CLASS Actinopterygii
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Sturgeon Chub (Macrhybopsis Gelida): a Technical Conservation Assessment
Sturgeon Chub (Macrhybopsis gelida): A Technical Conservation Assessment Prepared for the USDA Forest Service, Rocky Mountain Region, Species Conservation Project August 31, 2004 Frank J. Rahel and Laura A. Thel Department of Zoology and Physiology University of Wyoming, Laramie, Wyoming 82071 Peer Review Administered by American Fisheries Society Rahel, F.J. and L.A. Thel. (2004, August 31). Sturgeon Chub (Macrhybopsis gelida): a technical conservation assessment. [Online]. USDA Forest Service, Rocky Mountain Region. Available: http://www.fs.fed.us/r2/ projects/scp/assessments/sturgeonchub.pdf [date of access]. ACKNOWLEDGEMENTS We thank biologists from Colorado, Kansas, Nebraska, South Dakota, and Wyoming, and from the national forests and national grasslands within Region 2 who provided information about sturgeon chub within their jurisdictions. We especially thank Gregory Hayward and Richard Vacirca of the USDA Forest Service for their review of this species assessment. Comments also were provided by two anonymous reviewers. David B. McDonald of the University of Wyoming provided the population demographic matrix analysis. AUTHORS’ BIOGRAPHIES Frank J. Rahel is a professor in the Department of Zoology and Physiology at the University of Wyoming where he teaches courses in fi sheries management, ichthyology, and conservation biology. His research interests are centered around fi sh ecology and the infl uence of anthropogenic disturbances on fi sh assemblages. Laura A. Thel is a graduate research assistant in the Department of Zoology and Physiology at the University of Wyoming with research interests involving stream ecology, hydrology, and landscape ecology, especially as these are related to the management of native fi shes. COVER PHOTO CREDIT Sturgeon Chub (Macrhybopsis gelida). -

The Ecology and Management of the European Grayling Thymallus Thymallus (Linnaeus)
The ecology and management of the European grayling Thymallus thymallus (Linnaeus). Interim report Item Type monograph Authors Ingram, A.; Ibbotson, A.; Gallagher, M. Publisher Institute of Freshwater Ecology Download date 03/10/2021 22:03:11 Link to Item http://hdl.handle.net/1834/24874 The Ecology and Management of the European Grayling Thymallus thymallus (Linnaeus) Interim Report Ingram A Ibbotson A Gallagher M The Ecology and Management of the European Grayling Thymallus thymallus (Linnaeus) Interim Report Ingram A Ibbotson A Gallagher M INTELLECTUAL PROPERTY RIGHTS CONFIDENTIALITY STATEMENT 'In accordance with our normal practice, this report is for the use only of the party to whom it is addressed, and no responsibility is accepted to any third party for the whole or any part of its contents. Neither the whole nor any part of this report or any reference thereto may be included in any published document, circular or statement, nor published or referred to in any way without our written approval of the form and context in which it may appear.' 11 CHAPTER 1 1 Overall aim A collaborative research and development project between the Institute of Freshwater Ecology and the Environment Agency in the UK, to review the ecology, status and management of grayling in order to provide recommendations for future management of grayling fisheries in England and Wales. 2 Objectives • To review grayling ecology, status and management practice in concentrating on England and Wales but including published literature from Europe and North America, where appropriate. • To recommend best management practices on the basis of objective 1 and produce a guidance leaflet for internal and external circulation which promotes the key issues. -

