Safety Data Sheet
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

(Vocs and Svocs) from Warm Mix Asphalt Incorporating Natural Zeolite and Reclaimed Asphalt Pavement for Sustainable Pavements
sustainability Article Evaluation of Reductions in Fume Emissions (VOCs and SVOCs) from Warm Mix Asphalt Incorporating Natural Zeolite and Reclaimed Asphalt Pavement for Sustainable Pavements Javier Espinoza 1,2 , Cristian Medina 1,2, Alejandra Calabi-Floody 3 , Elsa Sánchez-Alonso 3 , Gonzalo Valdés 3 and Andrés Quiroz 1,2,* 1 Chemical Ecology Laboratory, Department of Chemical Sciences and Natural Resources, University of La Frontera, Temuco 4811230, Chile; [email protected] (J.E.); [email protected] (C.M.) 2 Biotechnological Research Center Applied to the Environment (CIBAMA), University of La Frontera, Temuco 4811230, Chile 3 Department of Civil Works Engineering, University of La Frontera, Temuco 4811230, Chile; [email protected] (A.C.-F.); [email protected] (E.S.-A.); [email protected] (G.V.) * Correspondence: [email protected] Received: 26 September 2020; Accepted: 10 November 2020; Published: 17 November 2020 Abstract: Conventional asphalt mixtures used for road paving require high manufacturing temperatures and therefore high energy expenditure, which has a negative environmental impact and creates risk in the workplace owing to high emissions of pollutants, greenhouse gases, and toxic fumes. Reducing energy consumption and emissions is a continuous challenge for the asphalt industry. Previous studies have focused on the reduction of emissions without characterizing their composition, and detailed characterization of volatile organic compounds (VOCs) and semi-volatile organic compounds (SVOCs) in asphalt fumes is scarce. This communication describes the characterization and evaluation of VOCs and SVOCs from asphalt mixtures prepared at lower production temperatures using natural zeolite; in some cases, reclaimed asphalt pavement (RAP) was used. -

Part I: Carbonyl-Olefin Metathesis of Norbornene
Part I: Carbonyl-Olefin Metathesis of Norbornene Part II: Cyclopropenimine-Catalyzed Asymmetric Michael Reactions Zara Maxine Seibel Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate School of Arts and Sciences COLUMBIA UNIVERSITY 2016 1 © 2016 Zara Maxine Seibel All Rights Reserved 2 ABSTRACT Part I: Carbonyl-Olefin Metathesis of Norbornene Part II: Cyclopropenimine-Catalyzed Asymmetric Michael Reactions Zara Maxine Seibel This thesis details progress towards the development of an organocatalytic carbonyl- olefin metathesis of norbornene. This transformation has not previously been done catalytically and has not been done in practical manner with stepwise or stoichiometric processes. Building on the previous work of the Lambert lab on the metathesis of cyclopropene and an aldehyde using a hydrazine catalyst, this work discusses efforts to expand to the less stained norbornene. Computational and experimental studies on the catalytic cycle are discussed, including detailed experimental work on how various factors affect the difficult cycloreversion step. The second portion of this thesis details the use of chiral cyclopropenimine bases as catalysts for asymmetric Michael reactions. The Lambert lab has previously developed chiral cyclopropenimine bases for glycine imine nucleophiles. The scope of these catalysts was expanded to include glycine imine derivatives in which the nitrogen atom was replaced with a carbon atom, and to include imines derived from other amino acids. i Table of Contents List of Abbreviations…………………………………………………………………………..iv Part I: Carbonyl-Olefin Metathesis…………………………………………………………… 1 Chapter 1 – Metathesis Reactions of Double Bonds………………………………………….. 1 Introduction………………………………………………………………………………. 1 Olefin Metathesis………………………………………………………………………… 2 Wittig Reaction…………………………………………………………………………... 6 Tebbe Olefination………………………………………………………………………... 9 Carbonyl-Olefin Metathesis……………………………………………………………. -

