Functional Signature Ontology-Based Identification and Validation of Novel Therapeutic Targets and Natural Products for the Treatment of Cancer
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hearing Aging Is 14.1±0.4% GWAS-Heritable
medRxiv preprint doi: https://doi.org/10.1101/2021.07.05.21260048; this version posted July 7, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. It is made available under a CC-BY-NC 4.0 International license . Predicting age from hearing test results with machine learning reveals the genetic and environmental factors underlying accelerated auditory aging Alan Le Goallec1,2+, Samuel Diai1+, Théo Vincent1, Chirag J. Patel1* 1Department of Biomedical Informatics, Harvard Medical School, Boston, MA, 02115, USA 2Department of Systems, Synthetic and Quantitative Biology, Harvard University, Cambridge, MA, 02118, USA +Co-first authors *Corresponding author Contact information: Chirag J Patel [email protected] Abstract With the aging of the world population, age-related hearing loss (presbycusis) and other hearing disorders such as tinnitus become more prevalent, leading to reduced quality of life and social isolation. Unveiling the genetic and environmental factors leading to age-related auditory disorders could suggest lifestyle and therapeutic interventions to slow auditory aging. In the following, we built the first machine learning-based hearing age predictor by training models to predict chronological age from hearing test results (root mean squared error=7.10±0.07 years; R-Squared=31.4±0.8%). We defined hearing age as the prediction outputted by the model on unseen samples, and accelerated auditory aging as the difference between a participant’s hearing age and age. We then performed a genome wide association study [GWAS] and found that accelerated hearing aging is 14.1±0.4% GWAS-heritable. -
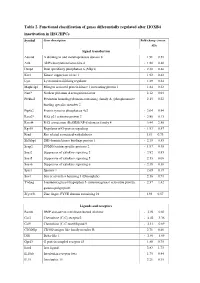
Table 2. Functional Classification of Genes Differentially Regulated After HOXB4 Inactivation in HSC/Hpcs
Table 2. Functional classification of genes differentially regulated after HOXB4 inactivation in HSC/HPCs Symbol Gene description Fold-change (mean ± SD) Signal transduction Adam8 A disintegrin and metalloprotease domain 8 1.91 ± 0.51 Arl4 ADP-ribosylation factor-like 4 - 1.80 ± 0.40 Dusp6 Dual specificity phosphatase 6 (Mkp3) - 2.30 ± 0.46 Ksr1 Kinase suppressor of ras 1 1.92 ± 0.42 Lyst Lysosomal trafficking regulator 1.89 ± 0.34 Mapk1ip1 Mitogen activated protein kinase 1 interacting protein 1 1.84 ± 0.22 Narf* Nuclear prelamin A recognition factor 2.12 ± 0.04 Plekha2 Pleckstrin homology domain-containing. family A. (phosphoinosite 2.15 ± 0.22 binding specific) member 2 Ptp4a2 Protein tyrosine phosphatase 4a2 - 2.04 ± 0.94 Rasa2* RAS p21 activator protein 2 - 2.80 ± 0.13 Rassf4 RAS association (RalGDS/AF-6) domain family 4 3.44 ± 2.56 Rgs18 Regulator of G-protein signaling - 1.93 ± 0.57 Rrad Ras-related associated with diabetes 1.81 ± 0.73 Sh3kbp1 SH3 domain kinase bindings protein 1 - 2.19 ± 0.53 Senp2 SUMO/sentrin specific protease 2 - 1.97 ± 0.49 Socs2 Suppressor of cytokine signaling 2 - 2.82 ± 0.85 Socs5 Suppressor of cytokine signaling 5 2.13 ± 0.08 Socs6 Suppressor of cytokine signaling 6 - 2.18 ± 0.38 Spry1 Sprouty 1 - 2.69 ± 0.19 Sos1 Son of sevenless homolog 1 (Drosophila) 2.16 ± 0.71 Ywhag 3-monooxygenase/tryptophan 5- monooxygenase activation protein. - 2.37 ± 1.42 gamma polypeptide Zfyve21 Zinc finger. FYVE domain containing 21 1.93 ± 0.57 Ligands and receptors Bambi BMP and activin membrane-bound inhibitor - 2.94 ± 0.62 -

Mathiomica: an Integrative Platform for Dynamic Omics George I
