View U.S. Patent Application Publication No. US-2020-0056988
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Treatment of Herpes Simplex Virus Epithelial Keratitis
THE TREATMENT OF HERPES SIMPLEX VIRUS EPITHELIAL KERATITIS BY Kirk R. Wilhelmus, MD, MPH ABSTRACT Purpose: Epithelial keratitis is the most common presentation of ocular infection by herpes simplex virus (HSV). Quantitative assessment of available therapy is needed to guide evidence-based ophthalmology. This study aimed to compare the efficacy of various treatments for dendritic or geographic HSV epithelial keratitis and to evaluate the role of various clinical characteristics on epithelial healing. Methods: Following a systematic review of the literature, information from clinical trials of HSV dendritic or geographic epithelial keratitis was extracted, and the methodological quality of each study was scored. Methods of epithelial cau- terization and curettage were grouped as relatively equivalent physicochemical therapy, and solution and ointment for- mulations of a given topical antiviral agent were combined. The proportion healed with 1 week of therapy, a scheduled follow-up day that approximated the average time of resolution with antiviral therapy, was selected as the primary out- come based on a masked evaluation of maximum treatment differences in published healing curves. The proportion healed at 14 days was recorded as supplemental information. Fixed-effects and random-effects meta-analysis models were used to obtain summary estimates by pooling results from comparative treatment trials. Hypotheses about which prognostic factors might affect epithelial healing during antiviral therapy were developed by multivariate analysis of the Herpetic Eye Disease Study dataset. Results: After excluding 48 duplicate reports, 14 nonrandomized studies, 15 studies with outdated or similar treatments, and 29 trials lacking sufficient data on healing or accessibility, 76 primary reports were identified. -
(12) United States Patent (10) Patent No.: US 8,993,581 B2 Perrine Et Al
US00899.3581B2 (12) United States Patent (10) Patent No.: US 8,993,581 B2 Perrine et al. (45) Date of Patent: Mar. 31, 2015 (54) METHODS FOR TREATINGVIRAL (58) Field of Classification Search DSORDERS CPC ... A61K 31/00; A61K 31/166; A61K 31/185: A61K 31/233; A61K 31/522: A61K 38/12: (71) Applicant: Trustees of Boston University, Boston, A61K 38/15: A61K 45/06 MA (US) USPC ........... 514/263.38, 21.1, 557, 565, 575, 617; 424/2011 (72) Inventors: Susan Perrine, Weston, MA (US); Douglas Faller, Weston, MA (US) See application file for complete search history. (73) Assignee: Trustees of Boston University, Boston, (56) References Cited MA (US) U.S. PATENT DOCUMENTS (*) Notice: Subject to any disclaimer, the term of this 3,471,513 A 10, 1969 Chinn et al. patent is extended or adjusted under 35 3,904,612 A 9/1975 Nagasawa et al. U.S.C. 154(b) by 0 days. (Continued) (21) Appl. No.: 13/915,092 FOREIGN PATENT DOCUMENTS (22) Filed: Jun. 11, 2013 CA 1209037 A 8, 1986 CA 2303268 A1 4f1995 (65) Prior Publication Data (Continued) US 2014/OO45774 A1 Feb. 13, 2014 OTHER PUBLICATIONS Related U.S. Application Data (63) Continuation of application No. 12/890,042, filed on PCT/US 10/59584 Search Report and Written Opinion mailed Feb. Sep. 24, 2010, now abandoned. 11, 2011. (Continued) (60) Provisional application No. 61/245,529, filed on Sep. 24, 2009, provisional application No. 61/295,663, filed on Jan. 15, 2010. Primary Examiner — Savitha Rao (74) Attorney, Agent, or Firm — Nixon Peabody LLP (51) Int. -
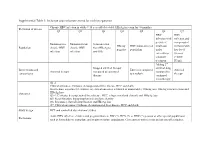
Inclusion and Exclusion Criteria for Each Key Question
Supplemental Table 1: Inclusion and exclusion criteria for each key question Chronic HBV infection in adults ≥ 18 year old (detectable HBsAg in serum for >6 months) Definition of disease Q1 Q2 Q3 Q4 Q5 Q6 Q7 HBV HBV infection with infection and persistent compensated Immunoactive Immunotolerant Seroconverted HBeAg HBV mono-infected viral load cirrhosis with Population chronic HBV chronic HBV from HBeAg to negative population under low level infection infection anti-HBe entecavir or viremia tenofovir (<2000 treatment IU/ml) Adding 2nd Stopped antiviral therapy antiviral drug Interventions and Entecavir compared Antiviral Antiviral therapy compared to continued compared to comparisons to tenofovir therapy therapy continued monotherapy Q1-2: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Intermediate outcomes (if evidence on clinical outcomes is limited or unavailable): HBsAg loss, HBeAg seroconversion and Outcomes HBeAg loss Q3-4: Cirrhosis, decompensated liver disease, HCC, relapse (viral and clinical) and HBsAg loss Q5: Renal function, hypophosphatemia and bone density Q6: Resistance, flare/decompensation and HBeAg loss Q7: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Study design RCT and controlled observational studies Acute HBV infection, children and pregnant women, HIV (+), HCV (+) or HDV (+) persons or other special populations Exclusions such as hemodialysis, transplant, and treatment failure populations. Co treatment with steroids and uncontrolled studies. Supplemental Table 2: Detailed Search Strategy: Ovid Database(s): Embase 1988 to 2014 Week 37, Ovid MEDLINE(R) In-Process & Other Non- Indexed Citations and Ovid MEDLINE(R) 1946 to Present, EBM Reviews - Cochrane Central Register of Controlled Trials August 2014, EBM Reviews - Cochrane Database of Systematic Reviews 2005 to July 2014 Search Strategy: # Searches Results 1 exp Hepatitis B/dt 26410 ("hepatitis B" or "serum hepatitis" or "hippie hepatitis" or "injection hepatitis" or 2 178548 "hepatitis type B").mp. -

