3 Recent Actions by the Food And
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Photoaging & Skin Damage
Use_for_Revised_OFC_Only_2006_PhotoagingSkinDamage 5/21/13 9:11 AM Page 2 PEORIA (309) 674-7546 MORTON (309) 263-7546 GALESBURG (309) 344-5777 PERU (815) 224-7400 NORMAL (309) 268-9980 CLINTON, IA (563) 242-3571 DAVENPORT, IA (563) 344-7546 SoderstromSkinInstitute.comsoderstromskininstitute.com FROMFrom YOUR Your DERMATOLOGISTDermatologist [email protected]@skinnews.com PHOTOAGING & SKIN DAMAGE Before You Worship The Sun Who’s At Risk? Today, many researchers and dermatologists Skin types that burn easily and tan rarely are believe that wrinkling and aging changes of the skin much more susceptible to the ravages of the sun on the are much more related to sun damage than to age! skin than are those that tan easily, rather than burn. Many of the signs of skin damage from the sun are Light complected, blue-eyed, red-haired people such as pictured on these pages. The decrease in the ozone Swedish, Irish, and English, are usually more suscep- layer, increasing the sun’s intensity, and the increasing tible to photo damage, and their skin shows the signs sun exposure among our population – through work, of photo damage earlier in life and in a more pro- sports, sunbathing and tanning parlors – have taken a nounced manner. Dark complexions give more protec- tremendous toll on our skin. Sun damage to the skin tion from light and the sun. ranks with other serious health dangers of smoking, alcohol, and increased cholesterol, and is being seen in younger and younger people. NO TAN IS A SAFE TAN! Table of Contents Sun Damage .............................................Pg. 1 Skin Cancer..........................................Pgs. 2-3 Mohs Micrographic Surgery ......................Pg. -

Late Stent Thrombosis After Paclitaxel-Eluting Stent Placement in a Patient with Essential Thrombocytosis
558 Türk Kardiyol Dern Arş - Arch Turk Soc Cardiol 2010;38(8):558-560 Late stent thrombosis after paclitaxel-eluting stent placement in a patient with essential thrombocytosis Esansiyel trombositozlu bir hastada paklitaksel salınımlı stent yerleştirme sonrası gelişen geç stent trombozu Telat Keleş, M.D., Nihal Akar Bayram, M.D., Tahir Durmaz, M.D., Engin Bozkurt, M.D. Department of Cardiology, Ankara Atatürk Education and Research Hospital, Ankara We report on a case of late stent thrombosis after drug- Bu yazıda, esansiyel trombositozlu bir hastada ilaç sa- eluting stent placement in a patient with essential throm- lınımlı stent yerleştirme sonrası gelişen geç stent trom- bocytosis. A 51-year-old male patient with a three-month bozu sunuldu. Üç ay önce sol ön inen artere paklitak- history of paclitaxel-eluting stent placement to the left sel salınımlı stent yerleştirilen 51 yaşındaki erkek hasta anterior descending artery presented with a complaint şiddetli retrosternal göğüs ağrısı yakınmasıyla başvur- of severe retrosternal chest pain. A high platelet count du. İki ay öncesinde hastanın trombosit sayımı yüksek (1,063,000/mm3) was detected two months prior to (1063000/mm3) bulunmuş ve durumu esansiyel trombo- presentation, which was interpreted as essential throm- sitoz olarak yorumlanmıştı. Hasta standart ikili antitrom- bocytosis. He was on standard dual antiplatelet therapy bosit tedavi (aspirin ve klopidogrel) görmekteydi. Elekt- (aspirin and clopidogrel). The electrocardiogram showed rokardiyografide V1-V6 derivasyonlarında ST-segment ST-segment elevation in leads V1-V6. Emergent coro- yükselmesi izlendi. Acil koroner anjiyografide paklitaksel nary angiography revealed thrombotic total occlusion salınımlı stent yerinde trombotik tam tıkanıklık gözlendi. at the location of the paclitaxel-eluting stent. -

