PTPN6 (SHP1), Active Human Recombinant Protein Expressed in E.Coli Cells
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Inflammatory Cytokine Signalling by Protein Tyrosine Phosphatases in Pancreatic Β-Cells
59 4 W J STANLEY and others PTPN1 and PTPN6 modulate 59: 4 325–337 Research cytokine signalling in β-cells Differential regulation of pro- inflammatory cytokine signalling by protein tyrosine phosphatases in pancreatic β-cells William J Stanley1,2, Prerak M Trivedi1,2, Andrew P Sutherland1, Helen E Thomas1,2 and Esteban N Gurzov1,2,3 Correspondence should be addressed 1 St. Vincent’s Institute of Medical Research, Melbourne, Australia to E N Gurzov 2 Department of Medicine, St. Vincent’s Hospital, The University of Melbourne, Melbourne, Australia Email 3 ULB Center for Diabetes Research, Universite Libre de Bruxelles (ULB), Brussels, Belgium esteban.gurzov@unimelb. edu.au Abstract Type 1 diabetes (T1D) is characterized by the destruction of insulin-producing β-cells Key Words by immune cells in the pancreas. Pro-inflammatory including TNF-α, IFN-γ and IL-1β f pancreatic β-cells are released in the islet during the autoimmune assault and signal in β-cells through f protein tyrosine phosphorylation cascades, resulting in pro-apoptotic gene expression and eventually phosphatases β-cell death. Protein tyrosine phosphatases (PTPs) are a family of enzymes that regulate f PTPN1 phosphorylative signalling and are associated with the development of T1D. Here, we f PTPN6 observed expression of PTPN6 and PTPN1 in human islets and islets from non-obese f cytokines diabetic (NOD) mice. To clarify the role of these PTPs in β-cells/islets, we took advantage f inflammation Journal of Molecular Endocrinology of CRISPR/Cas9 technology and pharmacological approaches to inactivate both proteins. We identify PTPN6 as a negative regulator of TNF-α-induced β-cell death, through JNK- dependent BCL-2 protein degradation. -

The Regulatory Roles of Phosphatases in Cancer
Oncogene (2014) 33, 939–953 & 2014 Macmillan Publishers Limited All rights reserved 0950-9232/14 www.nature.com/onc REVIEW The regulatory roles of phosphatases in cancer J Stebbing1, LC Lit1, H Zhang, RS Darrington, O Melaiu, B Rudraraju and G Giamas The relevance of potentially reversible post-translational modifications required for controlling cellular processes in cancer is one of the most thriving arenas of cellular and molecular biology. Any alteration in the balanced equilibrium between kinases and phosphatases may result in development and progression of various diseases, including different types of cancer, though phosphatases are relatively under-studied. Loss of phosphatases such as PTEN (phosphatase and tensin homologue deleted on chromosome 10), a known tumour suppressor, across tumour types lends credence to the development of phosphatidylinositol 3--kinase inhibitors alongside the use of phosphatase expression as a biomarker, though phase 3 trial data are lacking. In this review, we give an updated report on phosphatase dysregulation linked to organ-specific malignancies. Oncogene (2014) 33, 939–953; doi:10.1038/onc.2013.80; published online 18 March 2013 Keywords: cancer; phosphatases; solid tumours GASTROINTESTINAL MALIGNANCIES abs in sera were significantly associated with poor survival in Oesophageal cancer advanced ESCC, suggesting that they may have a clinical utility in Loss of PTEN (phosphatase and tensin homologue deleted on ESCC screening and diagnosis.5 chromosome 10) expression in oesophageal cancer is frequent, Cao et al.6 investigated the role of protein tyrosine phosphatase, among other gene alterations characterizing this disease. Zhou non-receptor type 12 (PTPN12) in ESCC and showed that PTPN12 et al.1 found that overexpression of PTEN suppresses growth and protein expression is higher in normal para-cancerous tissues than induces apoptosis in oesophageal cancer cell lines, through in 20 ESCC tissues. -
