systemic carboplatin had a 30% reduction in her target tumor lesions following pembrolizumab treatment with a PD‑L1 positive tumor. Treatment was discontinued for grade 3 mucositis after cycle 5. Pembrolizumab may provide some clinical beneft to some patients with vaginal or vulvar SCC and is overall safe to utilize in this population. Future studies are needed to evaluate the efcacy of pembrolizumab in these rare tumor types and to identify predictive biomarkers of response. -

Influence of Age on Histologic Outcome of Cervical Intraepithelial Neoplasia
www.nature.com/scientificreports OPEN Infuence of age on histologic outcome of cervical intraepithelial neoplasia during observational Received: 10 May 2017 Accepted: 6 April 2018 management: results from large Published: xx xx xxxx cohort, systematic review, meta- analysis Christine Bekos1, Richard Schwameis1, Georg Heinze2, Marina Gärner1, Christoph Grimm1, Elmar Joura1,4, Reinhard Horvat3, Stephan Polterauer1,4 & Mariella Polterauer1 Aim of this study was to investigate the histologic outcome of cervical intraepithelial neoplasia (CIN) during observational management. Consecutive women with histologically verifed CIN and observational management were included. Histologic fndings of initial and follow-up visits were collected and persistence, progression and regression rates at end of observational period were assessed. Uni- and multivariate analyses were performed. A systematic review of the literature and meta-analysis was performed. In 783 women CIN I, II, and III was diagnosed by colposcopically guided biopsy in 42.5%, 26.6% and 30.9%, respectively. Younger patients had higher rates of regression (p < 0.001) and complete remission (< 0.001) and lower rates of progression (p = 0.003). Among women aged < 25, 25 < 30, 30 < 35, 35 < 40 years, and > 40 years, regression rates were 44.7%, 33.7%, 30.9%, 27.3%, and 24.9%, respectively. Pooled analysis of published data showed similar results. Multivariable analysis showed that with each fve years of age, the odds for regression reduced by 21% (p < 0.001) independently of CIN grade (p < 0.001), and presence of HPV high-risk infection (p < 0.001). Patient’s age has a considerable infuence on the natural history of CIN – independent of CIN grade and HPV high- risk infection. -

Clinical Radiation Oncology Review
Clinical Radiation Oncology Review Daniel M. Trifiletti University of Virginia Disclaimer: The following is meant to serve as a brief review of information in preparation for board examinations in Radiation Oncology and allow for an open-access, printable, updatable resource for trainees. Recommendations are briefly summarized, vary by institution, and there may be errors. NCCN guidelines are taken from 2014 and may be out-dated. This should be taken into consideration when reading. 1 Table of Contents 1) Pediatrics 6) Gastrointestinal a) Rhabdomyosarcoma a) Esophageal Cancer b) Ewings Sarcoma b) Gastric Cancer c) Wilms Tumor c) Pancreatic Cancer d) Neuroblastoma d) Hepatocellular Carcinoma e) Retinoblastoma e) Colorectal cancer f) Medulloblastoma f) Anal Cancer g) Epndymoma h) Germ cell, Non-Germ cell tumors, Pineal tumors 7) Genitourinary i) Craniopharyngioma a) Prostate Cancer j) Brainstem Glioma i) Low Risk Prostate Cancer & Brachytherapy ii) Intermediate/High Risk Prostate Cancer 2) Central Nervous System iii) Adjuvant/Salvage & Metastatic Prostate Cancer a) Low Grade Glioma b) Bladder Cancer b) High Grade Glioma c) Renal Cell Cancer c) Primary CNS lymphoma d) Urethral Cancer d) Meningioma e) Testicular Cancer e) Pituitary Tumor f) Penile Cancer 3) Head and Neck 8) Gynecologic a) Ocular Melanoma a) Cervical Cancer b) Nasopharyngeal Cancer b) Endometrial Cancer c) Paranasal Sinus Cancer c) Uterine Sarcoma d) Oral Cavity Cancer d) Vulvar Cancer e) Oropharyngeal Cancer e) Vaginal Cancer f) Salivary Gland Cancer f) Ovarian Cancer & Fallopian -

