List of Withdrawn Drugs
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

(19) United States (12) Patent Application Publication (10) Pub
US 20130289061A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2013/0289061 A1 Bhide et al. (43) Pub. Date: Oct. 31, 2013 (54) METHODS AND COMPOSITIONS TO Publication Classi?cation PREVENT ADDICTION (51) Int. Cl. (71) Applicant: The General Hospital Corporation, A61K 31/485 (2006-01) Boston’ MA (Us) A61K 31/4458 (2006.01) (52) U.S. Cl. (72) Inventors: Pradeep G. Bhide; Peabody, MA (US); CPC """"" " A61K31/485 (201301); ‘4161223011? Jmm‘“ Zhu’ Ansm’ MA. (Us); USPC ......... .. 514/282; 514/317; 514/654; 514/618; Thomas J. Spencer; Carhsle; MA (US); 514/279 Joseph Biederman; Brookline; MA (Us) (57) ABSTRACT Disclosed herein is a method of reducing or preventing the development of aversion to a CNS stimulant in a subject (21) App1_ NO_; 13/924,815 comprising; administering a therapeutic amount of the neu rological stimulant and administering an antagonist of the kappa opioid receptor; to thereby reduce or prevent the devel - . opment of aversion to the CNS stimulant in the subject. Also (22) Flled' Jun‘ 24’ 2013 disclosed is a method of reducing or preventing the develop ment of addiction to a CNS stimulant in a subj ect; comprising; _ _ administering the CNS stimulant and administering a mu Related U‘s‘ Apphcatlon Data opioid receptor antagonist to thereby reduce or prevent the (63) Continuation of application NO 13/389,959, ?led on development of addiction to the CNS stimulant in the subject. Apt 27’ 2012’ ?led as application NO_ PCT/US2010/ Also disclosed are pharmaceutical compositions comprising 045486 on Aug' 13 2010' a central nervous system stimulant and an opioid receptor ’ antagonist. -

Réglementation De La Pharmacie
R E C U E I L D E T E X T E S S U R L A P H A R M A C I E Mis à jour le 13 février 2017 par l’Inspection de la pharmacie P R É A M B U L E La réglementation relative à la pharmacie en vigueur en Nouvelle-Calédonie résulte de la coexistence des dispositions adoptées par la Nouvelle-Calédonie au titre de ses compétences en matières d’hygiène publique, de santé et de professions de la pharmacie1, et de celles adoptées par l’Etat au titre de ses compétences en matières de garanties des libertés publiques, de droit civil et de droit commercial2. Sur le contenu du recueil En 1954, la Nouvelle-Calédonie s’est vue étendre les articles L. 511 à L. 520 et L. 549 à L. 665 de l’ancien Livre V relatif à la Pharmacie du code de la santé publique métropolitain par la loi n° 54-418 du 15 avril 1954 étendant aux territoires d'outre-mer, au Togo et au Cameroun certaines dispositions du Code de la santé publique relatives à l'exercice de la pharmacie3, dont les modalités d’application ont été fixées par le décret modifié n° 55-1122 du 16 août 1955 fixant les modalités d'application de la loi n° 54-418 du 15 avril 1954 étendant aux territoires d'outre-mer, au Togo et au Cameroun certaines dispositions du code de la santé publique relatives à l'exercice de la pharmacie4. Depuis sont intervenues la loi- cadre Defferre5, la loi référendaire de 19886 et la loi organique n° 99-209 du 19 mars 1999 dont les apports ont eu pour résultat le transfert de ces articles de la compétence de l’Etat à la compétence de la Nouvelle-Calédonie, permettant à celle-ci de s’en approprier et de les modifier à sa guise par des délibérations du congrès de la Nouvelle-Calédonie7. -