May 2020 for Health Professionals Who Care for Cancer Patients
Systemic Therapy Update Volume 23 Issue 5 May 2020 For Health Professionals Who Care for Cancer Patients Inside This Issue: Editor’s Choice Benefit Drug List New Programs: Neoadjuvant Carboplatin for Triple-Negative New: BRLACTWAC, UGUPAPA, Pegaspargase (LKNOS, Breast Cancer (BRLACTWAC) | Apalutamide for Non- LYNOS), Bevacizumab (Pediatric), IGEV (Pediatric) | Metastatic Castration-Resistant Prostate Cancer (UGUPAPA) Deleted: GIGAVEOCAP, GIGAVEOF, SMAJIFN Drug Update NEW Protocols, PPPOs and Patient Handouts Pharmacare Update – Anticoagulants | Patient Assistance BR: BRLACTWAC | GU: UGUPAPA Programs REVISED Protocols, PPPOs and Patient Handouts Drug Shortages BR: BRAJAC, BRAJACT, BRAJACTG, BRAJACTT, BRAJACTTG, New: Alemtuzumab, Anagrelide | Updated: Hydroxyurea BRAJACTW, BRAJDAC, BRAJFEC, BRAJFECD, BRAJFECDT, Cancer Drug Manual© BRAVAC, BRLAACD, BRLAACDT, BRLATACG, BRLATWAC | New: Abemaciclib, Lurbinectedin | Revised: Apalutamide, GI: GIPGEMABR | GU: UGUPABI, UGUPENZ | LK: ULKBLIN, Durvalumab, Panitumumab, Pembrolizumab ULKMDSA | SA: SAIME | SC: SCDRUGRX | SM: USMAVDAB, ST Update Editorial Board USMAVDT, USMAVTRA, USMAVVC, USMAVVEM Membership Update Resources and Contact Information Editor’s Choice New Programs Effective 01 May 2020, the BC Cancer Provincial Systemic Therapy Program has approved the following new treatment programs. The full details of these programs can be found on the BC Cancer website in the Chemotherapy Protocols section. Breast Neoadjuvant Carboplatin for Locally Advanced, Triple-Negative Breast Cancer (BRLACTWAC) — The Provincial Systemic Therapy Program has approved the addition of carboplatin to the standard taxane- anthracycline neoadjuvant chemotherapy regimens, for patients with stage II and higher triple-negative breast cancer. This regimen consists of carboplatin (given every 3 weeks) in combination with weekly paclitaxel, followed by doxorubicin-cyclophosphamide (AC given every 2 or 3 weeks). Filgrastim support is available for the dose-dense AC option. -

BC Cancer Benefit Drug List September 2021
Page 1 of 65 BC Cancer Benefit Drug List September 2021 DEFINITIONS Class I Reimbursed for active cancer or approved treatment or approved indication only. Reimbursed for approved indications only. Completion of the BC Cancer Compassionate Access Program Application (formerly Undesignated Indication Form) is necessary to Restricted Funding (R) provide the appropriate clinical information for each patient. NOTES 1. BC Cancer will reimburse, to the Communities Oncology Network hospital pharmacy, the actual acquisition cost of a Benefit Drug, up to the maximum price as determined by BC Cancer, based on the current brand and contract price. Please contact the OSCAR Hotline at 1-888-355-0355 if more information is required. 2. Not Otherwise Specified (NOS) code only applicable to Class I drugs where indicated. 3. Intrahepatic use of chemotherapy drugs is not reimbursable unless specified. 4. For queries regarding other indications not specified, please contact the BC Cancer Compassionate Access Program Office at 604.877.6000 x 6277 or [email protected] DOSAGE TUMOUR PROTOCOL DRUG APPROVED INDICATIONS CLASS NOTES FORM SITE CODES Therapy for Metastatic Castration-Sensitive Prostate Cancer using abiraterone tablet Genitourinary UGUMCSPABI* R Abiraterone and Prednisone Palliative Therapy for Metastatic Castration Resistant Prostate Cancer abiraterone tablet Genitourinary UGUPABI R Using Abiraterone and prednisone acitretin capsule Lymphoma reversal of early dysplastic and neoplastic stem changes LYNOS I first-line treatment of epidermal -