WO 2014/167554 A2 16 October 2014 (16.10.2014) P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2014/167554 A2 16 October 2014 (16.10.2014) P O P C T (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every A61K 47/10 (2006.01) kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, (21) International Application Number: BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, PCT/IB20 14/060675 DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (22) International Filing Date: HN, HR, HU, ID, IL, IN, IR, IS, JP, KE, KG, KN, KP, KR, 12 April 2014 (12.04.2014) KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, (25) Filing Language: English OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, (26) Publication Language: English SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, (30) Priority Data: zw. 1103/DEL/2013 12 April 2013 (12.04.2013) IN (84) Designated States (unless otherwise indicated, for every (71) Applicant: VYOME BIOSCIENCES PVT. LTD. kind of regional protection available): ARIPO (BW, GH, [IN/IN]; 459 F.I.E, First Floor, Patparganj Industrial Area, GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, SZ, TZ, New Delhi 110092 (IN). -

)&F1y3x PHARMACEUTICAL APPENDIX to THE
)&f1y3X PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE )&f1y3X PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 3 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABAMECTIN 65195-55-3 ACTODIGIN 36983-69-4 ABANOQUIL 90402-40-7 ADAFENOXATE 82168-26-1 ABCIXIMAB 143653-53-6 ADAMEXINE 54785-02-3 ABECARNIL 111841-85-1 ADAPALENE 106685-40-9 ABITESARTAN 137882-98-5 ADAPROLOL 101479-70-3 ABLUKAST 96566-25-5 ADATANSERIN 127266-56-2 ABUNIDAZOLE 91017-58-2 ADEFOVIR 106941-25-7 ACADESINE 2627-69-2 ADELMIDROL 1675-66-7 ACAMPROSATE 77337-76-9 ADEMETIONINE 17176-17-9 ACAPRAZINE 55485-20-6 ADENOSINE PHOSPHATE 61-19-8 ACARBOSE 56180-94-0 ADIBENDAN 100510-33-6 ACEBROCHOL 514-50-1 ADICILLIN 525-94-0 ACEBURIC ACID 26976-72-7 ADIMOLOL 78459-19-5 ACEBUTOLOL 37517-30-9 ADINAZOLAM 37115-32-5 ACECAINIDE 32795-44-1 ADIPHENINE 64-95-9 ACECARBROMAL 77-66-7 ADIPIODONE 606-17-7 ACECLIDINE 827-61-2 ADITEREN 56066-19-4 ACECLOFENAC 89796-99-6 ADITOPRIM 56066-63-8 ACEDAPSONE 77-46-3 ADOSOPINE 88124-26-9 ACEDIASULFONE SODIUM 127-60-6 ADOZELESIN 110314-48-2 ACEDOBEN 556-08-1 ADRAFINIL 63547-13-7 ACEFLURANOL 80595-73-9 ADRENALONE -

