Anti-Histone H2B Phospho (Ser14) Antibody [RM238] (ARG57238)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Analysis of Trans Esnps Infers Regulatory Network Architecture
Analysis of trans eSNPs infers regulatory network architecture Anat Kreimer Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate School of Arts and Sciences COLUMBIA UNIVERSITY 2014 © 2014 Anat Kreimer All rights reserved ABSTRACT Analysis of trans eSNPs infers regulatory network architecture Anat Kreimer eSNPs are genetic variants associated with transcript expression levels. The characteristics of such variants highlight their importance and present a unique opportunity for studying gene regulation. eSNPs affect most genes and their cell type specificity can shed light on different processes that are activated in each cell. They can identify functional variants by connecting SNPs that are implicated in disease to a molecular mechanism. Examining eSNPs that are associated with distal genes can provide insights regarding the inference of regulatory networks but also presents challenges due to the high statistical burden of multiple testing. Such association studies allow: simultaneous investigation of many gene expression phenotypes without assuming any prior knowledge and identification of unknown regulators of gene expression while uncovering directionality. This thesis will focus on such distal eSNPs to map regulatory interactions between different loci and expose the architecture of the regulatory network defined by such interactions. We develop novel computational approaches and apply them to genetics-genomics data in human. We go beyond pairwise interactions to define network motifs, including regulatory modules and bi-fan structures, showing them to be prevalent in real data and exposing distinct attributes of such arrangements. We project eSNP associations onto a protein-protein interaction network to expose topological properties of eSNPs and their targets and highlight different modes of distal regulation. -

Download Download
Supplementary Figure S1. Results of flow cytometry analysis, performed to estimate CD34 positivity, after immunomagnetic separation in two different experiments. As monoclonal antibody for labeling the sample, the fluorescein isothiocyanate (FITC)- conjugated mouse anti-human CD34 MoAb (Mylteni) was used. Briefly, cell samples were incubated in the presence of the indicated MoAbs, at the proper dilution, in PBS containing 5% FCS and 1% Fc receptor (FcR) blocking reagent (Miltenyi) for 30 min at 4 C. Cells were then washed twice, resuspended with PBS and analyzed by a Coulter Epics XL (Coulter Electronics Inc., Hialeah, FL, USA) flow cytometer. only use Non-commercial 1 Supplementary Table S1. Complete list of the datasets used in this study and their sources. GEO Total samples Geo selected GEO accession of used Platform Reference series in series samples samples GSM142565 GSM142566 GSM142567 GSM142568 GSE6146 HG-U133A 14 8 - GSM142569 GSM142571 GSM142572 GSM142574 GSM51391 GSM51392 GSE2666 HG-U133A 36 4 1 GSM51393 GSM51394 only GSM321583 GSE12803 HG-U133A 20 3 GSM321584 2 GSM321585 use Promyelocytes_1 Promyelocytes_2 Promyelocytes_3 Promyelocytes_4 HG-U133A 8 8 3 GSE64282 Promyelocytes_5 Promyelocytes_6 Promyelocytes_7 Promyelocytes_8 Non-commercial 2 Supplementary Table S2. Chromosomal regions up-regulated in CD34+ samples as identified by the LAP procedure with the two-class statistics coded in the PREDA R package and an FDR threshold of 0.5. Functional enrichment analysis has been performed using DAVID (http://david.abcc.ncifcrf.gov/) -

Integrating Single-Step GWAS and Bipartite Networks Reconstruction Provides Novel Insights Into Yearling Weight and Carcass Traits in Hanwoo Beef Cattle