United States Patent (19) 11 Patent Number: 5,731,483 Stabel Et Al
US005731483A United States Patent (19) 11 Patent Number: 5,731,483 Stabel et al. 45 Date of Patent: Mar. 24, 1998 54 RECYCLING OF PLASTICS IN A STEAM 52 U.S. Cl. .......................... 585/241; 585/648; 208/130 CRACKER 58 Field of Search ..................................... 585/241, 648; 208/130 75) Inventors: Uwe Stabel, Edingen-Neckarhausen; Helmut Woerz, Mannheim; Ruediger 56) References Cited Kotkamp, Limburgerhof; Andreas U.S. PATENT DOCUMENTS Fried, Bobenheim-Roxheim, all of Germany 5,364.995 11/1994 Kirkwood et al. ...................... 585,241 73) Assignee: BASFAktiengesellschaft, FOREIGN PATENT DOCUMENTS Ludwigshafen, Germany 2108.968 9/1983 Canada. 2094.456 10/1993 Canada. 21) Appl. No.: 553,658 502 618 9/1992 European Pat. Off. 567 292 10/1993 European Pat. Off. 22 PCT Filed: May 20, 1994 WO93/1812 9/1993 WIPO. (86 PCT No.: PCT/EP94/01647 Primary Examiner-Glenn Caldarola S371 Date: Nov. 17, 1995 Assistant Examiner-Bekir L. Yildirim Attorney, Agent, or Firm-Keil & Weinkauf S 102(e) Date: Nov. 17, 1995 57 ABSTRACT 87 PCT Pub. No.: WO95/03375 A process for recycling plastic waste in a steam cracker, PCT Pub. Date: Feb. 2, 1995 wherein a melt obtained from plastic waste is converted into 30 Foreign Application Priority Data products at from 400° 550° C., and a distillate fraction is separated off from the products at from 180° to 280° C. and Jul. 20, 1993 DEl Germany .......................... 43 24, 112.3 is fed as feed material to a steam cracker, Jan. 10, 1994 DEl Germany .......................... 4400 366.8 (51 int. Cl. .................. C07C 1/00; CO7C 4/22 9 Claims, 2 Drawing Sheets - N 5 AAA' U.S. -

Catalytic Pyrolysis of Plastic Wastes for the Production of Liquid Fuels for Engines
Electronic Supplementary Material (ESI) for RSC Advances. This journal is © The Royal Society of Chemistry 2019 Supporting information for: Catalytic pyrolysis of plastic wastes for the production of liquid fuels for engines Supattra Budsaereechaia, Andrew J. Huntb and Yuvarat Ngernyen*a aDepartment of Chemical Engineering, Faculty of Engineering, Khon Kaen University, Khon Kaen, 40002, Thailand. E-mail:[email protected] bMaterials Chemistry Research Center, Department of Chemistry and Center of Excellence for Innovation in Chemistry, Faculty of Science, Khon Kaen University, Khon Kaen, 40002, Thailand Fig. S1 The process for pelletization of catalyst PS PS+bentonite PP ) t e PP+bentonite s f f o % ( LDPE e c n a t t LDPE+bentonite s i m s n HDPE a r T HDPE+bentonite Gasohol 91 Diesel 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) Fig. S2 FTIR spectra of oil from pyrolysis of plastic waste type. Table S1 Compounds in oils (%Area) from the pyrolysis of plastic wastes as detected by GCMS analysis PS PP LDPE HDPE Gasohol 91 Diesel Compound NC C Compound NC C Compound NC C Compound NC C 1- 0 0.15 Pentane 1.13 1.29 n-Hexane 0.71 0.73 n-Hexane 0.65 0.64 Butane, 2- Octane : 0.32 Tetradecene methyl- : 2.60 Toluene 7.93 7.56 Cyclohexane 2.28 2.51 1-Hexene 1.05 1.10 1-Hexene 1.15 1.16 Pentane : 1.95 Nonane : 0.83 Ethylbenzen 15.07 11.29 Heptane, 4- 1.81 1.68 Heptane 1.26 1.35 Heptane 1.22 1.23 Butane, 2,2- Decane : 1.34 e methyl- dimethyl- : 0.47 1-Tridecene 0 0.14 2,2-Dimethyl- 0.63 0 1-Heptene 1.37 1.46 1-Heptene 1.32 1.35 Pentane, -