MathIOmica: An Integrative Platform for Dynamic Omics George I. Mias1,*, Tahir Yusufaly2, Raeuf Roushangar1, Lavida R. K. Brooks1, Vikas V. Singh1, and Christina Christou3 1Michigan State University, Biochemistry and Molecular Biology, East Lansing, MI 48824, USA 2University of Southern California, Department of Physics and Astronomy, Los Angeles, CA, 90089, USA 3Mercy Cancer Center, Department of Radiation Oncology, Mason City, IA 50401, USA *[email protected] SUPPLEMENTARY NOTE 1 1 1 MathIOmica: Omics Analysis Tutorial Loading the MathIOmica Package Metabolomic Data Data in MathIOmica Combined Data Clustering Transcriptome Data Visualization Proteomic Data Annotation and Enrichment MathIOmica is an omics analysis package designed to facilitate method development for the analysis of multiple omics in Mathematica, particularly for dynamics (time series/longitudinal data). This extensive tutorial follows the analysis of multiple dynamic omics data (transcriptomics, proteomics, and metabolomics from human samples). Various MathIOmica functions are introduced in the tutorial, including additional discussion of related functionality. We should note that the approach methods are simply an illustration of MathIOmica functionality, and should not be considered as a definitive appoach. Additionally, certain details are included to illustrate common complications (e.g. renaming samples, combining datasets, transforming accessions from one database to another, dealing with replicates and Missing data, etc.). After a brief discussion of data in MathIOmica, each example data (transcriptome, proteome and metabolome) are imported and preprocessed. Next a simulation is carried out to obtain datasets for each omics used to assess statistical significance cutoffs. The datasets are combined, and classified for time series patterns, followed by clustering. The clusters are visualized, and biological annotation of Gene Ontology (GO) and pathway analysis (KEGG: Kyoto Encyclopedia of Genes and Genomes) are finally considered. -

Allosteric Activation of Functionally Asymmetric RAF Kinase Dimers
Allosteric Activation of Functionally Asymmetric RAF Kinase Dimers Jiancheng Hu,1,2 Edward C. Stites,1 Haiyang Yu,1 Elizabeth A. Germino,1 Hiruy S. Meharena,3,4 Philip J.S. Stork,6 Alexandr P. Kornev,3,4,5 Susan S. Taylor,3,4,5,* and Andrey S. Shaw1,2,* 1Department of Pathology and Immunology 2Howard Hughes Medical Institute Washington University School of Medicine, 660 South Euclid, Box 8118, St. Louis, MO 63110, USA 3Department of Chemistry/Biochemistry 4Department of Pharmacology 5Howard Hughes Medical Institute University of California San Diego, 9500 Gilman Drive, Mail code 0654, La Jolla, CA 92093, USA 6Vollum Institute, Oregon Health & Science University, 3181 SW Sam Jackson Park Road, Portland, OR 97239, USA *Correspondence: [email protected] (S.S.T.), [email protected] (A.S.S.) http://dx.doi.org/10.1016/j.cell.2013.07.046 SUMMARY There are three isoforms of RAF: A-RAF, B-RAF, and C-RAF. Each appears to have a distinct mechanism of activation (Baljuls Although RAF kinases are critical for controlling et al., 2007; Galabova-Kovacs et al., 2006; Wimmer and Baccar- cell growth, their mechanism of activation is incom- ini, 2010). BRAF is considered to be more active and have a pletely understood. Recently, dimerization was simpler mechanism of activation in comparison to CRAF and shown to be important for activation. Here we show ARAF (Chong et al., 2001; Rebocho and Marais, 2013; Zhang that the dimer is functionally asymmetric with one and Guan, 2000). This difference can potentially explain why kinase functioning as an activator to stimulate activ- BRAF mutations are so common in cancer, in contrast to CRAF and ARAF, where mutations are rarely found (Davies ity of the partner, receiver kinase. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Transcriptomic Analysis of Native Versus Cultured Human and Mouse Dorsal Root Ganglia Focused on Pharmacological Targets Short