Topical and Systemic Antifungal Therapy for Chronic Rhinosinusitis (Protocol)
CORE Metadata, citation and similar papers at core.ac.uk Provided by University of East Anglia digital repository Cochrane Database of Systematic Reviews Topical and systemic antifungal therapy for chronic rhinosinusitis (Protocol) Head K, Sacks PL, Chong LY, Hopkins C, Philpott C Head K, Sacks PL, Chong LY, Hopkins C, Philpott C. Topical and systemic antifungal therapy for chronic rhinosinusitis. Cochrane Database of Systematic Reviews 2016, Issue 11. Art. No.: CD012453. DOI: 10.1002/14651858.CD012453. www.cochranelibrary.com Topical and systemic antifungal therapy for chronic rhinosinusitis (Protocol) Copyright © 2016 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd. TABLE OF CONTENTS HEADER....................................... 1 ABSTRACT ...................................... 1 BACKGROUND .................................... 1 OBJECTIVES ..................................... 3 METHODS ...................................... 3 ACKNOWLEDGEMENTS . 8 REFERENCES ..................................... 9 APPENDICES ..................................... 10 CONTRIBUTIONSOFAUTHORS . 25 DECLARATIONSOFINTEREST . 26 SOURCESOFSUPPORT . 26 NOTES........................................ 26 Topical and systemic antifungal therapy for chronic rhinosinusitis (Protocol) i Copyright © 2016 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd. [Intervention Protocol] Topical and systemic antifungal therapy for chronic rhinosinusitis Karen Head1, Peta-Lee Sacks2, Lee Yee Chong1, Claire Hopkins3, Carl Philpott4 1UK Cochrane Centre, -

A Screening-Based Approach to Circumvent Tumor Microenvironment
JBXXXX10.1177/1087057113501081Journal of Biomolecular ScreeningSingh et al. 501081research-article2013 Original Research Journal of Biomolecular Screening 2014, Vol 19(1) 158 –167 A Screening-Based Approach to © 2013 Society for Laboratory Automation and Screening DOI: 10.1177/1087057113501081 Circumvent Tumor Microenvironment- jbx.sagepub.com Driven Intrinsic Resistance to BCR-ABL+ Inhibitors in Ph+ Acute Lymphoblastic Leukemia Harpreet Singh1,2, Anang A. Shelat3, Amandeep Singh4, Nidal Boulos1, Richard T. Williams1,2*, and R. Kiplin Guy2,3 Abstract Signaling by the BCR-ABL fusion kinase drives Philadelphia chromosome–positive acute lymphoblastic leukemia (Ph+ ALL) and chronic myelogenous leukemia (CML). Despite their clinical activity in many patients with CML, the BCR-ABL kinase inhibitors (BCR-ABL-KIs) imatinib, dasatinib, and nilotinib provide only transient leukemia reduction in patients with Ph+ ALL. While host-derived growth factors in the leukemia microenvironment have been invoked to explain this drug resistance, their relative contribution remains uncertain. Using genetically defined murine Ph+ ALL cells, we identified interleukin 7 (IL-7) as the dominant host factor that attenuates response to BCR-ABL-KIs. To identify potential combination drugs that could overcome this IL-7–dependent BCR-ABL-KI–resistant phenotype, we screened a small-molecule library including Food and Drug Administration–approved drugs. Among the validated hits, the well-tolerated antimalarial drug dihydroartemisinin (DHA) displayed potent activity in vitro and modest in vivo monotherapy activity against engineered murine BCR-ABL-KI–resistant Ph+ ALL. Strikingly, cotreatment with DHA and dasatinib in vivo strongly reduced primary leukemia burden and improved long-term survival in a murine model that faithfully captures the BCR-ABL-KI–resistant phenotype of human Ph+ ALL. -