Catalytic Enantioselective Carbon-Carbon Bond Formation Using Cycloisomerization Reactions
View Online / Journal Homepage / Table of Contents for this issue Chemical Science Dynamic Article LinksC< Cite this: Chem. Sci., 2012, 3, 2899 www.rsc.org/chemicalscience MINIREVIEW Catalytic enantioselective carbon-carbon bond formation using cycloisomerization reactions Iain D. G. Watsona and F. Dean Toste*b Received 30th April 2012, Accepted 7th June 2012 DOI: 10.1039/c2sc20542d This review describes important recent advancements in asymmetric cycloisomerization reactions. A wide variety of catalytic and asymmetric strategies have been applied to these reactions over the past twenty years. Cycloisomerization reactions have the ability to produce diverse polycyclic compounds in excellent yields and selectivity. They constitute a powerful and efficient strategy for asymmetric carbon- carbon bond formation in cyclic compounds. Enyne and related olefin cyclizations comprise the majority of reactions of this type and important advances have recently occurred in this area. However, significant changes have also occurred in the area of classical cyclization as well as intramolecular hydroacylation and C–H activation initiated cyclization and these will also be described. 1 Introduction The purpose of this review is to describe important new advances in asymmetric cycloisomerization reactions. In partic- The synthesis of rings is central to the art of organic synthesis. ular, this review will focus on enantioselective carbon-carbon Cyclic compounds abound in chemistry, from strained three bond forming cycloisomerizations. Many aspects of cyclo- membered rings to macrocyclic monsters. A common synthetic isomerization reactions have already been reviewed,3 including challenge is the creation of a ring within a certain target mechanistic4 and asymmetric aspects of the reaction.5 This compound. -

7Alkenes and Alkynes I: Properties and Synthesis
P1: PBU/OVY P2: PBU/OVY QC: PBU/OVY T1: PBU Printer: Bind Rite JWCL234-07 JWCL234-Solomons-v1 December 8, 2009 21:37 7 ALKENES AND ALKYNES I: PROPERTIES AND SYNTHESIS. ELIMINATION REACTIONS OF ALKYL HALIDES SOLUTIONS TO PROBLEMS 7.1 (a) (E )-1-Bromo-1-chloro-1-pentene or (E )-1-Bromo-1-chloropent-1-ene (b) (E )-2-Bromo-1-chloro-1-iodo-1-butene or (E )-2-Bromo-1-chloro-1-iodobut-1-ene (c) (Z )-3,5-Dimethyl-2-hexene or (Z )-3,5-Dimethylhex-2-ene (d) (Z )-1-Chloro-1-iodo-2-methyl-1-butene or (Z )-1-Chloro-1-iodo-2-methylbut-1-ene (e) (Z,4S )-3,4-Dimethyl-2-hexene or (Z,4S )-3,4-Dimethylhex-2-ene (f) (Z,3S )-1-Bromo-2-chloro-3-methyl-1-hexene or (Z,3S )-1-Bromo-2-chloro-3-methylhex-1-ene 7.2 <<Order of increasing stability 7.3 (a), (b) H 2 Δ H° = − 119 kJ mol−1 Pt 2-Methyl-1-butene pressure (disubstituted) H 2 Δ H° = − 127 kJ mol−1 Pt 3-Methyl-1-butene pressure (monosubstituted) H2 − Δ H° = − 113 kJ mol 1 Pt 2-Methyl-2-butene pressure (trisubstituted) (c) Yes, because hydrogenation converts each alkene into the same product. 106 CONFIRMING PAGES P1: PBU/OVY P2: PBU/OVY QC: PBU/OVY T1: PBU Printer: Bind Rite JWCL234-07 JWCL234-Solomons-v1 December 8, 2009 21:37 ALKENES AND ALKYNES I: PROPERTIES AND SYNTHESIS 107 H H (d) >> H H H (trisubstituted) (disubstituted) (monosubstituted) Notice that this predicted order of stability is confirmed by the heats of hydro- genation. -