bioRxiv preprint doi: https://doi.org/10.1101/766865; this version posted September 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. Transcriptomic analysis of native versus cultured human and mouse dorsal root ganglia focused on pharmacological targets Short title: Comparative transcriptomics of acutely dissected versus cultured DRGs Andi Wangzhou1, Lisa A. McIlvried2, Candler Paige1, Paulino Barragan-Iglesias1, Carolyn A. Guzman1, Gregory Dussor1, Pradipta R. Ray1,#, Robert W. Gereau IV2, # and Theodore J. Price1, # 1The University of Texas at Dallas, School of Behavioral and Brain Sciences and Center for Advanced Pain Studies, 800 W Campbell Rd. Richardson, TX, 75080, USA 2Washington University Pain Center and Department of Anesthesiology, Washington University School of Medicine # corresponding authors [email protected], [email protected] and [email protected] Funding: NIH grants T32DA007261 (LM); NS065926 and NS102161 (TJP); NS106953 and NS042595 (RWG). The authors declare no conflicts of interest Author Contributions Conceived of the Project: PRR, RWG IV and TJP Performed Experiments: AW, LAM, CP, PB-I Supervised Experiments: GD, RWG IV, TJP Analyzed Data: AW, LAM, CP, CAG, PRR Supervised Bioinformatics Analysis: PRR Drew Figures: AW, PRR Wrote and Edited Manuscript: AW, LAM, CP, GD, PRR, RWG IV, TJP All authors approved the final version of the manuscript. 1 bioRxiv preprint doi: https://doi.org/10.1101/766865; this version posted September 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. -

Functional Parsing of Driver Mutations in the Colorectal Cancer Genome Reveals Numerous Suppressors of Anchorage-Independent
Supplementary information Functional parsing of driver mutations in the colorectal cancer genome reveals numerous suppressors of anchorage-independent growth Ugur Eskiocak1, Sang Bum Kim1, Peter Ly1, Andres I. Roig1, Sebastian Biglione1, Kakajan Komurov2, Crystal Cornelius1, Woodring E. Wright1, Michael A. White1, and Jerry W. Shay1. 1Department of Cell Biology, University of Texas Southwestern Medical Center, 5323 Harry Hines Boulevard, Dallas, TX 75390-9039. 2Department of Systems Biology, University of Texas M.D. Anderson Cancer Center, Houston, TX 77054. Supplementary Figure S1. K-rasV12 expressing cells are resistant to p53 induced apoptosis. Whole-cell extracts from immortalized K-rasV12 or p53 down regulated HCECs were immunoblotted with p53 and its down-stream effectors after 10 Gy gamma-radiation. ! Supplementary Figure S2. Quantitative validation of selected shRNAs for their ability to enhance soft-agar growth of immortalized shTP53 expressing HCECs. Each bar represents 8 data points (quadruplicates from two separate experiments). Arrows denote shRNAs that failed to enhance anchorage-independent growth in a statistically significant manner. Enhancement for all other shRNAs were significant (two tailed Studentʼs t-test, compared to none, mean ± s.e.m., P<0.05)." ! Supplementary Figure S3. Ability of shRNAs to knockdown expression was demonstrated by A, immunoblotting for K-ras or B-E, Quantitative RT-PCR for ERICH1, PTPRU, SLC22A15 and SLC44A4 48 hours after transfection into 293FT cells. Two out of 23 tested shRNAs did not provide any knockdown. " ! Supplementary Figure S4. shRNAs against A, PTEN and B, NF1 do not enhance soft agar growth in HCECs without oncogenic manipulations (Student!s t-test, compared to none, mean ± s.e.m., ns= non-significant). -

Epigenome-Wide Exploratory Study of Monozygotic Twins Suggests Differentially Methylated Regions to Associate with Hand Grip Strength
Biogerontology (2019) 20:627–647 https://doi.org/10.1007/s10522-019-09818-1 (0123456789().,-volV)( 0123456789().,-volV) RESEARCH ARTICLE Epigenome-wide exploratory study of monozygotic twins suggests differentially methylated regions to associate with hand grip strength Mette Soerensen . Weilong Li . Birgit Debrabant . Marianne Nygaard . Jonas Mengel-From . Morten Frost . Kaare Christensen . Lene Christiansen . Qihua Tan Received: 15 April 2019 / Accepted: 24 June 2019 / Published online: 28 June 2019 Ó The Author(s) 2019 Abstract Hand grip strength is a measure of mus- significant CpG sites or pathways were found, how- cular strength and is used to study age-related loss of