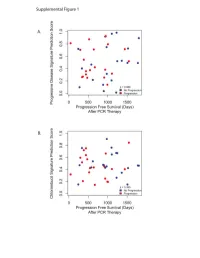
Supplementary Data
Progressive Disease Signature Upregulated probes with progressive disease U133Plus2 ID Gene Symbol Gene Name 239673_at NR3C2 nuclear receptor subfamily 3, group C, member 2 228994_at CCDC24 coiled-coil domain containing 24 1562245_a_at ZNF578 zinc finger protein 578 234224_at PTPRG protein tyrosine phosphatase, receptor type, G 219173_at NA NA 218613_at PSD3 pleckstrin and Sec7 domain containing 3 236167_at TNS3 tensin 3 1562244_at ZNF578 zinc finger protein 578 221909_at RNFT2 ring finger protein, transmembrane 2 1552732_at ABRA actin-binding Rho activating protein 59375_at MYO15B myosin XVB pseudogene 203633_at CPT1A carnitine palmitoyltransferase 1A (liver) 1563120_at NA NA 1560098_at AKR1C2 aldo-keto reductase family 1, member C2 (dihydrodiol dehydrogenase 2; bile acid binding pro 238576_at NA NA 202283_at SERPINF1 serpin peptidase inhibitor, clade F (alpha-2 antiplasmin, pigment epithelium derived factor), m 214248_s_at TRIM2 tripartite motif-containing 2 204766_s_at NUDT1 nudix (nucleoside diphosphate linked moiety X)-type motif 1 242308_at MCOLN3 mucolipin 3 1569154_a_at NA NA 228171_s_at PLEKHG4 pleckstrin homology domain containing, family G (with RhoGef domain) member 4 1552587_at CNBD1 cyclic nucleotide binding domain containing 1 220705_s_at ADAMTS7 ADAM metallopeptidase with thrombospondin type 1 motif, 7 232332_at RP13-347D8.3 KIAA1210 protein 1553618_at TRIM43 tripartite motif-containing 43 209369_at ANXA3 annexin A3 243143_at FAM24A family with sequence similarity 24, member A 234742_at SIRPG signal-regulatory protein gamma -

Activation of Islet Inflammation by Cytokine Signalling in Pancreatic Β
Activation of Islet Inflammation by Cytokine Signalling in Pancreatic b-cells Understanding the Role of Protein Tyrosine Phosphatases William James Stanley ORCID: 0000-0001-8001-2359 Student Number: 515467 Submitted in Total Fulfilment of The Degree of Doctor of Philosophy March 2020 St. Vincent’s Institute of Medical Research Department of Medicine (St. Vincent’s Hospital) The University of Melbourne Abstract: Type 1 diabetes is characterised by the autoimmune destruction of insulin producing β- cells in the islets of Langerhans of the pancreas. Immune cells release pro-inflammatory cytokines such as interferon-g (IFN-g), tumour necrosis factor-a (TNF-a) and interleukin- 1b (IL-1b) into the islet microenvironment which activate phosphorylation cascades and gene expression in b-cells that increase their susceptibility to autoimmune attack and destruction. Protein tyrosine phosphatases (PTPs) regulate phosphorylation based signalling pathways and have previously been shown to negatively regulate IFN-g induced cell death of β-cells in vitro. We previously showed that during immune infiltration to the islet PTPs, including PTPN1 and PTPN6, are rendered catalytically inactive through oxidation resulting in loss of signal regulation. The overall aim of this thesis is to observe if antioxidant treatment can reduce autoimmune development in the NOD/Lt mouse through reduction of oxidised PTPs and dissect the role of PTPN1 and PTPN6 in the regulation of cytotoxic signalling events in the NIT-1 β-cell line and isolated NODPI islets in vitro. Chapter 3 studies the effect of the mitochondrial targeted antioxidant mito-TEMPO on insulitis and diabetes development in the NOD/Lt mouse. Delivery of mito-TEMPO through drinking water reduced levels of oxidised PTPs in the pancreas of NOD/Lt mice but had no effect on the development of insulitis, activity or number of CD8+ and CD4+ T- cells in the periphery in NOD/Lt mice or diabetes development in a diabetes transfer model. -