Conformational Studies of Aliphatic Secondary Ozonides (Propene, 1-Butene and 1-Heptene) by Means of FTIR Spectroscopy
DOI: 10.2478/s11532-006-0031-3 Research article CEJC 4(4) 2006 578–591 Conformational studies of aliphatic secondary ozonides (propene, 1-butene and 1-heptene) by means of FTIR spectroscopy Ruta Bariseviciute, Justinas Ceponkus, Alytis Gruodis, Valdas Sablinskas∗ Department of General Physics and Spectroscopy, Vilnius University, Vilnius-01513, Lithuania Received 6 January 2006; accepted 31 May 2006 Abstract: Ozonization reaction of simple alkenes was studied by means of FT infrared absorption gas spectroscopy. The reaction was performed at 95 K in neat films of the reactants. IR absorption spectra of the gaseous products were recorded. The spectra were analyzed combining experimental results with theoretical calculations performed at B3LYP 6-311++G (3df, 3pd) level. We found that among all theoretically predicted conformers of propene secondary ozonide, only one which has the O-O half-chair configuration for the five membered ring and the radical attached in the equatorial position was present in the sample. Samples of 1-butene and 1-heptene secondary ozonides consist from two conformers of very similar energy (ΔH =0.3 kJ/mol). The most stable conformer for both ozonides is the one with O-O half-chair configuration of the five membered ring and the radical attached in equatorial position and the aliphatic chain in gauche position. The second stable conformer has the aliphatic chain in anti position. c Versita Warsaw and Springer-Verlag Berlin Heidelberg. All rights reserved. Keywords: Ozonization, conformational analysis, secondary ozonides, IR spectroscopy 1 Introduction The ability of ozone to cleave double bonds in olefins has been known for more than 50 years. -

