Condensed and Dried Milk
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

CONDENSED and DESICCATED MILK, Milk Is a Bulky Product, Expensive to Transport, and Very Suscep
CONDENSED AND DESICCATED MILK, By LEVI WELLS, Dairy Inspector, Dairy Division, Bureau of Animal Industry. INTRODUCTION. Milk is a bulky product, expensive to transport, and very suscep- tible to contamination, which in a short time renders it unpalatable. In its natural state it contains al^out 87 per cent of water, which is a comparatively worthless constituent. ' Efforts to reduce the water content of milk, leaving the solids in ; a more concentrated form without destroying their food value, and at the same time improving the keeping qualities, have resulted in developing the manufacture of both condensed milk and desiccated milk or milk flour. The condensing processes now used reduce the volume of milk to one-half or one-fifth its original bulk, and if the product is carefully sterilized or preserved with cane sugar and sealed in air-tight containers it becomes easily transportable and keeps for long periods in any climate. The desiccating processes now perfected remove practically all the ■water in milk, leaving a dry powder soluble in water. In the manu- facture of this product whole milk is reduced to about one-eighth, and skimmed milk to about one-eleventh the original volume. By this means the volume is reduced to a minimum, and the keeping quality, particularly of dried skim milk, is superior. CONDENSED JVIILK. Eemoving a portion of the water from milk, leaving a product of good keeping quality which may be restored to its normal consistency without injuring its natural flavor, is a problem that has been studied for many years. It is claimed that during the first half of the last century foreign inventors evaporated a part of the water from milk, and, with the addition of cane sugar, made what was then known a& condensed milk (see Scientific American, export edition, July, 1905). -

Chapter 1530 Department of Agriculture Milk, Milk Products, and Standards
MINNESOTA RULES 2001 250 CHAPTER 1530 DEPARTMENT OF AGRICULTURE MILK, MILK PRODUCTS, AND STANDARDS MILK AND CREAM AND RELATED PRODUCTS 1530.0550 PASTEURIZATION OF CREAMING MIXTURE. 1530.0010 DEFINITIONS. 1530.0560 DIACETYL STARTER DISTILLATE OR 1530.0020 MILK. OTHER FLAVORING SUBSTANCES. 1530.0030 GOAT MILK. 1530.0570 DEFINITIONS. 1530.0040 EWE MILK. 1530.0580 LABEL STATEMENTS. 1530.0050 CREAM. 1530.0590 LABEL STATEMENT REGARDING 1530.0060 HOMOGENIZED CREAM. ARTIFICIAL FLAVORING. 1530.0070 SOUR CREAM, CULTURED SOUR CREAM. 1530.0600 LABEL DECLARATIONS TO BE USED IN 1530.0080 LIGHT CREAM, COFFEE CREAM. OR TABLE CONJUNCTION WITH "CREAMED COTTAGE CREAM. CHEESE." 1530.0090 WHIPPED LIGHT CREAM, COFFEE CREAM. 1530.0610 VITAMIN D MILK AND MILK PRODUCTS. OR TABLE CREAM. 1530.0620 FORTIFIED MILK PRODUCTS. 1530.0100 WHIPPING CREAM. 1530.0630 RECONSTITUTED OR RECOMBINED MILK 1530.0110 LIGHT WHIPPING CREAM. AND MILK PRODUCTS. 1530.0120 HEAVY CREAM OR HEAVY WHIPPING 1530.0640 STERILIZED MILK. CREAM. 1530.0650 HOMOGENIZED MILK AND HOMOGENIZED 1530.0130 WHIPPED CREAM. 1530.0140 HALF AND HALF. MILK PRODUCTS. 1530.0150 SOUR HALF AiND HALF OR CULTURED 1530.0660 WHEY. 1530.0670 CONCENTRATED MILK TO BE HALF AND HALF. 1530.0160 DRY MILK, DRY MILK SOLIDS. RECONSTITUTED FOR FLUID USE. 1530.0680 CONCENTRATED MILK PRODUCTS. 1530.0170 NONFAT DRY MILK. 1530.0690 ACIDIFIED MILK AND MILK PRODUCTS. 1530.0180 INSTANT NONFAT DRY MILK. 1530.0700 MILK PRODUCTS. 1530.0190 CONDENSED SKIM MILK, EVAPORATED 1530.0710 OPTIONAL INGREDIENTS. SKJM MILK, CONCENTRATED SKIM MILK. 1530.0200 SWEETENED CONDENSED SKIM MILK. GRADE A REQUIREMENTS FOR MILK, MILK 1530.0210 DRY BUTTERMILK. PRODUCrS, AND GOAT MILK 1530.0220 DRY WHEY. -