1 441 702 B1
(19) TZZ__Z _T (11) EP 1 441 702 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61K 31/4045 (2006.01) A61P 25/20 (2006.01) 10.05.2017 Bulletin 2017/19 A61K 9/20 (2006.01) (21) Application number: 02760523.7 (86) International application number: PCT/IL2002/000662 (22) Date of filing: 12.08.2002 (87) International publication number: WO 2003/015690 (27.02.2003 Gazette 2003/09) (54) METHOD FOR TREATING PRIMARY INSOMNIA VERFAHREN ZUR BEHANDLUNG PRIMÄRER INSOMNIA METHODE DE TRAITEMENT DE L’INSOMNIE PRIMAIRE (84) Designated Contracting States: • ROTH T ET AL: "Consensus for the AT BE BG CH CY CZ DE DK EE ES FI FR GB GR pharmacological management of insomnia in th IE IT LI LU MC NL PT SE SK TR enew millennium", INTERNATIONAL JOURNAL Designated Extension States: OF CLINICAL PRACTICE, MEDICON LT LV RO SI INTERNATIONAL, ESHER, GB, vol. 55, no. 1, 1 January 2001 (2001-01-01), page 10PAGES, (30) Priority: 14.08.2001 IL 14490001 XP002990688, ISSN: 1368-5031 • PERLIS M L ET AL: "Psychophysiological (43) Date of publication of application: insomnia: the behavioural model and a 04.08.2004 Bulletin 2004/32 neurocognitive perspective", JOURNAL OF SLEEP RESEARCH, BLACKWELL SCIENTIFIC, (60) Divisional application: OXFORD, GB, vol. 6, 1 January 1997 (1997-01-01), 16172415.8 / 3 103 443 pages 179-188, XP002990686, ISSN: 0962-1105, DOI: DOI:10.1046/J.1365-2869.1997.00045.X (73) Proprietor: NEURIM PHARMACEUTICALS (1991) • SILVA J A C E ET AL: "Special report from a LIMITED symposium held by the WHO and the World Tel Aviv 69710 (IL) Federation of sleep research societies: an overview of insomnias and related disorders - (72) Inventor: ZISAPEL, Nava recognition, epidemiology and rational 69355 Tel Aviv (IL) management", SLEEP, ALLEN PRESS, LAWRENCE, KS,US, vol. -

(12) United States Patent (10) Patent No.: US 9,365,521 B2 Blackburn Et Al
USOO9365521B2 (12) United States Patent (10) Patent No.: US 9,365,521 B2 Blackburn et al. (45) Date of Patent: Jun. 14, 2016 (54) NON-HYGROSCOPICSALTS OF 5-HT, 5,178,786 A 1/1993 Jahnke et al. AGONSTS 5,247,080 A 9/1993 Berger et al. 5,275,915 A 1/1994 Kojima et al. 5,387,685 A 2f1995 Powell et al. (75) Inventors: Anthony C. Blackburn, San Diego, CA 5,397.793 A 3, 1995 Shaber et al. (US); Yun Shan, San Diego, CA (US); 5,412,119 A 5/1995 Brussee et al. Anna Shifrina, San Diego, CA (US); 5,422,355 A 6/1995 White et al. Scott Stirn, San Diego, CA (US) 5,691.362 A 11/1997 McCormicket al. 5,750,520 A 5/1998 Danilewicz et al. (73) Assignee: Arena Pharmaceuticals, Inc., San 5,856.5035,795,895 A 8,1/1999 1998 AnchAS al. Diego, CA (US) 5,861,393 A 1/1999 Danilewicz et al. 5,908,830 A 6/1999 Smith et al. (*) Notice: Subject to any disclaimer, the term of this 5,925,651 A 7/1999 Hutchinson patent is extended or adjusted under 35 3. A 3: As al U.S.C. 154(b) by 567 days. 5,958,943- 4 A 9/1999 Laufera etca. al. 6,087,346 A 7/2000 Glennon et al. (21) Appl. No.: 13/820,095 6,218,385 B1 4/2001 Adam et al. 6,900,313 B2 5/2005 Wasserscheid et al. (22) PCT Filed: Aug. 31, 2011 6,953,787 B2 * 10/2005 Smith ................. -