WHO Guidelines for Treatment of Cervical Intraepithelial Neoplasia 2–3 and Adenocarcinoma in Situ
WHO guidelines WHO guidelines for treatment of cervical intraepithelial neoplasia 2–3 and adenocarcinoma in situ Cryotherapy Large loop excision of the transformation zone Cold knife conization WHO guidelines WHO guidelines for treatment of cervical intraepithelial neoplasia 2–3 and adenocarcinoma in situ: cryotherapy, large loop excision of the transformation zone, and cold knife conization Catalogage à la source : Bibliothèque de l’OMS WHO guidelines for treatment of cervical intraepithelial neoplasia 2–3 and adenocarcinoma in situ: cryotherapy, large loop excision of the transformation zone, and cold knife conization. 1.Cervical Intraepithelial Neoplasia – diagnosis. 2.Cervical Intraepithelial Neoplasia – therapy. 3.Cervical Intraepithelial Neoplasia – surgery. 4.Adenocarcinoma – diagnosis. 5.Adenocarcinoma – therapy. 6.Cryotherapy – utilization. 7.Conization – methods. 8.Uterine Cervical Neoplasms – prevention and control. 9.Guideline. I.World Health Organization. ISBN 978 92 4 150677 9 (Classification NLM : WP 480) © World Health Organization 2014 All rights reserved. Publications of the World Health Organization are available on the WHO website (www.who.int) or can be purchased from WHO Press, World Health Organization, 20 Avenue Appia, 1211 Geneva 27, Switzerland (tel.: +41 22 791 3264; fax: +41 22 791 4857; e-mail: [email protected]). Requests for permission to reproduce or translate WHO publications – whether for sale or for non-commercial distribution – should be addressed to WHO Press through the WHO website (www.who.int/about/licensing/copyright_form/en/index.html). The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. -

Squamous Metaplasia of the Tracheal Epithelium in Children
Thorax: first published as 10.1136/thx.31.2.167 on 1 April 1976. Downloaded from Thorax (1976), 31, 167. Squamous metaplasia of the tracheal epithelium in children AVINASH MITHAL' and JOHN L. EMERY2 The Chest Clinic, Lincoln' and The Children's Hospital, Sheffield' Mithal, A. and Emery, J. L. (1976). Thorax, 31, 167-171. Squamous metaplasia of the tracheal epithelium in children. Thirty-seven (16%) tracheas from 2170 children showed squamous metaplasia. (Cases with tracheo-oesophageal fistula and congenital heart disease were excluded.) The metaplasia extended into the bronchi in 15 cases. Features of pulmonary retention were present in seven cases. Respiratory infection, probably viral, seemed to be the most significant causative factor in 20 children, including those with cystic fibrosis. Tracheal instrumentation was a possible factor in 11 cases but oxygen therapy alone did not seem important. The metaplasia was almost certainly congenital in one child and probably in two others but no stillborn infants showed metaplasia. In many children the metaplasia seemed to be due to a combination of factors. Squamous metaplasia of the trachea in childhood Tracheas from children with tracheo-oesophageal has been described in cases of measles (Gold- fistula and those with congenital heart disease or zieher, 1918), influenza (Askanazy, 1919), cystic other gross deformities were excluded. There were fibrosis of the pancreas (Zuelzer and Newton, thus 2331 tracheas available for study. Epithelium 1949), and following intubation of the trachea was absent in 16 cases. This left 2170 tracheas for http://thorax.bmj.com/ (Rasche and Kuhns, 1972) and tracheostomy histological analysis. (Sara, 1967; Sara and Reye, 1969). -