T Cells Deficient in the Tyrosine Phosphatase SHP-1 Resist Suppression by Regulatory T Cells
T Cells Deficient in the Tyrosine Phosphatase SHP-1 Resist Suppression by Regulatory T Cells This information is current as Emily R. Mercadante and Ulrike M. Lorenz of September 29, 2021. J Immunol published online 26 May 2017 http://www.jimmunol.org/content/early/2017/05/26/jimmun ol.1602171 Downloaded from Supplementary http://www.jimmunol.org/content/suppl/2017/05/26/jimmunol.160217 Material 1.DCSupplemental Why The JI? Submit online. http://www.jimmunol.org/ • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average by guest on September 29, 2021 Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2017 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. Published May 26, 2017, doi:10.4049/jimmunol.1602171 The Journal of Immunology T Cells Deficient in the Tyrosine Phosphatase SHP-1 Resist Suppression by Regulatory T Cells Emily R. Mercadante and Ulrike M. Lorenz The balance between activation of T cells and their suppression by regulatory T cells (Tregs) is dysregulated in autoimmune diseases and cancer. Autoimmune diseases feature T cells that are resistant to suppression by Tregs, whereas in cancer, T cells are unable to mount antitumor responses due to the Treg-enriched suppressive microenvironment. -

Prognostic and Therapeutic Significance of Phosphorylated
Han et al. Blood Cancer Journal (2018) 8:110 DOI 10.1038/s41408-018-0138-8 Blood Cancer Journal ARTICLE Open Access Prognostic and therapeutic significance of phosphorylated STAT3 and protein tyrosine phosphatase-6 in peripheral-T cell lymphoma Jing Jing Han1,MeganO’byrne2,MaryJ.Stenson1, Matthew J. Maurer 2, Linda E. Wellik1,AndrewL.Feldman 3, Ellen D. McPhail3,ThomasE.Witzig1 and Mamta Gupta1,4 Abstract Peripheral T cell lymphomas (PTCL) is a heterogenous group of non-Hodgkin lymphoma and many patients remain refractory to the frontline therapy. Identifying new prognostic markers and treatment is an unmet need in PTCL. We analyzed phospho-STAT3 (pSTAT3) expression in a cohort of 169 PTCL tumors and show overall 38% positivity with varied distribution among PTCL subtypes with 27% (16/59) in PTCL-NOS; 29% (11/38) in AITL, 57% (13/28) in ALK- negative ALCL, and 93% in ALK-pos ALCL (14/15), respectively. Correlative analysis indicated an adverse correlation between pSTAT3 and overall survival (OS). PTPN6, a tyrosine phosphatase and potential negative regulator of STAT3 activity, was suppressed in 62% of PTCL-NOS, 42% of AITL, 60% ALK-neg ALCL, and 86% of ALK-pos ALCL. Loss of PTPN6 combined with pSTAT3 positivity predicted an infwere considered significantferior OS in PTCL cases. In vitro treatment of TCL lines with azacytidine (aza), a DNA methyltransferase inhibitor (DNMTi), restored PTPN6 expression 1234567890():,; 1234567890():,; 1234567890():,; 1234567890():,; and decreased pSTAT3. Combining DNMTi with JAK3 inhibitor resulted in synergistic antitumor activity in SUDHL1 cell line. Overall, our results suggest that PTPN6 and activated STAT3 can be developed as prognostic markers, and the combination of DNMTi and JAK3 inhibitors as a novel treatment for patients with PTCL subtypes. -

Alteration of the MYB Locus in MYST3-Linked Cases