Status of Use of Nonfat Dry Milk and Plain Condensed Milk in the Production of Market Milk Products
77 STATUS OF USE OF NONFAT DRY MILK AND PLAIN CONDENSED MILK IN THE PRODUCTION OF MARKET MILK PRODUCTS K. G. WECKEL Department of Dairy and Food Industries Univer8ity of Wisconsin Nonfat dry milk and condensed skim milk have A milk plants in the State of Wisconsin. A question long been used. as a source of solids for ice cream and naire was submitted to the regulatory office 1 in each other frozen desserts. Within the past several years of 25 cities in the state during the months of May there has been rapid increase in the use of added non and June. The health officer was requested to ascer fat milk solids for various dairy products processed tain the actual use of nonfat dry milk and condensed in fluid milk plants. The increased use is of signifi skim milk in the dairy products produced· in each of cance in the light of the widespread adoption of the dairy plants in the jurisdiction on any given day. Grade A standards for fluid milk and cream, and, 'I'he results of the survey are believed to represent regionally, for cottage cheese, and in the absence of at least 80-90 per cent of the total utilization within similar standards for condensed and dried milk. the state. The results are presented in Table No. 1. The Wisconsin Conference on Intra-State Milk Nonfat dry milk was used in 18, or 72 per cent of the Shipments, at its September, 1957 meeting, adopted cities. Condensed skim milk was used in 14, or 56 per as interim standards the proposed draft Supplement cent of the cities on any given day. -

354 Subpart B—Food Additives in Standardized Foods PART 131—MILK and CREAM Subpart A—General Provisions
§ 130.20 21 CFR Ch. I (4–1–11 Edition) Subpart B—Food Additives in 131.130 Evaporated milk. 131.147 Dry whole milk. Standardized Foods 131.149 Dry cream. 131.150 Heavy cream. § 130.20 Food additives proposed for 131.155 Light cream. use in foods for which definitions 131.157 Light whipping cream. and standards of identity are estab- 131.160 Sour cream. lished. 131.162 Acidified sour cream. (a) Where a petition is received for 131.170 Eggnog. the issuance or amendment of a regula- 131.180 Half-and-half. tion establishing a definition and 131.200 Yogurt. standard of identity for a food under 131.203 Lowfat yogurt. section 401 of the act, which proposes 131.206 Nonfat yogurt. the inclusion of a food additive in such AUTHORITY: 21 U.S.C. 321, 341, 343, 348, 371, definition and standard of identity, the 379e. provisions of the regulations in part 171 SOURCE: 42 FR 14360, Mar. 15, 1977, unless of this chapter shall apply with respect otherwise noted. to the information that must be sub- EDITORIAL NOTE: Nomenclature changes to mitted with respect to the food addi- part 131 appear at 63 FR 14035, Mar. 24, 1998. tive. Since section 409(b)(5) of the act requires that the Commissioner publish notice of a petition for the establish- Subpart A—General Provisions ment of a food additive regulation § 131.3 Definitions. within 30 days after filing, notice of a petition relating to a definition and (a) Cream means the liquid milk prod- standard of identity shall also be pub- uct high in fat separated from milk, lished within that time limitation if it which may have been adjusted by add- includes a request, so designated, for ing thereto: Milk, concentrated milk, the establishment of a regulation per- dry whole milk, skim milk, con- taining to a food additive. -