Squamous Metaplasia of Normal and Carcinoma in Situ of HPV 16-Immortalized Human Endocervical Cells1
[CANCER RESEARCH 52. 4254-4260, August I, 1992] Squamous Metaplasia of Normal and Carcinoma in Situ of HPV 16-Immortalized Human Endocervical Cells1 Qi Sun, Kouichiro Tsutsumi, M. Brian Kelleher, Alan Pater, and Mary M. Pater2 Division of Basic Medical Sciences, Faculty of Medicine, Memorial University of Newfoundland, St. John's, Newfoundland, Canada A1B ÌV6 ABSTRACT genomic DNA, most frequently of HPV 16, has been detected in 90% of the cervical carcinomas and are found to be actively The importance of cervical squamous metaplasia and human papil- expressed (6, 7). HPV 16 DNA has been used to transform lomavirus 16 (HPV 16) infection for cervical carcinoma has been well human foreskin and ectocervical keratinocytes (8, 9). It immor established. Nearly 87% of the intraepithelial neoplasia of the cervix occur in the transformation zone, which is composed of squamous meta- talizes human keratinocytes efficiently, producing cell clones plastic cells with unclear origin. HPV DNA, mostly HPV 16, has been with indefinite life span in culture. Different approaches have found in 90% of cervical carcinomas, but only limited experimental data been taken to examine the behavior of these immortalized cell are available to discern the role of HPV 16 in this tissue specific onco- lines in conditions allowing squamous differentiation (10, 11). genesis. We have initiated in vivo studies of cultured endocervical cells After transplantation in vivo, the HPV 16-immortalized kerat as an experimental model system for development of cervical neoplasia. inocytes retain thépotential for squamous differentiation, Using a modified in vivo implantation system, cultured normal endocer forming abnormal epithelium without dysplastic changes at vical epithelial cells formed epithelium resembling squamous metapla early passages and with various dysplastic changes only after sia, whereas those immortalized by HPV 16 developed into lesions long periods of time in culture (10). -

Squamous Cell Carcinoma of the Breast As a Clinical Diagnostic Challenge
582 MOLECULAR AND CLINICAL ONCOLOGY 8: 582-586, 2018 Squamous cell carcinoma of the breast as a clinical diagnostic challenge KATARZYNA JAKUBOWSKA1, LUIZA KAŃCZUGA‑KODA1, WOJCIECH KISIELEWSKI2, MARIUSZ KODA3 and WALDEMAR FAMULSKI1,2 1Department of Pathomorphology, Comprehensive Cancer Center, 15‑027 Białystok; Departments of 2Medical Pathomorphology and 3General Pathomorphology, Medical University of Białystok, 15‑269 Białystok, Poland Received September 17, 2017; Accepted December 14, 2017 DOI: 10.3892/mco.2018.1581 Abstract. Squamous cell carcinoma (SqCC) of the breast metaplasia of ductal and lobular epithelial cells can be should be differentiated between the primary skin keratinizing linked with fat necrosis and infracted ademonas. Squamous squamous carcinoma and squamous metaplastic cancer. In the cell carcinoma should be differentiated between lesions of current study, the cases of two patients who were diagnosed keratinizing squamous carcinoma and squamous metaplasia with SqCC originated from skin and the breast were discussed. associated to mammary carcinoma (2). The characteristic A fine-needle aspiration biopsy confirmed the presence features of metaplastic cell carcinoma include: i) primary of atypical squamous cells. In both cases, the microscopic carcinoma without other neoplastic components such as ductal examination of the surgical specimen revealed a malignant or mesenchymal elements, ii) the tumor origin is independent neoplasm differentiated into SqCC characterized by keratin- of the overlying skin and nipple and iii) absence of primary izing cancer cells with abundant eosiphilic cytoplasm with epidermoid tumors present in other site (oral cavity, bronchus, large, hyperchromatic vesicular nuclei. Immunohistochemical esophagus, bladder, cervix ect.) (3). However, squamous studies showed negative for progesterone and estrogen recep- metaplastic carcinoma should be also differentiated with pure tors and human epidermal growth factor receptor 2.