Properties and Synthesis. Elimination Reactions of Alkyl Halides
P1: PBU/OVY P2: PBU/OVY QC: PBU/OVY T1: PBU Printer: Bind Rite JWCL234-07 JWCL234-Solomons-v1 December 8, 2009 21:37 7 ALKENES AND ALKYNES I: PROPERTIES AND SYNTHESIS. ELIMINATION REACTIONS OF ALKYL HALIDES SOLUTIONS TO PROBLEMS 7.1 (a) ( E )-1-Bromo-1-chloro-1-pentene or ( E )-1-Bromo-1-chloropent-1-ene (b) ( E )-2-Bromo-1-chloro-1-iodo-1-butene or ( E )-2-Bromo-1-chloro-1-iodobut-1-ene (c) ( Z )-3,5-Dimethyl-2-hexene or ( Z )-3,5-Dimethylhex-2-ene (d) ( Z )-1-Chloro-1-iodo-2-methyl-1-butene or ( Z )-1-Chloro-1-iodo-2-methylbut-1-ene (e) ( Z,4 S )-3,4-Dimethyl-2-hexene or ( Z,4 S )-3,4-Dimethylhex-2-ene (f) ( Z,3 S )-1-Bromo-2-chloro-3-methyl-1-hexene or (Z,3 S )-1-Bromo-2-chloro-3-methylhex-1-ene 7.2 < < Order of increasing stability 7.3 (a), (b) H − 2 ∆ H° = − 119 kJ mol 1 Pt 2-Methyl-1-butene pressure (disubstituted) H2 − ∆ H° = − 127 kJ mol 1 Pt 3-Methyl-1-butene pressure (monosubstituted) H2 − ∆ H° = − 113 kJ mol 1 Pt 2-Methyl-2-butene pressure (trisubstituted) (c) Yes, because hydrogenation converts each alkene into the same product. 106 CONFIRMING PAGES P1: PBU/OVY P2: PBU/OVY QC: PBU/OVY T1: PBU Printer: Bind Rite JWCL234-07 JWCL234-Solomons-v1 December 8, 2009 21:37 ALKENES AND ALKYNES I: PROPERTIES AND SYNTHESIS 107 H H (d) > > H H H (trisubstituted) (disubstituted) (monosubstituted) Notice that this predicted order of stability is confirmed by the heats of hydro- genation. -
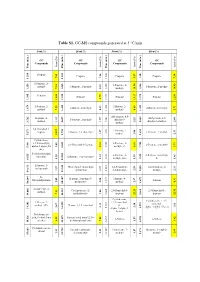
Table S1. GC-MS Compounds Generated at 5 °C/Min
Table S1. GC-MS compounds generated at 5 °C/min. 0 wt.% 10 wt.% 30 wt.% 50 wt.% GC GC GC GC Compounds Compounds Compounds Compounds Area (%) Area (%) Area (%) Area (%) Time (min.) (min.) Time Time (min.) (min.) Time (min.) Time (min.) Time Propene Propene Propene Propene 7.42 7.42 1.26 5.47 1.20 7.38 1.200 1.200 1.200 1.214 1.214 1-Propene, 2- 1-Propene, 2- methyl- 1-Propene, 2-methyl- 1-Propene, 2-methyl- 7.34 7.34 2.41 methyl- 1.245 1.245 1.245 1.259 1.259 10.54 1.246 14.90 Pentane Pentane Pentane Pentane 3.79 3.79 1.375 1.375 1.395 1.395 11.81 1.381 13.17 1.375 12.83 1-Pentene, 2- 2-Butene, 2- 2-Butene, 2-methyl- 2-Butene, 2-methyl- methyl- 7.25 0.93 methyl- 4.65 5.71 1.426 1.426 1.420 1.420 1.679 1.679 1H-Pyrazole, 4,5- Heptane, 4- 1H-Pyrazole, 4,5- 1-Pentene, 2-methyl- dihydro-5- methyl- 1.92 1.57 2.25 dihydro-5-methyl- 2.44 1.659 1.659 1.562 1.569 3.762 3.762 methyl- 2,4-Dimethyl-1- 1-Pentene, 2- heptene 2-Butene, 2,3-dimethyl- 1-Pentene, 2-methyl- 1.27 1.27 methyl- 3.08 2.33 1.653 1.653 1.653 5.671 5.671 34.57 1.750 Cyclohexane, 1,3,5-trimethyl-, 2-Pentene, 3- 2,4-Dimethyl-1-heptene 2-Pentene, 2-methyl- alpha.,3.alpha.,5.b 2.03 9.11 methyl-, €- 6.11 5.73 5.605 5.605 1.743 1.744 5.981 5.981 eta.)- 5-Aminoisoxazole 2-Pentene, 3- 2-Pentene, 3-methyl-, Spermine Ethanone, 1-cyclopentyl- 1.55 1.55 1.32 methyl-, (Z)- 1.59 €- 1.46 1.828 1.828 1.828 6.925 6.925 10.560 Ethanone, 1- 1R,2c,3t,4t-Tetramethyl- 1,3-Pentadiene, 1,4-Hexadiene, 5- cyclopentyl- 3.57 3.57 cyclohexan 0.97 2,3-dimethyl- 2.28 methyl- 2.31 2.584 2.584 2.585 10.613 10.613 10.638 N- Heptane, 2-methyl-3- Heptane, 4- Methylallylamine Toluene 2.49 2.49 methylene- 2.50 methyl- 2.97 4.31 3.697 3.697 3.697 14.047 14.047 10.684 10.684 2-Undecene, 4- Cyclopentane, (2- 2,4-Dimethyl-1- 2,4-Dimethyl-1- methyl- 7.81 7.81 methylbutyl)- 1.00 heptene heptene 5.605 5.605 22.29 22.29 5.605 12.95 14.086 14.086 14.163 Cyclohexane, Cyclohexane, 1,3,5- 2-Decene, 7- 1,3,5-trimethyl-, trimethyl- methyl-, (Z)- Hexane, 2,3,4-trimethyl- (1. -