Leukemia (2009) 23, 85–94 & 2009 Macmillan Publishers Limited All rights reserved 0887-6924/09 $32.00 www.nature.com/leu ORIGINAL ARTICLE Genome profiling of acute myelomonocytic leukemia: alteration of the MYB locus in MYST3-linked cases A Murati1,13, C Gervais2,13, N Carbuccia1,13, P Finetti1, N Cervera1, J Ade´laı¨de1, S Struski2, E Lippert3, F Mugneret4, I Tigaud5, D Penther6, C Bastard6, B Poppe7, F Speleman7, L Baranger8, I Luquet9, P Cornillet-Lefebvre9, N Nadal10, F Nguyen-Khac11, CPe´rot12, S Olschwang1, F Bertucci1, M Chaffanet1, M Lessard2, M-J Mozziconacci1 and D Birnbaum1 on behalf of the Groupe Francophone de Cytoge´ne´tique He´matologique (GFCH) 1De´partement d’Oncologie Mole´culaire, Centre de Recherche en Cance´rologie de Marseille, Institut Paoli-Calmettes, Marseille, France; 2Laboratoire d’He´matologie, CHU de Hautepierre, Strasbourg, France; 3Laboratoire d’He´matologie, Hoˆpital Cardiologique-Haut Le´veˆque, Bordeaux, France; 4Laboratoire de Cytoge´ne´tique, CHU du Bocage, Dijon, France; 5Laboratoire d’He´matologie, CHU Lyon Sud, Lyon, France; 6De´partement de Ge´ne´tique, Centre Henri Becquerel, Rouen, France; 7Centre de Ge´ne´tique Me´dicale, Ghent, Belgique; 8Laboratoire de Ge´ne´tique, CHU d’Angers, Angers, France; 9Service de Ge´ne´tique, Hoˆpital Maison Blanche, Reims, France; 10Laboratoire d’He´matologie, Hoˆpital Nord, Saint-Etienne, France; 11Laboratoire de Cytoge´ne´tique He´matologique, Hoˆpital Pitie´-Salpeˆtrie`re, Paris, France and 12Laboratoire de Cytoge´ne´tique Onco-He´matologique, Hoˆpital Saint-Antoine, Paris, France The t(8;16)(p11;p13) is a rare translocation involved in de novo NCOA3 at 20q12.6 MYST3-linked acute myeloid leukemias and therapy-related myelomonocytic and monocytic acute (AMLs) share specific features, such as frequent extramedullary leukemia. -

Mycoplasma Mycoides Subsp
RESEARCH ARTICLE Whole Blood Transcriptome Analysis of Mycoplasma mycoides Subsp. mycoides-Infected Cattle Confirms Immunosuppression but Does Not Reflect Local Inflammation Valérie Rodrigues1,2*, Philippe Holzmuller1,2, Carinne Puech1,2, Hezron Wesonga3, François Thiaucourt1,2, Lucía Manso-Silván1,2 1 CIRAD, UMR15 CMAEE, F-34398 Montpellier, France, 2 INRA, UMR1309 CMAEE, F-34398 Montpellier, France, 3 Kenyan Agricultural Research Institute, Nairobi, Kenya * [email protected] OPEN ACCESS Abstract Citation: Rodrigues V, Holzmuller P, Puech C, Wesonga H, Thiaucourt F, Manso-Silván L (2015) Contagious bovine pleuropneumonia (CBPP), caused by Mycoplasma mycoides subsp. Mycoplasma Whole Blood Transcriptome Analysis of mycoides (Mmm), is a severe respiratory disease of cattle responsible for major economic mycoides Subsp. mycoides-Infected Cattle Confirms Immunosuppression but Does Not Reflect Local losses in sub-Saharan Africa. Disease control relies mainly on the use of empirically attenu- Inflammation. PLoS ONE 10(10): e0139678. ated vaccines that provide limited protection. Thus, understanding the virulence mechanisms doi:10.1371/journal.pone.0139678 used by Mmm as well as the role of the host immune system in disease development, persis- Editor: Bernhard Kaltenboeck, Auburn University, tence, and control is a prerequisite for the development of new, rationally designed control UNITED STATES strategies. The aim of this study was to assess the use of whole blood transcriptome analysis Received: February 12, 2015 to study cattle-Mmm interactions, starting by the characterization of the bovine response to Accepted: September 16, 2015 Mmm infection during the acute form of the disease. For that purpose, we compared the tran- scriptome profile of whole blood from six cattle, before challenge by contact with Mmm- Published: October 2, 2015 infected animals and at the appearance of first clinical signs, using a bovine microarray. -