Wo 2008/127291 A2
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (43) International Publication Date PCT (10) International Publication Number 23 October 2008 (23.10.2008) WO 2008/127291 A2 (51) International Patent Classification: Jeffrey, J. [US/US]; 106 Glenview Drive, Los Alamos, GOlN 33/53 (2006.01) GOlN 33/68 (2006.01) NM 87544 (US). HARRIS, Michael, N. [US/US]; 295 GOlN 21/76 (2006.01) GOlN 23/223 (2006.01) Kilby Avenue, Los Alamos, NM 87544 (US). BURRELL, Anthony, K. [NZ/US]; 2431 Canyon Glen, Los Alamos, (21) International Application Number: NM 87544 (US). PCT/US2007/021888 (74) Agents: COTTRELL, Bruce, H. et al.; Los Alamos (22) International Filing Date: 10 October 2007 (10.10.2007) National Laboratory, LGTP, MS A187, Los Alamos, NM 87545 (US). (25) Filing Language: English (81) Designated States (unless otherwise indicated, for every (26) Publication Language: English kind of national protection available): AE, AG, AL, AM, AT,AU, AZ, BA, BB, BG, BH, BR, BW, BY,BZ, CA, CH, (30) Priority Data: CN, CO, CR, CU, CZ, DE, DK, DM, DO, DZ, EC, EE, EG, 60/850,594 10 October 2006 (10.10.2006) US ES, FI, GB, GD, GE, GH, GM, GT, HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, KR, KZ, LA, LC, LK, (71) Applicants (for all designated States except US): LOS LR, LS, LT, LU, LY,MA, MD, ME, MG, MK, MN, MW, ALAMOS NATIONAL SECURITY,LLC [US/US]; Los MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PG, PH, PL, Alamos National Laboratory, Lc/ip, Ms A187, Los Alamos, PT, RO, RS, RU, SC, SD, SE, SG, SK, SL, SM, SV, SY, NM 87545 (US). -

Ep 2089382 B1
(19) TZZ Z¥ _T (11) EP 2 089 382 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: C07D 401/14 (2006.01) C07D 413/14 (2006.01) 24.10.2012 Bulletin 2012/43 C07D 471/04 (2006.01) C07D 487/04 (2006.01) C07D 491/048 (2006.01) C07D 491/052 (2006.01) (2006.01) (2006.01) (21) Application number: 07862400.4 C07D 495/04 A61K 31/551 A61P 3/04 (2006.01) A61P 25/20 (2006.01) C07D 403/14 (2006.01) (22) Date of filing: 30.11.2007 (86) International application number: PCT/US2007/024690 (87) International publication number: WO 2008/069997 (12.06.2008 Gazette 2008/24) (54) SUBSTITUTED DIAZEPAN COMPOUNDS AS OREXIN RECEPTOR ANTAGONISTS SUBSTITUIERTE DIAZEPANVERBINDUNGEN ALS OREXIN-REZEPTOR-ANTAGONISTEN ANTAGONISTES DES RÉCEPTEURS DE L’OREXINE SOUS FORME DE COMPOSÉS DE DIAZÉPANE SUBSTITUÉS (84) Designated Contracting States: • COX, Christopher D. AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Rahway, NJ 07065-0907 (US) HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE • MERCER, Swati P. SI SK TR Rahway, NJ 07065-0907 (US) Designated Extension States: • ROECKER, Anthony J. HR RS Rahway, NJ 07065-0907 (US) (30) Priority: 01.12.2006 US 872393 P (74) Representative: Buchan, Gavin MacNicol 16.07.2007 US 959742 P Merck Sharp & Dohme Limited European Patent Department (43) Date of publication of application: Hertford Road 19.08.2009 Bulletin 2009/34 Hoddesdon Hertfordshire EN11 9BU (GB) (60) Divisional application: 11177692.8 / 2 392 572 (56) References cited: EP-A- 0 794 178 WO-A-03/041711 (73) Proprietor: Merck Sharp & Dohme Corp. -