Herpes Simplex Virus
HSV Herpes simplex virus HSV (Herpes simplex virus) can be spread when an infected person is producing and shedding the virus. Herpes simplex can be spread through contact with saliva, such as sharing drinks. Symptoms of herpes simplex virus infection include watery blisters in the skin or mucous membranes of the mouth, lips or genitals. Lesions heal with ascab characteristic of herpetic disease. As neurotropic and neuroinvasive viruses, HSV-1 and -2 persist in the body by becoming latent and hiding from the immune system in the cell bodies of neurons. After the initial or primary infection, some infected people experience sporadic episodes of viral reactivation or outbreaks. www.MedChemExpress.com 1 HSV Inhibitors (Z)-Capsaicin 1-Docosanol (Zucapsaicin; Civamide; cis-Capsaicin) Cat. No.: HY-B1583 (Behenyl alcohol) Cat. No.: HY-B0222 (Z)-Capsaicin is the cis isomer of capsaicin, acts 1-Docosanol is a saturated fatty alcohol used as an orally active TRPV1 agonist, and is used in traditionally as an emollient, emulsifier, and the research of neuropathic pain. thickener in cosmetics, and nutritional supplement; inhibitor of lipid-enveloped viruses including herpes simplex. Purity: 99.96% Purity: ≥98.0% Clinical Data: Launched Clinical Data: Launched Size: 10 mM × 1 mL, 10 mg, 50 mg Size: 500 mg 2-Deoxy-D-glucose 20(R)-Ginsenoside Rh2 (2-DG; 2-Deoxy-D-arabino-hexose; D-Arabino-2-deoxyhexose) Cat. No.: HY-13966 Cat. No.: HY-N1401 2-Deoxy-D-glucose is a glucose analog that acts as 20(R)-Ginsenoside Rh2, a matrix a competitive inhibitor of glucose metabolism, metalloproteinase (MMP) inhibitor, acts as a inhibiting glycolysis via its actions on hexokinase. -

Stems for Nonproprietary Drug Names
USAN STEM LIST STEM DEFINITION EXAMPLES -abine (see -arabine, -citabine) -ac anti-inflammatory agents (acetic acid derivatives) bromfenac dexpemedolac -acetam (see -racetam) -adol or analgesics (mixed opiate receptor agonists/ tazadolene -adol- antagonists) spiradolene levonantradol -adox antibacterials (quinoline dioxide derivatives) carbadox -afenone antiarrhythmics (propafenone derivatives) alprafenone diprafenonex -afil PDE5 inhibitors tadalafil -aj- antiarrhythmics (ajmaline derivatives) lorajmine -aldrate antacid aluminum salts magaldrate -algron alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol combined alpha and beta blockers labetalol medroxalol -amidis antimyloidotics tafamidis -amivir (see -vir) -ampa ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) subgroup: -ampanel antagonists becampanel -ampator modulators forampator -anib angiogenesis inhibitors pegaptanib cediranib 1 subgroup: -siranib siRNA bevasiranib -andr- androgens nandrolone -anserin serotonin 5-HT2 receptor antagonists altanserin tropanserin adatanserin -antel anthelmintics (undefined group) carbantel subgroup: -quantel 2-deoxoparaherquamide A derivatives derquantel -antrone antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine antineoplastics (arabinofuranosyl derivatives) fazarabine fludarabine aril-, -aril, -aril- antiviral (arildone derivatives) pleconaril arildone fosarilate -arit antirheumatics (lobenzarit type) lobenzarit clobuzarit -arol anticoagulants (dicumarol type) dicumarol -

National OTC Medicines List
National OTC Medicines List ‐ DraŌ 01 DRAFT National OTC Medicines List Draft 01 Ministry of Public Health of Lebanon This list was prepared under the guidance of His Excellency Minister Waêl Abou Faour andDRAFT the supervision of the Director General Dr. Walid Ammar. Editors Rita KARAM, Pharm D. PhD. Myriam WATFA, Pharm D Ghassan HAMADEH, MD.CPE FOREWORD According to the French National Agency for Medicines and Health Products Safety (ANSM), Over-the-counter (OTC) drugs are medicines that are accessible to patients in pharmacies, based on criteria set to safeguard patients’ safety. Due to their therapeutic class, these medicines could be dispensed without physician’s intervention for diagnostic, treatment initiation or maintenance purposes. Moreover, their dosage, treatment period and Package Insert Leaflet should be suitable for OTC classification. The packaging size should be in accordance with the dosage and treatment period. According to ArticleDRAFT 43 of the Law No.367 issued in 1994 related to the pharmacy practice, and the amendment of Articles 46 and 47 by Law No.91 issued in 2010, pharmacists do not have the right to dispense any medicine that is not requested by a unified prescription, unless the medicine is mentioned in a list which is established by pharmacists and physicians’ syndicates. In this regard, the Ministry of Public Health (MoPH) developed the National OTC Medicines List, and presentedit in a scientific, objective, reliable, and accessible listing. The OTC List was developed by a team of pharmacists and physicians from the Ministry of Public Health (MoPH). In order to ensure a safe and effective self- medicationat the pharmacy level, several pharmaceutical categories (e.g. -