ever two of the suggestive top CpG sites were mapped physical capacity. In order to explore the biological to the COL6A1 and CACNA1B genes, known to be mechanisms that influence hand grip strength varia- related to muscular dysfunction. By investigating tion, an epigenome-wide association study (EWAS) of genomic regions using the comb-p algorithm, several hand grip strength in 672 middle-aged and elderly differentially methylated regions in regulatory monozygotic twins (age 55–90 years) was performed, domains were identified as significantly associated to using both individual and twin pair level analyses, the hand grip strength, and pathway analyses of these latter controlling the influence of genetic variation. regions revealed significant pathways related to the Moreover, as measurements of hand grip strength immune system, autoimmune disorders, including performed over 8 years were available in the elderly diabetes type 1 and viral myocarditis, as well as twins (age 73–90 at intake), a longitudinal EWAS was negative regulation of cell differentiation. -

KSR1- and ERK-Dependent Translational Regulation of 2 the Epithelial-To-Mesenchymal Transition
bioRxiv preprint doi: https://doi.org/10.1101/2021.01.18.427224; this version posted January 21, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 KSR1- and ERK-dependent Translational Regulation of 2 the Epithelial-to-Mesenchymal Transition 3 Chaitra Rao1, Danielle E. Frodyma1, Siddesh Southekal2, Robert A. Svoboda3, Adrian R. Black1, 4 Chittibabu Guda2, Tomohiro Mizutani5, Hans Clevers5, Keith R. Johnson1,4, Kurt W. Fisher3, and 5 Robert E. Lewis1* 6 1 Eppley Institute, Fred & Pamela Buffett Cancer Center, University of Nebraska Medical Center, Omaha, NE 68198, USA. 7 2 Department of Genetics, Cell Biology and Anatomy, University of Nebraska Medical Center, Omaha, NE, University of 8 Nebraska Medical Center, Omaha, NE 68198, USA. 9 3 Department of Pathology and Microbiology, University of Nebraska Medical Center, Omaha, NE 68198, USA. 10 4 Department of Oral Biology, University of Nebraska Medical Center, Omaha, NE 68198, USA 11 5 Hubrecht Institute, Royal Netherlands Academy of Arts and Sciences (KNAW) and UMC Utrecht, 3584CT Utrecht, The 12 Netherlands 13 *Correspondence: [email protected] 14 Abstract: 15 The epithelial-to-mesenchymal transition (EMT) is considered a transcriptional process 16 that induces a switch in cells from a polarized state to a migratory phenotype. Here we show that 17 KSR1 and ERK promote EMT through the preferential translation of Epithelial-Stromal 18 Interaction 1 (EPSTI1), which is required to induce the switch from E- to N-cadherin and 19 coordinate migratory and invasive behavior. -

Bioinformatics Analyses of Genomic Imprinting
Bioinformatics Analyses of Genomic Imprinting Dissertation zur Erlangung des Grades des Doktors der Naturwissenschaften der Naturwissenschaftlich-Technischen Fakultät III Chemie, Pharmazie, Bio- und Werkstoffwissenschaften der Universität des Saarlandes von Barbara Hutter Saarbrücken 2009 Tag des Kolloquiums: 08.12.2009 Dekan: Prof. Dr.-Ing. Stefan Diebels Berichterstatter: Prof. Dr. Volkhard Helms Priv.-Doz. Dr. Martina Paulsen Vorsitz: Prof. Dr. Jörn Walter Akad. Mitarbeiter: Dr. Tihamér Geyer Table of contents Summary________________________________________________________________ I Zusammenfassung ________________________________________________________ I Acknowledgements _______________________________________________________II Abbreviations ___________________________________________________________ III Chapter 1 – Introduction __________________________________________________ 1 1.1 Important terms and concepts related to genomic imprinting __________________________ 2 1.2 CpG islands as regulatory elements ______________________________________________ 3 1.3 Differentially methylated regions and imprinting clusters_____________________________ 6 1.4 Reading the imprint __________________________________________________________ 8 1.5 Chromatin marks at imprinted regions___________________________________________ 10 1.6 Roles of repetitive elements ___________________________________________________ 12 1.7 Functional implications of imprinted genes _______________________________________ 14 1.8 Evolution and parental conflict ________________________________________________ -