Nsaids in Cats
ISFM and AAFP Consensus Guidelines Long-term use of NSAIDs in cats Clinical Practice A preprint from the Journal of Feline Medicine and Surgery Volume 12, July 2010 Journal of Feline Medicine and Surgery (2010) 12, 519 doi:10.1016/j.jfms.2010.05.003 EDITORIAL NSAIDs and cats – it’s been a long journey Although the first This issue of JFMS contains the first ever panel have covered much valuable ground: international consensus guidelines ✜ To set the scene they consider how use of NSAIDs on the long-term use of non-steroidal common chronic pain can be in cats, typically was probably anti-inflammatory drugs (NSAIDs) in cats. related to degenerative joint disease, This timely publication, which appears on idiopathic cystitis, trauma and cancer. by Hippocrates, pages 521–538 (doi:10.1016/j.jfms.2010.05.004), ✜ They then explain how and why NSAIDs is a collaborative enterprise by the International can have such positive and, potentially, it has taken until Society of Feline Medicine (ISFM) and negative actions. now for cats to American Association of Feline Practitioners ✜ They consider the best ways of enhancing (AAFP). It has been compiled by a panel of owner and cat compliance, make suggestions gain the benefit of world leaders in the understanding of pain about sensible dosing frequencies, timing of in cats and, without doubt, is essential reading medication and accuracy of dosing, and the long-term use for all small animal veterinary surgeons. emphasize the importance of always using of these drugs. It is interesting to reflect that although the ‘lowest effective dose’. -

Stembook 2018.Pdf
The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 WHO/EMP/RHT/TSN/2018.1 © World Health Organization 2018 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances. Geneva: World Health Organization; 2018 (WHO/EMP/RHT/TSN/2018.1). Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. -

Predicting the Function of Drug-Like Molecules Methods and Applications
Predicting the function of drug-like molecules methods and applications Inaugural-Dissertation to obtain the academic degree Doctor rerum naturalium (Dr. rer. nat.) submitted to the Department of Biology, Chemistry and Pharmacy of Freie Universität Berlin by Hao Wang from Liupanshui, Guizhou province, People’s Republic of China January 2017 Diese Arbeit wurde in dem Zeitraum von November 2012 bis Januar 2017 unter der Leitung von Prof. Dr. Ernst Walter Knapp am Institute für Chemie und Biochemie der Freien Universität Berlin im Fachbereich Biologie, Chemie und Pharmazie durchgeführt. 1. Gutacher: Prof. Dr. Ernst-Walter Knapp 2. Gutacher: Prof. Dr. Gerhard Wolber Disputation am ___16. 3. 2017_________ Statutory Declaration I hereby testify that this thesis is the result of my own work and research, except for any explicitly referenced material, whose source is listed in the bibliography. This work contains material that is the copyright property of others, which must not be reproduced without the permission of the copyright own. Acknowledgement First of all, I would like to thank Prof. Dr. Ernst Walter Knapp, China Scholarship Council, Free University of Berlin and Xiamen University for giving me this precious opportunity to study in Germany. I very much appreciate the supervision of Prof. Dr. Ernst Walter Knapp and valuable discussions during my doctoral work. In addition, I would also like to thank Dr. Bentzien Jörg and Dr. Ingo Mugge in Boehringer-Ingelheim group for their cooperation and valuable suggestions for my research projects. I am thankful for the support and great help of my colleagues in AG Knapp during my doctoral studies. -