Xagrid, INN-Anagrelide
SCIENTIFIC DISCUSSION 1. Introduction Anagrelide hydrochloride monohydrate is a novel imidazoquinazoline originally developed as an inhibitor of platelet aggregation but subsequently found to have value as a platelet-lowering agent for the treatment of patients suffering from Essential thrombocythaemia (ET). ET is one of a number of chronic myeloproliferative disorders characterised by an elevated platelet count due to an autonomous clonal proliferation of bone marrow megakaryocytes. The underlying cause of this proliferation is unknown. Platelet function is generally normal and the cells are able to undergo aggregation and to participate in the haemostatic process in a more or less normal manner. The diagnosis is one of exclusion of reactive causes and other myeloproliferative disorders (MPDs) according to internationally agreed criteria of the Polycythaemia Vera Study Group (PVSG). The main criterion is a sustained platelet level of >600 x 109/L for more than two months. It is estimated that the prevalence of ET in the European Union (EU) is 2-3 per 10,000 persons. ET is a condition that may affect anyone at any stage of life from childhood although the median age of presentation is 60 years. Literature and clinical experience indicate a preponderance of female patients between the ages of 25 and 50 years. Up to two thirds of patients with ET are asymptomatic and a significant number of patients are now being diagnosed as an incidental consequence of the analysis of full blood counts for other health reasons. Generally, the older patient is more likely to be symptomatic at presentation, although younger patients may present with major complications. -

ER Guide to Bleeding Disorders
Bleeding disorders ER guide to bleeding disorders 1 Table of contents 4 General Guidelines 4–5 national Hemophilia Foundation guidelines 5–10 Treatment options 10 HemopHilia a Name:__________________________________________________________________________________________________ 10–11 national Hemophilia Foundation guidelines Address:________________________________________________________________________________________________ 12 dosage chart Phone:__________________________________________________________________________________________________ 14–15 Treatment products 16 HemopHilia B In case of emergency, contact: ______________________________________________________________________________ 16 national Hemophilia Foundation guidelines Relation to patient:________________________________________________________________________________________ 17 dosage chart 18 Treatment products 19 HemopHilia a or B with inHiBiTors Diagnosis: Hemophilia A: Mild Moderate Severe 20 national Hemophilia Foundation guidelines Inhibitors Inhibitors Bethesda units (if known) ____________________________________ 21 Treatment products Hemophilia B: Mild Moderate Severe 22–23 Von willeBrand disease Inhibitors Inhibitors Bethesda units (if known) ____________________________________ 23–24 national Hemophilia Foundation guidelines von Willebrand disease: Type 1 Type 2 Type 3 Platelet type 25 Treatment products 27 Bibliography Preferred product:_________________________________________________________________________________________ Dose for life-threatening -
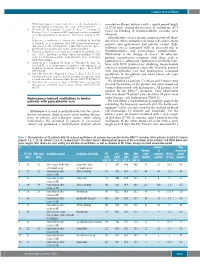
Hydroxyurea Induced Oscillations in Twelve Patients with Polycythemia
Letters to the Editor JAK2 haplotype is a major risk factor for the development of revealed oscillatory behavior with a typical period length myeloproliferative neoplasms. Nat Genet. 2009;41(4):446-9. of 27-30 days. During the presence of oscillations (25.5 6. Olcaydu D, Harutyunyan A, Jager R, Berg T, Gisslinger B, Pabinger I, et al. A common JAK2 haplotype confers susceptibil- years) no bleeding or thromboembolic episodes were ity to myeloproliferative neoplasms. Nat Genet. 2009;41(4):450- observed. 4. Polycythemia vera is a chronic acquired stem cell disor- 7. Kilpivaara O, Mukherjee S, Schram AM, Wadleigh M, Mullally der which affects primarily red blood cells and in many A, Ebert BL, et al. A germline JAK2 SNP is associated with pre- patients also granulocyte and platelet counts. Poly - disposition to the development of JAK2V617F-positive myelo- cythemia vera is associated with an increased risk of proliferative neoplasms. Nat Genet. 2009;41(4):455-9. 1 8. Olcaydu D, Skoda RC, Looser R, Li S, Cazzola M, Pietra D, et al. thromboembolic and hemorrhagic complications. The 'GGCC' haplotype of JAK2 confers susceptibility to JAK2 Phlebotomy is the therapy of choice.2 In refractory exon 12 mutation-positive polycythemia vera. Leukemia. patients cytoreductive treatment with drugs such as 2009;23(10):1924-6. hydroxyurea is advocated. Hydroxyurea primarily inter- 9. Harrison CN, Campbell PJ, Buck G, Wheatley K, East CL, Bareford D, et al. Hydroxyurea Compared with Anagrelide in feres with DNA synthesis by inhibiting ribonucleotide 3 High-Risk Essential Thrombocythemia. N Engl J Med. 2005; reductase in hematopoietic stem cells. -