Antagonists of Il-6 to Prevent Or Treat
(19) TZZ ¥_ _T (11) EP 2 376 126 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61K 39/00 (2006.01) C07K 16/24 (2006.01) 20.12.2017 Bulletin 2017/51 (86) International application number: (21) Application number: 09830695.4 PCT/US2009/006266 (22) Date of filing: 24.11.2009 (87) International publication number: WO 2010/065077 (10.06.2010 Gazette 2010/23) (54) ANTAGONISTS OF IL-6 TO PREVENT OR TREAT THROMBOSIS ANTAGONISTEN VON IL-6 ZUR PRÄVENTION ODER BEHANDLUNG VON THROMBOSE ANTAGONISTES D IL-6 POUR PRÉVENIR OU TRAITER LA THROMBOSE (84) Designated Contracting States: • MATSUYAMA MASASHI ET AL: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR "Anti-interleukin-6 receptor antibody HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL (tocilizumab) treatment of multicentric PT RO SE SI SK SM TR Castleman’s disease", INTERNAL MEDICINE, JAPANESE SOCIETY OF INTERNAL MEDICINE, (30) Priority: 05.02.2009 US 366567 TOKYO, JP; BIOSCIENCES INFORMATION 14.07.2009 US 502581 SERVICE, PHILADELPHIA, PA, US, vol. 46, 1 25.11.2008 US 117811 P January 2007 (2007-01-01), pages 771-774, 25.11.2008 US 117861 P XP002696891, ISSN: 0918-2918, DOI: 24.02.2009 US 391717 10.2169/INTERNALMEDICINE.46.6262 06.03.2009 US 399156 • EMILLE D ET AL: "ADMINISTRATION OF AN 25.11.2008 US 117839 P ANTI-INTERLEUKIN-6 MONOCLONAL 24.02.2009 US 391615 ANTIBODY TO PATIENTS WITH ACQUIRED IMUNODEFICIENCY SYNDROME AND (43) Date of publication of application: LYMPHOMA: EFFECT ON LYMPHOMA GROWTH 19.10.2011 Bulletin 2011/42 AND ON B CLINICAL SYMPTOMS", BLOOD, AMERICAN SOCIETY OFHEMATOLOGY, US, vol. -

Certain Pharmaceuticals and Intermediate Chemicals: Identification of Applicable 6-Digit HS Subheadings for Products Covered By
Second Interim Report to the Public on CERTAIN PHARMACEUTICALS AND INTERMEDIATE CHEMICALS: IDENTIFI CATION OF APPLICABLE 6-DIGIT HS SUBHEADINGS. .· FOR PRODUCTS ·coVERED BY THE PROPOSED URUGUAY ROUND PHARMACEUTICAL A,GREEMENT Investigation No. 332-322 USITC PUBLICATION 2500 APRIL 1992 ,f~· United States International Trade Commission . ·J, Washington, DC 20436 . ·-~}~~:; . _:, -~;<$f ·!•;. UNITED STATES INTERNATIONAL TRADE COMMISSION COMMISSIONERS Don E. Newquist, Chairman Anne E. Brunsdale, Vice Chairman David B. Rohr Carol T. Crawford Janet A. Nuzum Peter S. Watson Office of Operations Charles W. Ervin, Director Office of Industries Rohen A. Rogowsky, Director This report was prepared principally by Eli?.abcth R. Ncsbiu Project Leader Aimison Jonnard Edward Matusik David Michels James Raftery Office of Industries David Beck Office of Tariff Affairs and Trade Agreements With assistance from Paul Daniels James Gill Office ofInformation Resources Management Under the direction of John J. Gersic, Chief Energy & Chemicals Division and Edmund D. Cappuccilli, Chief Energy, Petroleum, Benzenoid Chemicals, and Rubber and Plastics Branch · Energy & Chemicals Division Additional assistance provided by Brenda Carroll and Keilh Hipp With special assistance from U.S. Customs Service Address all communications to Kenneth R. Mason, Secreta·ry to the Commission United States International Trade Commission Washington, DC 20436 PREFACE This report is the second of two interim reports to the public pertaining to Commission investigation No. 332-322, entitled Certain Pharmaceuticals and Intermediate Chemicals; Identification of Applicable 6-Digit HS Subheadings for Products Covered by the Proposed Uruguay Round Pharmaceutical Agreement. The investigation was instituted following receipt of a letter from the United States Trade Representative (USTR) on January 27, 1992, requesting that the Commission conduct an investigation under section 332(g) of the Tariff Act of 1930 (see appendix A for a copy of the USTR' s request)·.