United States Patent Office
Patented July 16, 1957 United States Patent Office 1. 2 be isolated from a solution in petroleum ether by frac tional crystallization. 2,799,685 The homologues, i. e. for example the pentachloro and tetrachloro-derivatives and the corresponding ana UNSATURATEDPROCESS FOR POLYCYCLIC THEIR MANUFACTURE SULFITES AND A logues of methyl and ethyl can be obtained in a corre einz Frensch, Helmut Goebel, Wilhelm Staudermann, sponding manner. : and Walter Finkenbrink, Frankfurt am Main, Ger. As Diels-Alder-components suitable for the manufac many, assignors to Farbwerke Hoechst Aktiengesell ture of Sulfites of the kind described above, there come schaft yormals Meister Lucius & Brining, Frankfurt also into consideration the halogenated alkyl derivatives, am Main, Germany; a corporation of the Federal Re O for example, dimethylcyclopentadiene. public of Germany The polycyclic halogenated sulfites are readily crystal lizing colorless compounds. No Drawing. Application April 8, 1955, The products of this invention may be used for pro Serial No. 500,264 tecting wood, paper, textiles and leather and as inter Claims priority, application Germany April 17, 1954 5 mediates for the manufacture of drugs. In the pure state, they are practically, odourless and are, therefore, 9 Claims. (CI. 260-327) especially suitable for the mentioned purposes. They The present invention provides new unsaturated poly have a curative and prophylactic effect and are prefer cyclic sulfites corresponding to the following general ably used, if desired in admixture with other insecticides, 20 ovicides, fungicides, herbicides or fertilizers, for combat formula: ing animal and fungoid pests. Y - In addition to their excellent action as pesticides the substances hereinbefore described are distinguished by 4R/V their chemical stability. -

Gfsorganics & Fragrances
Chemicals for Flavors GFSOrganics & Fragrances GFS offers a broad range of specialty organic chemicals Specialized chemistries we as building blocks and intermediates for the manufacture of offer include: flavors and fragrances. • Alkynes Over 5,500 materials, including 1,400 chemicals from natural sources, are used for flavor • Alkynols enhancements and aroma profiles. These aroma chemicals are integral components of • Olefins the continued growth within consumer products and packaged foods. The diversity of products can be attributed to the broad spectrum of organic compounds derived from • Unsaturated Acids esters, aldehydes, lactones, alcohols and several other functional groups. • Unsaturated Esters • DIPPN and other Products GFS Chemicals manufactures a wide range of organic intermediates that have been utilized in a multitude of personal care applications. For example, we support Why GFS? several market leading companies in the manufacture and supply of alkyne based aroma chemicals. • Specialized Chemistries • Tailored Specifications As such, we understand the demanding nature of this fast changing market and are fully • From Grams to Metric Tons equipped to overcome process challenges and manufacture the novel chemical products • Responsive Technical Staff that you need, when you need them. We offer flexible custom manufacturing services to produce high purity products with the assurance of quality and full confidentiality. • Uncompromised Product Quality Our state-of-the-art manufacturing facility, located in Columbus, OH is ISO 9001:2008 certified. As a U.S. based manufacturer we have a proven record of helping you take products from development to commercialization. Our technical sales experts are readily accessible to discuss your project needs and unique product specifications. -

Elimination Reactions Are Described
Introduction In this module, different types of elimination reactions are described. From a practical standpoint, elimination reactions widely used for the generation of double and triple bonds in compounds from a saturated precursor molecule. The presence of a good leaving group is a prerequisite in most elimination reactions. Traditional classification of elimination reactions, in terms of the molecularity of the reaction is employed. How the changes in the nature of the substrate as well as reaction conditions affect the mechanism of elimination are subsequently discussed. The stereochemical requirements for elimination in a given substrate and its consequence in the product stereochemistry is emphasized. ELIMINATION REACTIONS Objective and Outline beta-eliminations E1, E2 and E1cB mechanisms Stereochemical considerations of these reactions Examples of E1, E2 and E1cB reactions Alpha eliminations and generation of carbene I. Basics Elimination reactions involve the loss of fragments or groups from a molecule to generate multiple bonds. A generalized equation is shown below for 1,2-elimination wherein the X and Y from two adjacent carbon atoms are removed, elimination C C C C -XY X Y Three major types of elimination reactions are: α-elimination: two atoms or groups are removed from the same atom. It is also known as 1,1-elimination. H R R C X C + HX R Both H and X are removed from carbon atom here R Carbene β-elimination: loss of atoms or groups on adjacent atoms. It is also H H known as 1,2- elimination. R C C R R HC CH R X H γ-elimination: loss of atoms or groups from the 1st and 3rd positions as shown below.