animals Article Integrating Single-Step GWAS and Bipartite Networks Reconstruction Provides Novel Insights into Yearling Weight and Carcass Traits in Hanwoo Beef Cattle Masoumeh Naserkheil 1 , Abolfazl Bahrami 1 , Deukhwan Lee 2,* and Hossein Mehrban 3 1 Department of Animal Science, University College of Agriculture and Natural Resources, University of Tehran, Karaj 77871-31587, Iran; [email protected] (M.N.); [email protected] (A.B.) 2 Department of Animal Life and Environment Sciences, Hankyong National University, Jungang-ro 327, Anseong-si, Gyeonggi-do 17579, Korea 3 Department of Animal Science, Shahrekord University, Shahrekord 88186-34141, Iran; [email protected] * Correspondence: [email protected]; Tel.: +82-31-670-5091 Received: 25 August 2020; Accepted: 6 October 2020; Published: 9 October 2020 Simple Summary: Hanwoo is an indigenous cattle breed in Korea and popular for meat production owing to its rapid growth and high-quality meat. Its yearling weight and carcass traits (backfat thickness, carcass weight, eye muscle area, and marbling score) are economically important for the selection of young and proven bulls. In recent decades, the advent of high throughput genotyping technologies has made it possible to perform genome-wide association studies (GWAS) for the detection of genomic regions associated with traits of economic interest in different species. In this study, we conducted a weighted single-step genome-wide association study which combines all genotypes, phenotypes and pedigree data in one step (ssGBLUP). It allows for the use of all SNPs simultaneously along with all phenotypes from genotyped and ungenotyped animals. Our results revealed 33 relevant genomic regions related to the traits of interest. -

Genome-Wide Screen of Cell-Cycle Regulators in Normal and Tumor Cells
bioRxiv preprint doi: https://doi.org/10.1101/060350; this version posted June 23, 2016. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Genome-wide screen of cell-cycle regulators in normal and tumor cells identifies a differential response to nucleosome depletion Maria Sokolova1, Mikko Turunen1, Oliver Mortusewicz3, Teemu Kivioja1, Patrick Herr3, Anna Vähärautio1, Mikael Björklund1, Minna Taipale2, Thomas Helleday3 and Jussi Taipale1,2,* 1Genome-Scale Biology Program, P.O. Box 63, FI-00014 University of Helsinki, Finland. 2Science for Life laboratory, Department of Biosciences and Nutrition, Karolinska Institutet, SE- 141 83 Stockholm, Sweden. 3Science for Life laboratory, Division of Translational Medicine and Chemical Biology, Department of Medical Biochemistry and Biophysics, Karolinska Institutet, S-171 21 Stockholm, Sweden To identify cell cycle regulators that enable cancer cells to replicate DNA and divide in an unrestricted manner, we performed a parallel genome-wide RNAi screen in normal and cancer cell lines. In addition to many shared regulators, we found that tumor and normal cells are differentially sensitive to loss of the histone genes transcriptional regulator CASP8AP2. In cancer cells, loss of CASP8AP2 leads to a failure to synthesize sufficient amount of histones in the S-phase of the cell cycle, resulting in slowing of individual replication forks. Despite this, DNA replication fails to arrest, and tumor cells progress in an elongated S-phase that lasts several days, finally resulting in death of most of the affected cells. -

Whole Exome Sequencing in Families at High Risk for Hodgkin Lymphoma: Identification of a Predisposing Mutation in the KDR Gene