Notropis Girardi) and Peppered Chub (Macrhybopsis Tetranema)
Arkansas River Shiner and Peppered Chub SSA, October 2018 Species Status Assessment Report for the Arkansas River Shiner (Notropis girardi) and Peppered Chub (Macrhybopsis tetranema) Arkansas River shiner (bottom left) and peppered chub (top right - two fish) (Photo credit U.S. Fish and Wildlife Service) Arkansas River Shiner and Peppered Chub SSA, October 2018 Version 1.0a October 2018 U.S. Fish and Wildlife Service Region 2 Albuquerque, NM This document was prepared by Angela Anders, Jennifer Smith-Castro, Peter Burck (U.S. Fish and Wildlife Service (USFWS) – Southwest Regional Office) Robert Allen, Debra Bills, Omar Bocanegra, Sean Edwards, Valerie Morgan (USFWS –Arlington, Texas Field Office), Ken Collins, Patricia Echo-Hawk, Daniel Fenner, Jonathan Fisher, Laurence Levesque, Jonna Polk (USFWS – Oklahoma Field Office), Stephen Davenport (USFWS – New Mexico Fish and Wildlife Conservation Office), Mark Horner, Susan Millsap (USFWS – New Mexico Field Office), Jonathan JaKa (USFWS – Headquarters), Jason Luginbill, and Vernon Tabor (Kansas Field Office). Suggested reference: U.S. Fish and Wildlife Service. 2018. Species status assessment report for the Arkansas River shiner (Notropis girardi) and peppered chub (Macrhybopsis tetranema), version 1.0, with appendices. October 2018. Albuquerque, NM. 172 pp. Arkansas River Shiner and Peppered Chub SSA, October 2018 EXECUTIVE SUMMARY ES.1 INTRODUCTION (CHAPTER 1) The Arkansas River shiner (Notropis girardi) and peppered chub (Macrhybopsis tetranema) are restricted primarily to the contiguous river segments of the South Canadian River basin spanning eastern New Mexico downstream to eastern Oklahoma (although the peppered chub is less widespread). Both species have experienced substantial declines in distribution and abundance due to habitat destruction and modification from stream dewatering or depletion from diversion of surface water and groundwater pumping, construction of impoundments, and water quality degradation. -

Burbot Management Plan
BURBOT MANAGEMENT PLAN DEPARTMENT OF INLAND FISHERIES AND WILDLIFE DIVISION OF FISHERIES AND HATCHERIES PREPARED BY SCOTT A. ROY ASSISTANT REGIONAL FISHERIES BIOLOGIST REGION E MARCH 2001 BURBOT LIFE HISTORY The burbot, Lota lota (Linnaeus), is a unique member of the cod family. It is the only species in the family, which spends its entire life in fresh water. However, the burbot is similar to its marine relatives in that its distribution is circumpolar. It can be found in cool, fresh waters throughout northern Europe, Asia and North America. In North America its range extends as far south as the northern tier of States across the United States. In Maine, the burbot is commonly known as the cusk, although in other areas it is also called the ling, eelpout, loche and lawyer. Unlike the salmonids and Maine’s other coldwater species, the burbot is not noted for its grace and beauty. The body is elongate, almost eel-shaped, with long, soft-rayed dorsal and anal fins that meet a rounded tail. Although it is smooth and slimy to the touch, the skin is embedded with very small, cycloid scales. The head of the burbot is broad and somewhat flattened. It has a large mouth containing several rows of small teeth on the jaws. A single, whisker-like barbell protrudes from the tip of the chin. There are no obvious external differences between males and females. In general, adults are olive brown to dark brown on the back and sides. This background is overlaid with distinctive patterns of dark brown or black markings and spots. -