Neural Progenitor Fate Decision Defects, Cortical Hypoplasia and Behavioral Impairment in Celsr1-Deficient Mice
OPEN Molecular Psychiatry (2018) 23, 723–734 www.nature.com/mp ORIGINAL ARTICLE Neural progenitor fate decision defects, cortical hypoplasia and behavioral impairment in Celsr1-deficient mice C Boucherie1, C Boutin1,5, Y Jossin2, O Schakman3, AM Goffinet1,LRis4, P Gailly3 and F Tissir1 The development of the cerebral cortex is a tightly regulated process that relies on exquisitely coordinated actions of intrinsic and extrinsic cues. Here, we show that the communication between forebrain meninges and apical neural progenitor cells (aNPC) is essential to cortical development, and that the basal compartment of aNPC is key to this communication process. We found that Celsr1, a cadherin of the adhesion G protein coupled receptor family, controls branching of aNPC basal processes abutting the meninges and thereby regulates retinoic acid (RA)-dependent neurogenesis. Loss-of-function of Celsr1 results in a decreased number of endfeet, modifies RA-dependent transcriptional activity and biases aNPC commitment toward self-renewal at the expense of basal progenitor and neuron production. The mutant cortex has a reduced number of neurons, and Celsr1 mutant mice exhibit microcephaly and behavioral abnormalities. Our results uncover an important role for Celsr1 protein and for the basal compartment of neural progenitor cells in fate decision during the development of the cerebral cortex. Molecular Psychiatry (2018) 23, 723–734; doi:10.1038/mp.2017.236; published online 19 December 2017 INTRODUCTION germinal zones,13 its function, after neural tube closure, has not The cerebral cortex is the seat of higher brain functions and its been assessed. formation requires the production and positioning of a right Here, we report that at the onset of neurogenesis, the Celsr1 number and diversity of neurons for intricate circuits assembly. -

PTPN6 Sirna Set I PTPN6 Sirna Set I
Catalog # Aliquot Size P33-911-05 3 x 5 nmol P33-911-20 3 x 20 nmol P33-911-50 3 x 50 nmol PTPN6 siRNA Set I siRNA duplexes targeted against three exon regions Catalog # P33-911 Lot # Z2085-25 Specificity Formulation PTPN6 siRNAs are designed to specifically knock-down The siRNAs are supplied as a lyophilized powder and human PTPN6 expression. shipped at room temperature. Product Description Reconstitution Protocol PTPN6 siRNA is a pool of three individual synthetic siRNA Briefly centrifuge the tubes (maximum RCF 4,000g) to duplexes designed to knock-down human PTPN6 mRNA collect lyophilized siRNA at the bottom of the tube. expression. Each siRNA is 19-25 bases in length. The gene Resuspend the siRNA in 50 µl of DEPC-treated water accession number is NM_080548. (supplied by researcher), which results in a 1x stock solution (10 µM). Gently pipet the solution 3-5 times to mix Gene Aliases and avoid the introduction of bubbles. Optional: aliquot SHP1; SHP-1; HCP; HCPH; HPTP1C; PTP-1C; SHP-1L; SH-PTP1 1x stock solutions for storage. Storage and Stability Related Products The lyophilized powder is stable for at least 4 weeks at room temperature. It is recommended that the Product Name Catalog Number lyophilized and resuspended siRNAs are stored at or PTPN1 (PTP1B), Active P28-20G below -20oC. After resuspension, siRNA stock solutions ≥2 PTPN11 (SHP2), Active P38-21G µM can undergo up to 50 freeze-thaw cycles without PTPN12 (PTP-PEST), Active P39-21G significant degradation. For long-term storage, it is PTPN13 (FAP-1), Active P40-21G recommended that the siRNA is stored at -70oC. -

Live-Cell Imaging Rnai Screen Identifies PP2A–B55α and Importin-Β1 As Key Mitotic Exit Regulators in Human Cells