Hodgkin Lymphoma SUPPLEMENTARY APPENDIX Whole exome sequencing in families at high risk for Hodgkin lymphoma: identification of a predisposing mutation in the KDR gene Melissa Rotunno, 1 Mary L. McMaster, 1 Joseph Boland, 2 Sara Bass, 2 Xijun Zhang, 2 Laurie Burdett, 2 Belynda Hicks, 2 Sarangan Ravichandran, 3 Brian T. Luke, 3 Meredith Yeager, 2 Laura Fontaine, 4 Paula L. Hyland, 1 Alisa M. Goldstein, 1 NCI DCEG Cancer Sequencing Working Group, NCI DCEG Cancer Genomics Research Laboratory, Stephen J. Chanock, 5 Neil E. Caporaso, 1 Margaret A. Tucker, 6 and Lynn R. Goldin 1 1Genetic Epidemiology Branch, Division of Cancer Epidemiology and Genetics, National Cancer Institute, NIH, Bethesda, MD; 2Cancer Genomics Research Laboratory, Division of Cancer Epidemiology and Genetics, National Cancer Institute, NIH, Bethesda, MD; 3Ad - vanced Biomedical Computing Center, Leidos Biomedical Research Inc.; Frederick National Laboratory for Cancer Research, Frederick, MD; 4Westat, Inc., Rockville MD; 5Division of Cancer Epidemiology and Genetics, National Cancer Institute, NIH, Bethesda, MD; and 6Human Genetics Program, Division of Cancer Epidemiology and Genetics, National Cancer Institute, NIH, Bethesda, MD, USA ©2016 Ferrata Storti Foundation. This is an open-access paper. doi:10.3324/haematol.2015.135475 Received: August 19, 2015. Accepted: January 7, 2016. Pre-published: June 13, 2016. Correspondence: [email protected] Supplemental Author Information: NCI DCEG Cancer Sequencing Working Group: Mark H. Greene, Allan Hildesheim, Nan Hu, Maria Theresa Landi, Jennifer Loud, Phuong Mai, Lisa Mirabello, Lindsay Morton, Dilys Parry, Anand Pathak, Douglas R. Stewart, Philip R. Taylor, Geoffrey S. Tobias, Xiaohong R. Yang, Guoqin Yu NCI DCEG Cancer Genomics Research Laboratory: Salma Chowdhury, Michael Cullen, Casey Dagnall, Herbert Higson, Amy A. -

IGF1R Deficiency Attenuates Acute Inflammatory Response in A
www.nature.com/scientificreports OPEN IGF1R deficiency attenuates acute inflammatory response in a bleomycin-induced lung injury Received: 7 November 2016 Accepted: 17 May 2017 mouse model Published: xx xx xxxx Sergio Piñeiro-Hermida1, Icíar P. López1, Elvira Alfaro-Arnedo1, Raquel Torrens1, María Iñiguez2, Lydia Alvarez-Erviti3, Carlos Ruíz-Martínez4 & José G. Pichel 1 IGF1R (Insulin-like Growth Factor 1 Receptor) is a tyrosine kinase with pleiotropic cellular functions. IGF activity maintains human lung homeostasis and is implicated in pulmonary diseases such as cancer, ARDS, COPD, asthma and fibrosis. Here we report that lung transcriptome analysis in mice with a postnatally-induced Igf1r gene deletion showed differentially expressed genes with potentially protective roles related to epigenetics, redox and oxidative stress. After bleomycin-induced lung injury, IGF1R-deficient mice demonstrated improved survival within a week. Three days post injury, IGF1R- deficient lungs displayed changes in expression of IGF system-related genes and reduced vascular fragility and permeability. Mutant lungs presented reduced inflamed area, down-regulation of pro- inflammatory markers and up-regulation of resolution indicators. Decreased inflammatory cell presence in BALF was reflected in diminished lung infiltration mainly affecting neutrophils, also corroborated by reduced neutrophil numbers in bone marrow, as well as reduced lymphocyte and alveolar macrophage counts. Additionally, increased SFTPC expression together with hindered HIF1A expression and augmented levels of Gpx8 indicate that IGF1R deficiency protects against alveolar damage. These findings identify IGF1R as an important player in murine acute lung inflammation, suggesting that targeting IGF1R may counteract the inflammatory component of many lung diseases. Inflammation is a relevant component of many lung diseases including ARDS, COPD, asthma, cancer, fibrosis and pneumonia1–5. -