Indiana Species April 2007
Fishes of Indiana April 2007 The Wildlife Diversity Section (WDS) is responsible for the conservation and management of over 750 species of nongame and endangered wildlife. The list of Indiana's species was compiled by WDS biologists based on accepted taxonomic standards. The list will be periodically reviewed and updated. References used for scientific names are included at the bottom of this list. ORDER FAMILY GENUS SPECIES COMMON NAME STATUS* CLASS CEPHALASPIDOMORPHI Petromyzontiformes Petromyzontidae Ichthyomyzon bdellium Ohio lamprey lampreys Ichthyomyzon castaneus chestnut lamprey Ichthyomyzon fossor northern brook lamprey SE Ichthyomyzon unicuspis silver lamprey Lampetra aepyptera least brook lamprey Lampetra appendix American brook lamprey Petromyzon marinus sea lamprey X CLASS ACTINOPTERYGII Acipenseriformes Acipenseridae Acipenser fulvescens lake sturgeon SE sturgeons Scaphirhynchus platorynchus shovelnose sturgeon Polyodontidae Polyodon spathula paddlefish paddlefishes Lepisosteiformes Lepisosteidae Lepisosteus oculatus spotted gar gars Lepisosteus osseus longnose gar Lepisosteus platostomus shortnose gar Amiiformes Amiidae Amia calva bowfin bowfins Hiodonotiformes Hiodontidae Hiodon alosoides goldeye mooneyes Hiodon tergisus mooneye Anguilliformes Anguillidae Anguilla rostrata American eel freshwater eels Clupeiformes Clupeidae Alosa chrysochloris skipjack herring herrings Alosa pseudoharengus alewife X Dorosoma cepedianum gizzard shad Dorosoma petenense threadfin shad Cypriniformes Cyprinidae Campostoma anomalum central stoneroller -
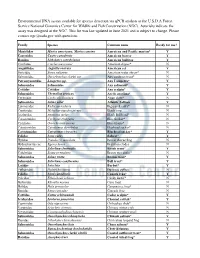
Edna Assay Development
Environmental DNA assays available for species detection via qPCR analysis at the U.S.D.A Forest Service National Genomics Center for Wildlife and Fish Conservation (NGC). Asterisks indicate the assay was designed at the NGC. This list was last updated in June 2021 and is subject to change. Please contact [email protected] with questions. Family Species Common name Ready for use? Mustelidae Martes americana, Martes caurina American and Pacific marten* Y Castoridae Castor canadensis American beaver Y Ranidae Lithobates catesbeianus American bullfrog Y Cinclidae Cinclus mexicanus American dipper* N Anguillidae Anguilla rostrata American eel Y Soricidae Sorex palustris American water shrew* N Salmonidae Oncorhynchus clarkii ssp Any cutthroat trout* N Petromyzontidae Lampetra spp. Any Lampetra* Y Salmonidae Salmonidae Any salmonid* Y Cottidae Cottidae Any sculpin* Y Salmonidae Thymallus arcticus Arctic grayling* Y Cyrenidae Corbicula fluminea Asian clam* N Salmonidae Salmo salar Atlantic Salmon Y Lymnaeidae Radix auricularia Big-eared radix* N Cyprinidae Mylopharyngodon piceus Black carp N Ictaluridae Ameiurus melas Black Bullhead* N Catostomidae Cycleptus elongatus Blue Sucker* N Cichlidae Oreochromis aureus Blue tilapia* N Catostomidae Catostomus discobolus Bluehead sucker* N Catostomidae Catostomus virescens Bluehead sucker* Y Felidae Lynx rufus Bobcat* Y Hylidae Pseudocris maculata Boreal chorus frog N Hydrocharitaceae Egeria densa Brazilian elodea N Salmonidae Salvelinus fontinalis Brook trout* Y Colubridae Boiga irregularis Brown tree snake* -

Tennessee Fish Species
The Angler’s Guide To TennesseeIncluding Aquatic Nuisance SpeciesFish Published by the Tennessee Wildlife Resources Agency Cover photograph Paul Shaw Graphics Designer Raleigh Holtam Thanks to the TWRA Fisheries Staff for their review and contributions to this publication. Special thanks to those that provided pictures for use in this publication. Partial funding of this publication was provided by a grant from the United States Fish & Wildlife Service through the Aquatic Nuisance Species Task Force. Tennessee Wildlife Resources Agency Authorization No. 328898, 58,500 copies, January, 2012. This public document was promulgated at a cost of $.42 per copy. Equal opportunity to participate in and benefit from programs of the Tennessee Wildlife Resources Agency is available to all persons without regard to their race, color, national origin, sex, age, dis- ability, or military service. TWRA is also an equal opportunity/equal access employer. Questions should be directed to TWRA, Human Resources Office, P.O. Box 40747, Nashville, TN 37204, (615) 781-6594 (TDD 781-6691), or to the U.S. Fish and Wildlife Service, Office for Human Resources, 4401 N. Fairfax Dr., Arlington, VA 22203. Contents Introduction ...............................................................................1 About Fish ..................................................................................2 Black Bass ...................................................................................3 Crappie ........................................................................................7 -