1 Supporting Information for a Microrna Network Regulates
Supporting Information for A microRNA Network Regulates Expression and Biosynthesis of CFTR and CFTR-ΔF508 Shyam Ramachandrana,b, Philip H. Karpc, Peng Jiangc, Lynda S. Ostedgaardc, Amy E. Walza, John T. Fishere, Shaf Keshavjeeh, Kim A. Lennoxi, Ashley M. Jacobii, Scott D. Rosei, Mark A. Behlkei, Michael J. Welshb,c,d,g, Yi Xingb,c,f, Paul B. McCray Jr.a,b,c Author Affiliations: Department of Pediatricsa, Interdisciplinary Program in Geneticsb, Departments of Internal Medicinec, Molecular Physiology and Biophysicsd, Anatomy and Cell Biologye, Biomedical Engineeringf, Howard Hughes Medical Instituteg, Carver College of Medicine, University of Iowa, Iowa City, IA-52242 Division of Thoracic Surgeryh, Toronto General Hospital, University Health Network, University of Toronto, Toronto, Canada-M5G 2C4 Integrated DNA Technologiesi, Coralville, IA-52241 To whom correspondence should be addressed: Email: [email protected] (M.J.W.); yi- [email protected] (Y.X.); Email: [email protected] (P.B.M.) This PDF file includes: Materials and Methods References Fig. S1. miR-138 regulates SIN3A in a dose-dependent and site-specific manner. Fig. S2. miR-138 regulates endogenous SIN3A protein expression. Fig. S3. miR-138 regulates endogenous CFTR protein expression in Calu-3 cells. Fig. S4. miR-138 regulates endogenous CFTR protein expression in primary human airway epithelia. Fig. S5. miR-138 regulates CFTR expression in HeLa cells. Fig. S6. miR-138 regulates CFTR expression in HEK293T cells. Fig. S7. HeLa cells exhibit CFTR channel activity. Fig. S8. miR-138 improves CFTR processing. Fig. S9. miR-138 improves CFTR-ΔF508 processing. Fig. S10. SIN3A inhibition yields partial rescue of Cl- transport in CF epithelia. -

Characterizing G-Quadruplex Mediated Regulation of the Amyloid Precursor Protein Expression by Ezekiel Matthew David Crenshaw Oc
I Characterizing G-Quadruplex Mediated Regulation of the Amyloid Precursor Protein Expression By Ezekiel Matthew David Crenshaw October, 2016 A Dissertation Presented to the Faculty of Drexel University Department of Biology In partial fulfillment of the Requirements for the Degree of Ph.D. Daniel Marenda, Ph.D. (Chair) Elias Spiliotis, Ph.D. Associate Professor, Associate Professor, Director, Graduate Student Program Director, Cell Imaging Center Co-Director, Cell Imaging Center Dept. Biology Dept. Biology Drexel University Drexel University Tali Gidalevitz, Ph.D. Aleister Saunders, Ph.D. (Advisor) Assistant Professor, Dept. Biology Associate Professor, Drexel University Director of the RNAi Resource Center, Dept. Biology Jeff Twiss, Ph.D. Senior Vice Provost for Research, Drexel University Professor, SmartState Chair, Childhood Neurotherapeutics Michael Akins, Ph.D.(Co-Advisor) Dept. Biology University of South Carolina Assistant Professor, Dept. Biology Drexel University II Acknowledgements Proverbs 3:5-6, “Trust in the Lord with all thine heart and lean not unto thine own understanding. In all thine ways acknowledge Him and He shall direct thy paths” I want to start off my acknowledging my LORD, who is known by many names. He is El Elyon, The Most High God; He is YAHWEH Shammah, for He has been there for me; He is YAHWEH Yireh, for He has provided for all of my needs; He is YAHWEH Shalom, for He is my peace. He is Yeshua Ha Mashiach, for He is my Savior. Coming from humble beginnings, my faith in the LORD has been the source of strength and encouragement to overcome the many obstacles I faced growing up, as well as in my pursuit for my Ph.D.