Ernas and Superenhancer Lncrnas Are Functional in Human Prostate Cancer
Hindawi Disease Markers Volume 2020, Article ID 8847986, 17 pages https://doi.org/10.1155/2020/8847986 Research Article eRNAs and Superenhancer lncRNAs Are Functional in Human Prostate Cancer Xiaona Zhang,1,2,3 Panpan Pang,2,3,4 Min Jiang,1,2,3 Qunfa Cao,2,3 Huili Li,2,3 Yi Xu,5 Yao Li ,4 Xue Chen ,1 and Junsong Han 2,3 1Department of Pathology, Tongji Hospital, Tongji University School of Medicine, Shanghai, China 2National Engineering Center for Biochip at Shanghai, Shanghai, China 3Shanghai Biochip Corporation, Shanghai, China 4Shanghai Engineering Research Center of Industrial Microorganisms, School of Life Science, Fudan University, Shanghai, China 5Yueyang Hospital of Integrated Traditional Chinese and Western Medicine, Shanghai University of Chinese Medicine, Shanghai, China Correspondence should be addressed to Yao Li; [email protected], Xue Chen; [email protected], and Junsong Han; [email protected] Received 23 June 2020; Revised 27 July 2020; Accepted 14 August 2020; Published 24 September 2020 Academic Editor: Mingjun Shi Copyright © 2020 Xiaona Zhang et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Prostate cancer (PCa) is one of the most commonly diagnosed cancers in males worldwide. lncRNAs (long noncoding RNAs) play a significant role in the occurrence and development of PCa. eRNAs (enhancer RNAs) and SE-lncRNAs (superenhancer lncRNAs) are important elements of lncRNAs, but the role of eRNAs and SE-lncRNAs in PCa remains largely unclear. In this work, we identified 681 eRNAs and 292 SE-lncRNAs that were expressed differentially in PCa using a microarray. -

Supplemental Data.Pdf
Supplementary material -Table of content Supplementary Figures (Fig 1- Fig 6) Supplementary Tables (1-13) Lists of genes belonging to distinct biological processes identified by GREAT analyses to be significantly enriched with UBTF1/2-bound genes Supplementary Table 14 List of the common UBTF1/2 bound genes within +/- 2kb of their TSSs in NIH3T3 and HMECs. Supplementary Table 15 List of gene identified by microarray expression analysis to be differentially regulated following UBTF1/2 knockdown by siRNA Supplementary Table 16 List of UBTF1/2 binding regions overlapping with histone genes in NIH3T3 cells Supplementary Table 17 List of UBTF1/2 binding regions overlapping with histone genes in HMEC Supplementary Table 18 Sequences of short interfering RNA oligonucleotides Supplementary Table 19 qPCR primer sequences for qChIP experiments Supplementary Table 20 qPCR primer sequences for reverse transcription-qPCR Supplementary Table 21 Sequences of primers used in CHART-PCR Supplementary Methods Supplementary Fig 1. (A) ChIP-seq analysis of UBTF1/2 and Pol I (POLR1A) binding across mouse rDNA. UBTF1/2 is enriched at the enhancer and promoter regions and along the entire transcribed portions of rDNA with little if any enrichment in the intergenic spacer (IGS), which separates the rDNA repeats. This enrichment coincides with the distribution of the largest subunit of Pol I (POLR1A) across the rDNA. All sequencing reads were mapped to the published complete sequence of the mouse rDNA repeat (Gene bank accession number: BK000964). The graph represents the frequency of ribosomal sequences enriched in UBTF1/2 and Pol I-ChIPed DNA expressed as fold change over those of input genomic DNA. -

HIST1H2BB and MAGI2 Methylation and Somatic Mutations As Precision Medicine Biomarkers for Diagnosis and Prognosis of High-Grade Serous Ovarian Cancer Blanca L