Aging Techniques & Population Dynamics of Blue Suckers (Cycleptus Elongatus) in the Lower Wabash River
Eastern Illinois University The Keep Masters Theses Student Theses & Publications Summer 2020 Aging Techniques & Population Dynamics of Blue Suckers (Cycleptus elongatus) in the Lower Wabash River Dakota S. Radford Eastern Illinois University Follow this and additional works at: https://thekeep.eiu.edu/theses Part of the Aquaculture and Fisheries Commons Recommended Citation Radford, Dakota S., "Aging Techniques & Population Dynamics of Blue Suckers (Cycleptus elongatus) in the Lower Wabash River" (2020). Masters Theses. 4806. https://thekeep.eiu.edu/theses/4806 This Dissertation/Thesis is brought to you for free and open access by the Student Theses & Publications at The Keep. It has been accepted for inclusion in Masters Theses by an authorized administrator of The Keep. For more information, please contact [email protected]. AGING TECHNIQUES & POPULATION DYNAMICS OF BLUE SUCKERS (CYCLEPTUS ELONGATUS) IN THE LOWER WABASH RIVER By Dakota S. Radford B.S. Environmental Biology Eastern Illinois University A thesis prepared for the requirements for the degree of Master of Science Department of Biological Sciences Eastern Illinois University May 2020 TABLE OF CONTENTS Thesis abstract .................................................................................................................... iii Acknowledgements ............................................................................................................ iv List of Tables .......................................................................................................................v -

Species Status Assessment (SSA) Report for the Ozark Chub (Erimystax Harryi) Version 1.2
Species Status Assessment (SSA) Report for the Ozark Chub (Erimystax harryi) Version 1.2 Ozark chub (Photo credit: Dustin Lynch, Arkansas Natural Heritage Commission) August 2019 U.S. Fish and Wildlife Service - Arkansas Ecological Services Field Office This document was prepared by Alyssa Bangs (U. S. Fish and Wildlife Service (USFWS) – Arkansas Ecological Services Field Office), Bryan Simmons (USFWS—Missouri Ecological Services Field Office), and Brian Evans (USFWS –Southeast Regional Office). We greatly appreciate the assistance of Jeff Quinn (Arkansas Game and Fish Commission), Brian Wagner (Arkansas Game and Fish Commission), and Jacob Westhoff (Missouri Department of Conservation) who provided helpful information and review of the draft document. We also thank the peer reviewers, who provided helpful comments. Suggested reference: U.S. Fish and Wildlife Service. 2019. Species status assessment report for the Ozark chub (Erimystax harryi). Version 1.2. August 2019. Atlanta, GA. CONTENTS Chapter 1: Executive Summary 1 1.1 Background 1 1.2 Analytical Framework 1 CHAPTER 2 – Species Information 4 2.1 Taxonomy and Genetics 4 2.2 Species Description 5 2.3 Range 6 Historical Range and Distribution 6 Current Range and Distribution 8 2.4 Life History Habitat 9 Growth and Longevity 9 Reproduction 9 Feeding 10 CHAPTER 3 –Factors Influencing Viability and Current Condition Analysis 12 3.1 Factors Influencing Viability 12 Sedimentation 12 Water Temperature and Flow 14 Impoundments 15 Water Chemistry 16 Habitat Fragmentation 17 3.2 Model 17 Analytical -

Endangered Species
FEATURE: ENDANGERED SPECIES Conservation Status of Imperiled North American Freshwater and Diadromous Fishes ABSTRACT: This is the third compilation of imperiled (i.e., endangered, threatened, vulnerable) plus extinct freshwater and diadromous fishes of North America prepared by the American Fisheries Society’s Endangered Species Committee. Since the last revision in 1989, imperilment of inland fishes has increased substantially. This list includes 700 extant taxa representing 133 genera and 36 families, a 92% increase over the 364 listed in 1989. The increase reflects the addition of distinct populations, previously non-imperiled fishes, and recently described or discovered taxa. Approximately 39% of described fish species of the continent are imperiled. There are 230 vulnerable, 190 threatened, and 280 endangered extant taxa, and 61 taxa presumed extinct or extirpated from nature. Of those that were imperiled in 1989, most (89%) are the same or worse in conservation status; only 6% have improved in status, and 5% were delisted for various reasons. Habitat degradation and nonindigenous species are the main threats to at-risk fishes, many of which are restricted to small ranges. Documenting the diversity and status of rare fishes is a critical step in identifying and implementing appropriate actions necessary for their protection and management. Howard L. Jelks, Frank McCormick, Stephen J. Walsh, Joseph S. Nelson, Noel M. Burkhead, Steven P. Platania, Salvador Contreras-Balderas, Brady A. Porter, Edmundo Díaz-Pardo, Claude B. Renaud, Dean A. Hendrickson, Juan Jacobo Schmitter-Soto, John Lyons, Eric B. Taylor, and Nicholas E. Mandrak, Melvin L. Warren, Jr. Jelks, Walsh, and Burkhead are research McCormick is a biologist with the biologists with the U.S. -