LETTERS Live-cell imaging RNAi screen identifies PP2A–B55α and importin-β1 as key mitotic exit regulators in human cells Michael H. A. Schmitz1,2,3, Michael Held1,2, Veerle Janssens4, James R. A. Hutchins5, Otto Hudecz6, Elitsa Ivanova4, Jozef Goris4, Laura Trinkle-Mulcahy7, Angus I. Lamond8, Ina Poser9, Anthony A. Hyman9, Karl Mechtler5,6, Jan-Michael Peters5 and Daniel W. Gerlich1,2,10 When vertebrate cells exit mitosis various cellular structures can contribute to Cdk1 substrate dephosphorylation during vertebrate are re-organized to build functional interphase cells1. This mitotic exit, whereas Ca2+-triggered mitotic exit in cytostatic-factor- depends on Cdk1 (cyclin dependent kinase 1) inactivation arrested egg extracts depends on calcineurin12,13. Early genetic studies in and subsequent dephosphorylation of its substrates2–4. Drosophila melanogaster 14,15 and Aspergillus nidulans16 reported defects Members of the protein phosphatase 1 and 2A (PP1 and in late mitosis of PP1 and PP2A mutants. However, the assays used in PP2A) families can dephosphorylate Cdk1 substrates in these studies were not specific for mitotic exit because they scored pro- biochemical extracts during mitotic exit5,6, but how this relates metaphase arrest or anaphase chromosome bridges, which can result to postmitotic reassembly of interphase structures in intact from defects in early mitosis. cells is not known. Here, we use a live-cell imaging assay and Intracellular targeting of Ser/Thr phosphatase complexes to specific RNAi knockdown to screen a genome-wide library of protein substrates is mediated by a diverse range of regulatory and targeting phosphatases for mitotic exit functions in human cells. We subunits that associate with a small group of catalytic subunits3,4,17. -

The Phosphatase JKAP/DUSP22 Inhibits T-Cell Receptor Signalling and Autoimmunity by Inactivating Lck
ARTICLE Received 8 Aug 2013 | Accepted 11 Mar 2014 | Published 9 Apr 2014 DOI: 10.1038/ncomms4618 The phosphatase JKAP/DUSP22 inhibits T-cell receptor signalling and autoimmunity by inactivating Lck Ju-Pi Li1, Chia-Yu Yang1,Ã, Huai-Chia Chuang1,Ã, Joung-Liang Lan2,3,4,Ã, Der-Yuan Chen2,5,Ã, Yi-Ming Chen2,5,Ã, Xiaohong Wang6, Alice J. Chen6,7, John W. Belmont7 & Tse-Hua Tan1,6 JNK pathway-associated phosphatase (JKAP, also known as DUSP22 or JSP-1) is a JNK activator. The in vivo role of JKAP in immune regulation remains unclear. Here we report that JKAP directly inactivates Lck by dephosphorylating tyrosine-394 residue during T-cell receptor (TCR) signalling. JKAP-knockout T cells display enhanced cell proliferation and cytokine production. JKAP-knockout mice show enhanced T-cell-mediated immune responses and are more susceptible to experimental autoimmune encephalomyelitis (EAE). In addition, the recipient mice that are adoptively transferred with JKAP-knockout T cells show exacerbated EAE symptoms. Aged JKAP-knockout mice spontaneously develop inflammation and autoimmunity. Thus, our results indicate that JKAP is an important phosphatase that inactivates Lck in the TCR signalling turn-off stage, leading to suppression of T-cell-mediated immunity and autoimmunity. 1 Immunology Research Center, National Health Research Institutes, Zhunan 35053, Taiwan. 2 Division of Allergy, Immunology, and Rheumatology, Taichung Veterans General Hospital, Taichung 40705, Taiwan. 3 Department of Medicine, China Medical University, Taichung 40402, Taiwan. 4 Division of Rheumatology & Immunology, Department of Internal Medicine, China Medical University Hospital, Taichung 40402, Taiwan. 5 Faculty of Medicine, National Yang-Ming University, Taipei 11221, Taiwan.