(CD-P-PH/PHO) Report Classification/Justifica
COMMITTEE OF EXPERTS ON THE CLASSIFICATION OF MEDICINES AS REGARDS THEIR SUPPLY (CD-P-PH/PHO) Report classification/justification of - Medicines belonging to the ATC group M01 (Antiinflammatory and antirheumatic products) Table of Contents Page INTRODUCTION 6 DISCLAIMER 8 GLOSSARY OF TERMS USED IN THIS DOCUMENT 9 ACTIVE SUBSTANCES Phenylbutazone (ATC: M01AA01) 11 Mofebutazone (ATC: M01AA02) 17 Oxyphenbutazone (ATC: M01AA03) 18 Clofezone (ATC: M01AA05) 19 Kebuzone (ATC: M01AA06) 20 Indometacin (ATC: M01AB01) 21 Sulindac (ATC: M01AB02) 25 Tolmetin (ATC: M01AB03) 30 Zomepirac (ATC: M01AB04) 33 Diclofenac (ATC: M01AB05) 34 Alclofenac (ATC: M01AB06) 39 Bumadizone (ATC: M01AB07) 40 Etodolac (ATC: M01AB08) 41 Lonazolac (ATC: M01AB09) 45 Fentiazac (ATC: M01AB10) 46 Acemetacin (ATC: M01AB11) 48 Difenpiramide (ATC: M01AB12) 53 Oxametacin (ATC: M01AB13) 54 Proglumetacin (ATC: M01AB14) 55 Ketorolac (ATC: M01AB15) 57 Aceclofenac (ATC: M01AB16) 63 Bufexamac (ATC: M01AB17) 67 2 Indometacin, Combinations (ATC: M01AB51) 68 Diclofenac, Combinations (ATC: M01AB55) 69 Piroxicam (ATC: M01AC01) 73 Tenoxicam (ATC: M01AC02) 77 Droxicam (ATC: M01AC04) 82 Lornoxicam (ATC: M01AC05) 83 Meloxicam (ATC: M01AC06) 87 Meloxicam, Combinations (ATC: M01AC56) 91 Ibuprofen (ATC: M01AE01) 92 Naproxen (ATC: M01AE02) 98 Ketoprofen (ATC: M01AE03) 104 Fenoprofen (ATC: M01AE04) 109 Fenbufen (ATC: M01AE05) 112 Benoxaprofen (ATC: M01AE06) 113 Suprofen (ATC: M01AE07) 114 Pirprofen (ATC: M01AE08) 115 Flurbiprofen (ATC: M01AE09) 116 Indoprofen (ATC: M01AE10) 120 Tiaprofenic Acid (ATC: -

Pharmaceuticals (Monocomponent Products) ………………………..………… 31 Pharmaceuticals (Combination and Group Products) ………………….……
DESA The Department of Economic and Social Affairs of the United Nations Secretariat is a vital interface between global and policies in the economic, social and environmental spheres and national action. The Department works in three main interlinked areas: (i) it compiles, generates and analyses a wide range of economic, social and environmental data and information on which States Members of the United Nations draw to review common problems and to take stock of policy options; (ii) it facilitates the negotiations of Member States in many intergovernmental bodies on joint courses of action to address ongoing or emerging global challenges; and (iii) it advises interested Governments on the ways and means of translating policy frameworks developed in United Nations conferences and summits into programmes at the country level and, through technical assistance, helps build national capacities. Note Symbols of United Nations documents are composed of the capital letters combined with figures. Mention of such a symbol indicates a reference to a United Nations document. Applications for the right to reproduce this work or parts thereof are welcomed and should be sent to the Secretary, United Nations Publications Board, United Nations Headquarters, New York, NY 10017, United States of America. Governments and governmental institutions may reproduce this work or parts thereof without permission, but are requested to inform the United Nations of such reproduction. UNITED NATIONS PUBLICATION Copyright @ United Nations, 2005 All rights reserved TABLE OF CONTENTS Introduction …………………………………………………………..……..……..….. 4 Alphabetical Listing of products ……..………………………………..….….…..….... 8 Classified Listing of products ………………………………………………………… 20 List of codes for countries, territories and areas ………………………...…….……… 30 PART I. REGULATORY INFORMATION Pharmaceuticals (monocomponent products) ………………………..………… 31 Pharmaceuticals (combination and group products) ………………….……........ -
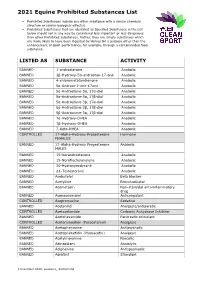
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s).