The DUEL USE of the VCRHTOB for Conde.Nsing Milk and Pasteurizing Ice Cream Mix
The DUEL USE of the VCRHTOB for Conde.nsing Milk and Pasteurizing Ice Cream Mix By G. H. WILSTER Oregon State System of Higher Education . Agricultural Experiment Station Oregon State College Corvallis Station Bulletin 430 july 1945 FOREWORD The method of pasteurizing cream under vacuum was first studied by the Oregon Agricultural Experimtht Sta- tion in 1938, the results being presented in Bulletin 368. The experimental use of this method of pasteurization for ice cream was studied by the Station in 1939.The operation was so successful that it became routine practice in the experiment station laboratory and its pilot plant. In 1942, stimulated by the deficiency of nonfat dry milk solids, the dairy research staff saw the possibility of adapting the Vacreator* vacuum pasteurizer for condens- ing milk.After repeated trials, the necessary adjustments were determined.A systematic study was made.The method developed is fully outlined herein. It is now apparent that in addition to manufacturing with this equipment ice cream of delectable flavor, excel- lenttexture, and irreproachable sanitaryquality,the whole field of milk concentration is opened to new pos- sibilities.Owners of this type of equipment who have access to an adequate supply of milk may economically evaporate it if such a procedure is desirable. The results from this research suggest not only revo- lutionary developments in the ice cream industry but the possibility of new methods of preparing milk solids for other uses in the dairy manufacturing industry. WM. A. SCHOENFELD Dean and Director Vacreator" is the registered trademark designating the Murray vacuum I pasteurizers. TABLE OF CONTENTS Page Summary 5 Introduction 7 Using the \Tacreator for Pasteurizing (vacreating) Ice Cream Mix . -

JBT's Filling and Closing Technology for Liquid Dairy Products and Nutraceuticals in Metal Cans
JBT’s Filling and Closing technology for liquid dairy products and nutraceuticals in metal cans High-quality liquid dairy products Deserve the best filling & closing Solutions Take a deeper look inside the JBT equipment and line solutions for canned liquid dairy. Get to know what makes the JBT offering unique, and how it satisfies quality conscious consumers, securing product safety while keeping your production cost per unit low. WHITE PAPER /JBT/ WHITE PAPER /JBT/ The increasing demand for Sweetened Condensed Milk and Evaporated Milk in these regions is mainly driven Longer shelf life for canned by the increasing buying power; the product also has a reputation as a highly nutritious product that can be kept liquid dairy products and nutraceuticals fresh for a few days after opening, without refrigeration. In addition, canned dairy products have a long shelf-life and are well adapted to the harsh logistical conditions in these There are many strong commercial advantages for the tropical and sub-tropical countries when fresh milk is not food processor, retailer and consumer if a dairy product available. It is also used as a substitute for breast milk, does not require refrigeration and can be shipped and for cooking and as coffee cream, or for recombination in stored unrefrigerated on the shelves without spoiling. various processed foods such as breads and confectionery. Manufacturers can reach geographically wider markets and reduce return of unsold products. Shelf handling becomes Both sweetened condensed milk and evaporated milk are also quite easier and less time consuming for the retailer, forms of concentrated milk in which approximately 60 and internal logistics are simplified. -

Course Title: Microbiology of Milk Product Course No
COURSE TITLE: MICROBIOLOGY OF MILK PRODUCT COURSE NO. - DTM-222: CREDIT HRS-2 (1+1) MICROBIOLOGY OF CONCENTRATED MILK (SCM) RAKESH KUMAR ASSOCIATE PROFESSOR (DAIRY MICROBIOLOGY) FACULTY OF DAIRY TECHNOLOGY S.G.I.D.T., BVC CAMPUS, P.O.- BVC, DIST.-PATNA-800014 MICROBIOLOGY OF CONCENTRATED MILK Concentrated milk is produced by partial removal of water from fresh milk for direct sale to the consumer, which can be used as food ingredients for providing a source of milk solids in manufacturing of a variety of other products. The removal of water from milk gives an advantages in reducing storage and transport costs, ease / convenience in use and increasing shelf life. Concentrated milk is further reconstituted by the consumer with water, to give a similar composition of fresh milk. It is generally produced as a 3:1 concentrate containing approximately 10 - 12% milk fat and 36% total milk solids. Introduction Condensed milk is an important source of milk solids in confectionery, bakery products, ice cream, concentrated yoghurt, choclates, bakely products and other newer kind of food mix preparation. It may be made from whole, skimmed, or reduced fat milks depending on the consumer need and requirements. Uses of Condensed milk 1- Condensed milks are mainly used for reconstitution into sweet milk drinks. 2- Condensed milks are very commonly utilised in the preparation of tea or coffee. 3- They are also used in the ice-cream preparation. 4- They are used in candy and confectionary products. 5- They are frequently used in prepared foods in various ways and form. History of condensed milk Invented by Gail Borden in the early 1850s, canned condensed milk proved invaluable as a military ration in the 1860s. -