Fish Survey for Calhoun, Gordon County, Georgia
Blacktail Redhorse (Moxostoma poecilurum) from Oothkalooga Creek Fish Survey for Calhoun, Gordon County, Georgia Prepared by: DECATUR, GA 30030 www.foxenvironmental.net January 2018 Abstract Biological assessments, in conjunction with habitat surveys, provide a time-integrated evaluation of water quality conditions. Biological and habitat assessments for fish were conducted on 3 stream segments in and around Calhoun, Gordon County, Georgia on October 3 and 5, 2017. Fish, physical habitat, and water chemistry data were evaluated according to Georgia Department of Natural Resources (GADNR), Wildlife Resources Division (WRD) – Fisheries Section protocol entitled “Standard Operating Procedures for Conducting Biomonitoring on Fish Communities in Wadeable Streams in Georgia”. All of the water quality parameters at all sites were within the typical ranges for streams although conductivity was somewhat high across the sites. Fish habitat scores ranged from 80 (Tributary to Oothkalooga Creek) to 132.7 (Oothkalooga Creek). Native fish species richness ranged from 6 species (Tributary to Oothkalooga Creek) to 17 (Oothkalooga and Lynn Creeks). Index of biotic integrity (IBI) scores ranged from 16 (Tributary to Oothkalooga Creek; “Very Poor”) to 34 (Lynn Creek; “Fair”). Overall, the results demonstrate that Oothkalooga and Lynn Creeks are in fair condition whereas the Tributary to Oothkalooga Creek is highly impaired. Although the data are only a snapshot of stream conditions during the sampling events, they provide a biological characterization from which to evaluate the effect of future changes in water quality and watershed management in Calhoun. We recommend continued monitoring of stream sites throughout the area to ensure that the future ecological health of Calhoun’s water resources is maintained. -

The Nature Conservancy's Merwin Preserve
CORE Metadata, citation and similar papers at core.ac.uk Provided by Illinois Digital Environment for Access to Learning and Scholarship Repository University of Illinois Prairie Research Institute William Shilts, Executive Director Illinois Natural History Survey Brian D. Anderson, Director 1816 South Oak Street Champaign, IL 61820 217-333-6830 The Nature Conservancy’s Merwin Preserve Fish and Aquatic Vegetation Monitoring Annual Report T.D. VanMiddlesworth and Andrew F. Casper INHS Technical Report 2014 (12) Prepared for The Nature Conservancy Illinois Natural History Survey Issue Date: 01/15/2014 Floodplain restoration monitoring of the aquatic vegetation and fish communities of The Nature Conservancy’s Merwin Preserve 2013 Grant/Contract Number: The Nature Conservancy CO7-33 Unrestricted; release online 2 years from issue date 2 Introduction In 1999, The Nature Conservancy (TNC) initiated restoration at the 2,000 acre Merwin Preserve at Spunky Bottoms, which lies alongside the Illinois River in Brown County, Illinois. Before its disconnection from the river due to levee construction and conversion to farmland in 1921, the Merwin Preserve consisted of two floodplain lakes known as Elbow and Long lakes and were both rich in natural resources characteristic of floodplain lakes of the Illinois River (Blodgett et al. 2007, TNC Personal Communication 2014). During the initial restoration phase, the Merwin Preserve was allowed to naturally fill with water from precipitation and a native plant community was established through natural colonization and establishment processes (Blodgett et al. 2007). Thus, restoration efforts at the Merwin Preserve have been largely occurring without dramatic interventions such as plantings or habitat construction, a process much like old field succession.