Pharmaceuticals As Environmental Contaminants
PharmaceuticalsPharmaceuticals asas EnvironmentalEnvironmental Contaminants:Contaminants: anan OverviewOverview ofof thethe ScienceScience Christian G. Daughton, Ph.D. Chief, Environmental Chemistry Branch Environmental Sciences Division National Exposure Research Laboratory Office of Research and Development Environmental Protection Agency Las Vegas, Nevada 89119 [email protected] Office of Research and Development National Exposure Research Laboratory, Environmental Sciences Division, Las Vegas, Nevada Why and how do drugs contaminate the environment? What might it all mean? How do we prevent it? Office of Research and Development National Exposure Research Laboratory, Environmental Sciences Division, Las Vegas, Nevada This talk presents only a cursory overview of some of the many science issues surrounding the topic of pharmaceuticals as environmental contaminants Office of Research and Development National Exposure Research Laboratory, Environmental Sciences Division, Las Vegas, Nevada A Clarification We sometimes loosely (but incorrectly) refer to drugs, medicines, medications, or pharmaceuticals as being the substances that contaminant the environment. The actual environmental contaminants, however, are the active pharmaceutical ingredients – APIs. These terms are all often used interchangeably Office of Research and Development National Exposure Research Laboratory, Environmental Sciences Division, Las Vegas, Nevada Office of Research and Development Available: http://www.epa.gov/nerlesd1/chemistry/pharma/image/drawing.pdfNational -

Preoperative Medication Directive a Clinical Practice Protocol Practitioner Information
Preoperative Medication Directive A Clinical Practice Protocol Practitioner Information Please note: this is a living document subject to change. We recommend that this not be printed but rather accessed on line for the most current information. Nov 2017 Table of Contents Regional practice protocol for Preoperative Medication Page Directives Purpose & Intent 1 Practice Outcomes 1 Background 2 Components 2 References 3 Authors 4 Guidelines- Section A (for select Nurses) 5 Guidelines- Section B (for Surgeons) 12 Guidelines- Section C (for Anaesthesiologists) pending PURPOSE AND INTENT Who this document is for: These sectioned documents are a resource and an information source for Preoperative Assessment/Anaesthesia Clinic Staff, Surgeons and Anaesthesiologists. Each Section (A, B, etc...) was created for a primary user group. Users of this document shall always employ critical thinking and seek additional guidance from the appropriate colleagues (ie: anaesthesiologist and/or surgeon/prescribing physician) when concerns or variances arise. 1. Practice Outcomes How to use this document: This tool is to supplement the practice at each site. Implementation of this document will be at the discretion of the site leaders, however the directions provided to patients regarding preoperative medications should be consistent with this document (under the consideration of critical thinking). Clinical judgment, critical thinking and patient specific needs & concerns should always be employed, considered and addressed for all parts of the document. Any care provider with a Preoperative Medication Directive medication question is asked to submit in writing the medication in question (along with any supporting evidence if available) to the PAC Working Group (chair) [email protected] for consideration. -

Cancer Drug Counselling: a Guide for Pharmacists
CANCER DRUG COUNSELLING: A GUIDE FOR PHARMACISTS CANCER DRUG COUNSELLING A Guide for Pharmacists 1st Edition, 2017 Pharmaceutical Services Division Ministry of Health, Malaysia Pharmaceutical Services Division, Ministry of Health Malaysia 1 DISCLAIMER This guide would serve as a handy reference for PHARMACIST ONLY and not as a complete drug information resource. It is NOT intended to replicate or replace the knowledge, skills and experience of trained oncology health professionals, nor is it a substitute for clinical judgement and advice. The nature of healthcare/drug information is that it is constantly evolving with ongoing research and clinical experience and is often subject to interpretation. While best effort has been made to ensure the accuracy of the information and recommendation presented, reader is advised that the contributors, editors, reviewers and publishers cannot be responsible for the continued updates of the information, of any errors or/and of any consequences arising from its application. ALL RIGHTS RESERVED This is a publication of the Pharmaceutical Services Division, Ministry of Health Malaysia (MOH). Enquiries are to be directed to the address below. Permission is hereby granted to reproduce information contained herein provided that such reproduction be given due acknowledgement and shall not modify the text. Pharmaceutical Services Division Ministry of Health Malaysia Lot 36, Jalan Universiti 46350 Petaling Jaya Selangor, Malaysia Tel: 603 - 7841 3200 Fax 603 - 7968 2222 Website: www.pharmacy.gov.my 2 Pharmaceutical Services Division, Ministry of Health Malaysia PREFACE Providing pharmaceutical care to patients through education and counselling can contribute towards enhancing patients’ adherence to cancer drug therapy as well as to reduce medication related problems. -