Published OnlineFirst June 24, 2020; DOI: 10.1158/1940-6207.CAPR-19-0412 CANCER PREVENTION RESEARCH | RESEARCH ARTICLE HIST1H2BB and MAGI2 Methylation and Somatic Mutations as Precision Medicine Biomarkers for Diagnosis and Prognosis of High-grade Serous Ovarian Cancer Blanca L. Valle1, Sebastian Rodriguez-Torres2,3, Elisabetta Kuhn4,5, Teresa Díaz-Montes6, Edgardo Parrilla-Castellar7, Fahcina P. Lawson1, Oluwasina Folawiyo1, Carmen Ili-Gangas8, Priscilla Brebi-Mieville8, James R. Eshleman9, James Herman3, Ie-Ming Shih4, David Sidransky1, and Rafael Guerrero-Preston1,10,11 ABSTRACT ◥ Molecular alterations that contribute to long-term (LT) (n ¼ 35). Immunoblot and clonogenic assays after pharma- and short-term (ST) survival in ovarian high-grade serous cologic unmasking show that HIST1H2BB and MAGI2 carcinoma (HGSC) may be used as precision medicine promoter methylation downregulates mRNA expression biomarkers. DNA promoter methylation is an early event levels in ovarian cancer cells. We then used qMSP in paired in tumorigenesis, which can be detected in blood and urine, tissue, ascites, plasma/serum, vaginal swabs, and urine from making it a feasible companion biomarker to somatic muta- a third cohort of patients with HGSC cancer (n ¼ 85) to test tions for early detection and targeted treatment workflows. the clinical potential of HIST1H2BB and MAGI2 in precision We compared the methylation profile in 12 HGSC tissue medicine workflows. We also performed next-generation samples to 30 fallopian tube epithelium samples, using the exome sequencing of 50 frequently mutated in human cancer Infinium Human Methylation 450K Array. We also used genes, using the Ion AmpliSeqCancer Hotspot Panel, to 450K methylation arrays to compare methylation among show that the somatic mutation profile found in tissue and HGSCs long-term survivors (more than 5 years) and short- plasma can be quantified in paired urine samples from term survivors (less than 3 years). -

The Pdx1 Bound Swi/Snf Chromatin Remodeling Complex Regulates Pancreatic Progenitor Cell Proliferation and Mature Islet Β Cell
Page 1 of 125 Diabetes The Pdx1 bound Swi/Snf chromatin remodeling complex regulates pancreatic progenitor cell proliferation and mature islet β cell function Jason M. Spaeth1,2, Jin-Hua Liu1, Daniel Peters3, Min Guo1, Anna B. Osipovich1, Fardin Mohammadi3, Nilotpal Roy4, Anil Bhushan4, Mark A. Magnuson1, Matthias Hebrok4, Christopher V. E. Wright3, Roland Stein1,5 1 Department of Molecular Physiology and Biophysics, Vanderbilt University, Nashville, TN 2 Present address: Department of Pediatrics, Indiana University School of Medicine, Indianapolis, IN 3 Department of Cell and Developmental Biology, Vanderbilt University, Nashville, TN 4 Diabetes Center, Department of Medicine, UCSF, San Francisco, California 5 Corresponding author: [email protected]; (615)322-7026 1 Diabetes Publish Ahead of Print, published online June 14, 2019 Diabetes Page 2 of 125 Abstract Transcription factors positively and/or negatively impact gene expression by recruiting coregulatory factors, which interact through protein-protein binding. Here we demonstrate that mouse pancreas size and islet β cell function are controlled by the ATP-dependent Swi/Snf chromatin remodeling coregulatory complex that physically associates with Pdx1, a diabetes- linked transcription factor essential to pancreatic morphogenesis and adult islet-cell function and maintenance. Early embryonic deletion of just the Swi/Snf Brg1 ATPase subunit reduced multipotent pancreatic progenitor cell proliferation and resulted in pancreas hypoplasia. In contrast, removal of both Swi/Snf ATPase subunits, Brg1 and Brm, was necessary to compromise adult islet β cell activity, which included whole animal glucose intolerance, hyperglycemia and impaired insulin secretion. Notably, lineage-tracing analysis revealed Swi/Snf-deficient β cells lost the ability to produce the mRNAs for insulin and other key metabolic genes without effecting the expression of many essential islet-enriched transcription factors. -