Low FODMAP Food Choices Foods to Eat Foods to Limit Poultry Fish, Eggs Dairy Meat
Low FODMAP Food Choices Foods to Eat Foods to Limit Poultry Fish, beef, chicken, canned tuna, foods made with high FODMAP Eggs eggs, egg whites, fish, lamb, fruit pork, shellfish, sauces or with HFCS turkey, cold cuts Dairy lactose free dairy, small buttermilk, chocolate, cottage amounts of: cheese, ice cream, cream cheese, half and half, creamy/cheesy sauces, milk hard cheeses (cheddar, colby, (from cow, sheep or goat), parmesan, swiss), mozzarella, sweetened condensed milk, sherbet evaporated milk, soft cheeses (brie, ricotta), sour cream, whipped cream, yogurt Meat, Non- almond milk, rice milk, rice milk coconut milk, coconut cream, Dairy ice cream, nuts, nut butters, beans, black eyed peas, seeds hummus, lentils, pistachios, soy Alternatives products Grains wheat free grains/wheat free chicory root, inulin, grains with flours (gluten free grains are HFCS or made from wheat wheat free): (terms for wheat: einkorn, bagels, breads, hot/cold cereals emmer, kamut, spelt), wheat (corn flakes, cheerios, cream of flours (terms for wheat rice, grits, oats, etc), crackers, flour: bromated, durum, noodles, pastas, quinoa, enriched, farina, graham, pancakes, pretzels, rice, semolina, white tapioca, tortillas, waffles flours), flour tortillas, rye Fruits bananas, berries, cantaloupe, avocado, apples, applesauce, grapes, grapefruit, honeydew, apricots, dates, canned fruit, kiwi, kumquat, lemon, lime, cherries, dried fruits, figs, mandarin, orange, passion guava, lychee, mango, fruit, pineapple, rhubarb, nectarines, pears, papaya, tangerine peaches, -

No Churn Ice Cream by Cleo Coyle
Cleo’s No Churn Ice Cream Chocolate, Vanilla, Coffee "No churn" ice cream appeals to me. It's easy to make, economical, takes little room in my freezer, and does not require reading appliance instructions (huzzah). Unfortunately, there is a problem with the most common recipe for "no churn" ice cream, one I have worked to remedy. Why I Revised the Popular "No Churn" Ice Cream Recipe "No churn" ice cream replaces the churning of air into the cream with whipping the air into the cream before freezing. This does a good job of keeping the end product as soft and fluffy as churned ice cream. But there is a problem with the most common "no churn" recipe I've seen shared across the internet (i.e., cream + sweetened condensed milk). It produces an ice cream that's far too soft, melts too easily, and leaves a waxy aftertaste on the tongue from too much butterfat. In short, it produces a product like ice cream but not as good. So I began experimenting with that ubiquitous no churn recipe and have come up with an improved version (IMO, of course). Why is it better? A few reasons... (1) Adding evaporated milk to the mix before freezing creates a final product that has a much cleaner, more ice-cream-like texture, eliminating that waxy butterfat coating on the tongue. It also... (2) Allows very fine ice crystals to form, which make the final product colder in the mouth and gives it a more stable form in the dish or on your cone. -
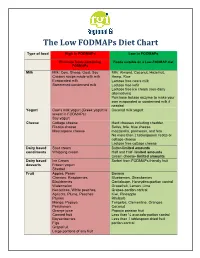
The Low Fodmaps Diet Chart
The Low FODMAPs Diet Chart Type of food High in FODMAPs Low in FODMAPs Eliminate foods containing Foods suitable on a Low-FODMAP diet FODMAPs Milk Milk: Cow, Sheep, Goat, Soy Milk: Almond, Coconut, Hazelnut, Creamy soups made with milk Hemp, Rice Evaporated milk Lactose free cow’s milk Sweetened condensed milk Lactose free kefir Lactose free ice cream (non-dairy alternatives) Purchase lactase enzyme to make your own evaporated or condensed milk if needed Yogurt Cow’s milk yogurt (Greek yogurt is Coconut milk yogurt lowest in FODMAPs) Soy yogurt Cheese Cottage cheese Hard cheeses including cheddar, Ricotta cheese Swiss, brie, blue cheese, Mascarpone cheese mozzarella, parmesan, and feta No more than 2 tablespoons ricotta or cottage cheese Lactose free cottage cheese Dairy based Sour cream Butter-limited amounts condiments Whipping cream Half and Half- limited amounts Cream cheese- limited amounts Dairy based Ice Cream Sorbet from FODMAPs friendly fruit desserts Frozen yogurt Sherbet Fruit Apples, Pears Banana Cherries, Raspberries, Blueberries, Strawberries Blackberries Cantaloupe, Honeydew-portion control Watermelon Grapefruit, Lemon, Lime Nectarines, White peaches, Grapes-portion control Apricots, Plums, Peaches Kiwi, Pineapple Prunes Rhubarb Mango, Papaya Tangelos, Clementine, Oranges Persimmon Coconut Orange juice Papaya passion fruit Canned fruit Less than ¼ avocado-portion control Boysenberries Less than 1 tablespoon dried fruit Figs portion control Grapefruit Large portions of any fruit Limit consumption to one low FODMAPs fruit -