Product List 2019 Finished Dosage Forms (Partner Label), Active Pharmaceutical Ingredients
Product List 2019 Finished Dosage Forms (Partner Label), Active Pharmaceutical Ingredients B2B DISCLAIMER Product exploitation, including development, sales and offer for sale are performed where permissible by patent law. This presentation is not and should not constitute an offer for sale in territories where it is not permitted by law. The availability of some of the listed products may be limited by geographical territory. All products are intended only to Sandoz B2B. 2 Finished Dosage Forms (Partner Label) Anti-Infectives LATIN ORAL EUROPE AMERICA APMA Aciclovir • • Amoxicillin Trihydrate • • • Amoxicillin Trihydrate + Potassium Clavulanate • • • Azithromycin Dihydrate • • Azithromycin Monohydrate • Cefadroxil • Cefalexin • • Cefuroxime Axetil • • Ciprofloxacin Hydrochloride • • Clarithromycin • • Doxycycline • Efavirenz • Efavirenz + Emtricitabine + Tenofovir Disoproxil (Succinate) • Entecavir • Lamivudine + Zidovudine • • Levofloxacin • Linezolid • Moxifloxacin Hydrochloride • • Norfloxacin • Penicillin V Potassium • • Rifampicin + Isoniazid • Rifampicin + Isoniazid + Pyrazinamide • Roxithromycin • Valganciclovir Hydrochloride • Voriconazole • • STERILE Aciclovir • Amoxicillin Sodium + Potassium Clavulanate • Cefazolin Sodium • Cefepime Hydrochloride • Cefotaxime Sodium • • Ceftazidime • Ceftriaxone Sodium • • Cefuroxime Sodium • Penicillin G Benzathine • Piperacillin Sodium + Tazobactam Sodium • • Vancomycin Hydrochloride • • Voriconazole • The availability of some of the listed products may be limited by geographical territory. -

Lists of Medicinal Products for Rare Diseases in Europe*
March 2021 Lists of medicinal products for rare diseases in Europe* the www.orpha.net www.orphadata.org General Table of contents PART 1: List of orphan medicinal products in Europe with European orphan designation and European marketing authorization 3 Table of contents 3 Methodology 3 Classification by tradename 5 Annex 1: Orphan medicinal products withdrawn from the European Community Register of orphan medicinal products 22 Annex 2: Orphan medicinal products withdrawn from use in the European Union 31 Classification by date of MA in descending order 33 Classification by ATC category 34 Classification by MA holder 35 PART 2 : 37 List of medicinal products intended for rare diseases in Europe with European marketing authorization without an orphan designation in Europe 37 Table of contents 37 Methodology 37 Classification by tradename 38 Classification by date of MA in descending order 104 Classification by ATC category 106 Classification by MA holder 108 For any questions or comments, please contact us: [email protected] Orphanet Report Series - Lists of medicinal products for rare diseases in Europe. March 2021 http://www.orpha.net/orphacom/cahiers/docs/GB/list_of_orphan_drugs_in_europe.pdf 2 PART 1: List of orphan medicinal products in Europe with European orphan designation and European marketing authorization* Table of contents List of orphan medicinal products in Europe with European orphan designation and European marketing authorisation* 3 Methodology 3 Classification by tradename 5 Annex 1: Orphan medicinal products removed or withdrawn from the European Community Register of orphan medicinal products 22 Annex 2: Orphan medicinal products withdrawn from use in the European Union 31 Classification by date of MA in descending order 33 Classification by ATC category 34 Classification by MA holder 35 Methodology This part of the document provides the list of all orphan with the list of medicinal products that have been granted a medicinal products that have received a European Marketing marketing authorization (http://ec.europa.