HIST1H2BB and MAGI2 Methylation and Somatic Mutations As Precision Medicine Biomarkers for Diagnosis and Prognosis of High-Grade Serous Ovarian Cancer Blanca L
Author Manuscript Published OnlineFirst on June 24, 2020; DOI: 10.1158/1940-6207.CAPR-19-0412 Author manuscripts have been peer reviewed and accepted for publication but have not yet been edited. HIST1H2BB and MAGI2 methylation and somatic mutations as precision medicine biomarkers for diagnosis and prognosis of high-grade serous ovarian cancer Blanca L. Valle1, Sebastian Rodriguez-Torres2,3, Elisabetta Kuhn4,5, Teresa Díaz-Montes6, Edgardo Parrilla-Castellar7, Fahcina P. Lawson1, Oluwasina Folawiyo1, Carmen Ili-Gangas8, Priscilla Brebi-Mieville8, James R. Eshleman9, James Herman3, Ie-Ming Shih4, David Sidransky1, Rafael Guerrero-Preston1, 10,11* 1Otolaryngology Department, Head and Neck Cancer Research Division, The Johns Hopkins University, School of Medicine, Baltimore, MD 2Department of Epidemiology, Harvard T.H. Chan School of Public Health, Boston, Massachusetts, USA 3Department of Medicine, University of Pittsburgh, School of Medicine, Pittsburgh, Pennsylvania, USA 4Division of Pathology, Fondazione IRCCS Ca' Granda, Ospedale Maggiore Policlinico; Department of Biomedical, Surgical, and Dental Sciences, University of Milan, Italy 5Departments of Pathology, Gynecology and Obstetrics, The Johns Hopkins University, School of Medicine, Baltimore, MD 6The Lya Segall Ovarian Cancer Institute, Mercy Medical Center, Baltimore, MD 7Department of Pathology, University of Washington, Seattle, WA 8Laboratory Integrative Biology (LIBi), Center for Excellence in Translational Medicine- Scientific and Technological Bioresources Nucleus (CEMT-BIOREN), -
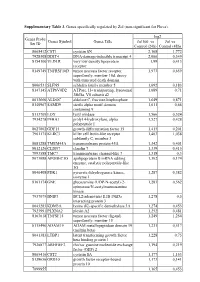
Supplementary Table 3. Genes Specifically Regulated by Zol (Non-Significant for Fluva)
Supplementary Table 3. Genes specifically regulated by Zol (non-significant for Fluva). log2 Genes Probe Genes Symbol Genes Title Zol100 vs Zol vs Set ID Control (24h) Control (48h) 8065412 CST1 cystatin SN 2,168 1,772 7928308 DDIT4 DNA-damage-inducible transcript 4 2,066 0,349 8154100 VLDLR very low density lipoprotein 1,99 0,413 receptor 8149749 TNFRSF10D tumor necrosis factor receptor 1,973 0,659 superfamily, member 10d, decoy with truncated death domain 8006531 SLFN5 schlafen family member 5 1,692 0,183 8147145 ATP6V0D2 ATPase, H+ transporting, lysosomal 1,689 0,71 38kDa, V0 subunit d2 8013660 ALDOC aldolase C, fructose-bisphosphate 1,649 0,871 8140967 SAMD9 sterile alpha motif domain 1,611 0,66 containing 9 8113709 LOX lysyl oxidase 1,566 0,524 7934278 P4HA1 prolyl 4-hydroxylase, alpha 1,527 0,428 polypeptide I 8027002 GDF15 growth differentiation factor 15 1,415 0,201 7961175 KLRC3 killer cell lectin-like receptor 1,403 1,038 subfamily C, member 3 8081288 TMEM45A transmembrane protein 45A 1,342 0,401 8012126 CLDN7 claudin 7 1,339 0,415 7993588 TMC7 transmembrane channel-like 7 1,318 0,3 8073088 APOBEC3G apolipoprotein B mRNA editing 1,302 0,174 enzyme, catalytic polypeptide-like 3G 8046408 PDK1 pyruvate dehydrogenase kinase, 1,287 0,382 isozyme 1 8161174 GNE glucosamine (UDP-N-acetyl)-2- 1,283 0,562 epimerase/N-acetylmannosamine kinase 7937079 BNIP3 BCL2/adenovirus E1B 19kDa 1,278 0,5 interacting protein 3 8043283 KDM3A lysine (K)-specific demethylase 3A 1,274 0,453 7923991 PLXNA2 plexin A2 1,252 0,481 8163618 TNFSF15 tumor necrosis