Coquito – Coconut Eggnog - Recipes | Goya Foods
11/11/2019 Coquito – Coconut Eggnog - Recipes | Goya Foods Coquito – Coconut Eggnog 7 Prep time 5m Total time 5m Yields Cups An Authentic Puerto Rican Beverage If you like coconut and eggnog, you’ll love Puerto Rican Coquito! Coquito is a thick and creamy coconut drink that mixes silky GOYA® Coconut Milk with sweet GOYA® Cream of Coconut, cinnamon and rum. This rich, authentic coquito recipe is commonly made during Christmas where it is served cold and meant to be shared with family and friends. Ingredients 2 GOYA® Evaporated Milk cans (12 oz. each) 1 GOYA® Cream of Coconut can (15 oz.) 1 GOYA® Coconut Milk can (13.5 oz.) ½ GOYA® Sweetened Condensed Milk cup ½ white rum (optional) cup 1 vanilla extract tsp. ½ ground cinnamon, plus more for garnish, if desired tsp. https://www.goya.com/en/recipes/coquito-coconut-eggnog 1/2 11/11/2019 Coquito – Coconut Eggnog - Recipes | Goya Foods Cinnamon sticks (optional) Directions 1 In bowl of blender, add evaporated milk, cream of coconut, coconut milk, sweetened condensed milk, rum (if using), vanilla extract and ground cinnamon. Blend on high until mixture is well combined, 1-2 minutes. 2 Pour coconut mixture into glass bottles; cover. Transfer to refrigerator. Chill until cold. 3 To serve, stir or shake bottle well to combine. Pour coquito into small serving glasses. Garnish with ground cinnamon and cinnamon sticks, if desired. Quality Goya products are available at many food stores and supermarkets. Copyright © 2019 Goya Foods, Inc., All Rights Reserved https://www.goya.com/en/recipes/coquito-coconut-eggnog 2/2. -

Total Fat Determination in Condensed Milk According to the Randall Method
APPLICATION NOTE F&F-S-005-2016/A1 Total Fat Determination in Condensed Milk according to the Randall method Reference: AOAC 933.05 Fat in Cheese Tested with VELP Scientifica SER 158/6 Solvent AutoExtractor (Code S303A0380) and HU6 Hydrolysis Unit (Code F30300110) Copyright © 2016 VELP Scientifica. All rights reserved. No part of this publication may be reproduced or transmitted in any form or for any purpose without the express permission of VELP. VELP Scientifica, Italy Tel: +39 039 628 811 Fax: +39 039 628 8120 www.velp.com TOTAL FAT DETERMINATION IN CONDENSED MILK RANDALL METHOD Introduction Condensed milk is cow's milk from which water has been removed. It is most often found in the form of sweetened condensed milk (SCM), with sugar added. Sweetened condensed milk is a very thick, sweet product which when canned can last for years without refrigeration if unopened. The production of condensed milk is obtained starting from raw milk that is clarified and standardized and is then heated to 85–90 °C for several seconds. This heating process destroys some microorganisms, decreases fat separation and inhibits oxidation. Some water is evaporated from the milk and sugar is added until a 9:11 (nearly half) ratio of sugar to (evaporated) milk is reached. The sugar extends the shelf life of sweetened condensed milk. Nowadays condensed milk is used in numerous dessert dishes in many countries all over the world. Fat Determination in Condensed milk Hot solvent extraction process with SER 158 Series can be summed up in 5 steps, for a fully automatic process completely unattended: STEP 1 STEP 2 STEP 3 STEP 4 STEP 5 IMMERSION REMOVING WASHING RECOVERY COOLING During IMMERSION the sample is immersed in boiling solvent.