Pesticidal Compound Mixtures
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Historical Perspectives on Apple Production: Fruit Tree Pest Management, Regulation and New Insecticidal Chemistries
Historical Perspectives on Apple Production: Fruit Tree Pest Management, Regulation and New Insecticidal Chemistries. Peter Jentsch Extension Associate Department of Entomology Cornell University's Hudson Valley Lab 3357 Rt. 9W; PO box 727 Highland, NY 12528 email: [email protected] Phone 845-691-7151 Mobile: 845-417-7465 http://www.nysaes.cornell.edu/ent/faculty/jentsch/ 2 Historical Perspectives on Fruit Production: Fruit Tree Pest Management, Regulation and New Chemistries. by Peter Jentsch I. Historical Use of Pesticides in Apple Production Overview of Apple Production and Pest Management Prior to 1940 Synthetic Pesticide Development and Use II. Influences Changing the Pest Management Profile in Apple Production Chemical Residues in Early Insect Management Historical Chemical Regulation Recent Regulation Developments Changing Pest Management Food Quality Protection Act of 1996 The Science Behind The Methodology Pesticide Revisions – Requirements For New Registrations III. Resistance of Insect Pests to Insecticides Resistance Pest Management Strategies IV. Reduced Risk Chemistries: New Modes of Action and the Insecticide Treadmill Fermentation Microbial Products Bt’s, Abamectins, Spinosads Juvenile Hormone Analogs Formamidines, Juvenile Hormone Analogs And Mimics Insect Growth Regulators Azadirachtin, Thiadiazine Neonicotinyls Major Reduced Risk Materials: Carboxamides, Carboxylic Acid Esters, Granulosis Viruses, Diphenyloxazolines, Insecticidal Soaps, Benzoyl Urea Growth Regulators, Tetronic Acids, Oxadiazenes , Particle Films, Phenoxypyrazoles, Pyridazinones, Spinosads, Tetrazines , Organotins, Quinolines. 3 I Historical Use of Pesticides in Apple Production Overview of Apple Production and Pest Management Prior to 1940 The apple has a rather ominous origin. Its inception is framed in the biblical text regarding the genesis of mankind. The backdrop appears to be the turbulent setting of what many scholars believe to be present day Iraq. -

WO 2010/128115 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 11 November 2010 (11.11.2010) WO 2010/128115 Al (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every C12N 9/16 (2006.01) kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, (21) International Application Number: CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, PCT/EP20 10/056204 DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (22) International Filing Date: HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, 6 May 2010 (06.05.2010) KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, (25) Filing Language: English NO, NZ, OM, PE, PG, PH, PL, PT, RO, RS, RU, SC, SD, (26) Publication Language: English SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: 09159520.7 6 May 2009 (06.05.2009) EP (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (71) Applicant (for all designated States except US): GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, NOVOZYMES A/S [DK/DK]; Krogshoejvej 36, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, DK-2880 Bagsvaerd (DK). -

Environmental Properties of Chemicals Volume 2
1 t ENVIRONMENTAL 1 PROTECTION Esa Nikunen . Riitta Leinonen Birgit Kemiläinen • Arto Kultamaa Environmental properties of chemicals Volume 2 1 O O O O O O O O OO O OOOOOO Ol OIOOO FINNISH ENVIRONMENT INSTITUTE • EDITA Esa Nikunen e Riitta Leinonen Birgit Kemiläinen • Arto Kultamaa Environmental properties of chemicals Volume 2 HELSINKI 1000 OlO 00000001 00000000000000000 Th/s is a second revfsed version of Environmental Properties of Chemica/s, published by VAPK-Pub/ishing and Ministry of Environment, Environmental Protection Department as Research Report 91, 1990. The pubiication is also available as a CD ROM version: EnviChem 2.0, a PC database runniny under Windows operating systems. ISBN 951-7-2967-2 (publisher) ISBN 952-7 1-0670-0 (co-publisher) ISSN 1238-8602 Layout: Pikseri Julkaisupalvelut Cover illustration: Jussi Hirvi Edita Ltd. Helsinki 2000 Environmental properties of chemicals Volume 2 _____ _____________________________________________________ Contents . VOLUME ONE 1 Contents of the report 2 Environmental properties of chemicals 3 Abbreviations and explanations 7 3.1 Ways of exposure 7 3.2 Exposed species 7 3.3 Fffects________________________________ 7 3.4 Length of exposure 7 3.5 Odour thresholds 8 3.6 Toxicity endpoints 9 3.7 Other abbreviations 9 4 Listofexposedspecies 10 4.1 Mammais 10 4.2 Plants 13 4.3 Birds 14 4.4 Insects 17 4.5 Fishes 1$ 4.6 Mollusca 22 4.7 Crustaceans 23 4.8 Algae 24 4.9 Others 25 5 References 27 Index 1 List of chemicals in alphabetical order - 169 Index II List of chemicals in CAS-number order -

Determination of Age-Related Differences in Activation and Detoxication of Organophosphates in Rat and Human Tissues
Mississippi State University Scholars Junction Theses and Dissertations Theses and Dissertations 8-10-2018 Determination of Age-Related Differences in Activation and Detoxication of Organophosphates in Rat and Human Tissues Edward Caldwell Meek Follow this and additional works at: https://scholarsjunction.msstate.edu/td Recommended Citation Meek, Edward Caldwell, "Determination of Age-Related Differences in Activation and Detoxication of Organophosphates in Rat and Human Tissues" (2018). Theses and Dissertations. 1339. https://scholarsjunction.msstate.edu/td/1339 This Dissertation - Open Access is brought to you for free and open access by the Theses and Dissertations at Scholars Junction. It has been accepted for inclusion in Theses and Dissertations by an authorized administrator of Scholars Junction. For more information, please contact [email protected]. Template A v3.0 (beta): Created by J. Nail 06/2015 Determination of age-related differences in activation and detoxication of organophosphates in rat and human tissues By TITLE PAGE Edward Caldwell Meek A Dissertation Submitted to the Faculty of Mississippi State University in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy in Environmental Toxicology in the College of Veterinary Medicine Mississippi State, Mississippi August 2018 Copyright by COPYRIGHT PAGE Edward Caldwell Meek 2018 Determination of age-related differences in activation and detoxication of organophosphates in rat and human tissues By APPROVAL PAGE Edward Caldwell Meek -

Development and Validation of a Method for the Simultaneous
CORE Metadata, citation and similar papers at core.ac.uk Provided by UGD Academic Repository American Journal of Applie d Chemistry 2014; 2(4): 46-54 Published online August 10, 2014 (http://www.sciencepublishinggroup.com/j/ajac) doi: 10.11648/j.ajac.20140204.11 ISSN: 2330-8753 (Print); ISSN: 2330-8745 (Online) Development and validation of a method for the simultaneous determination of 20 organophosphorus pesticide residues in corn by accelerated solvent extraction and gas chromatography with nitrogen phosphorus detection Vesna Kostik *, Biljana Gjorgeska, Bistra Angelovska Medical Faculty, Department of Pharmacy, University “Goce Delchev”, Shtip, Republic of Macedonia Email address: [email protected] (V. Kostik), [email protected] (B. Gjorgeska), [email protected] (B. Angelovska) To cite this article: Vesna Kostik, Biljana Gjorgeska, Bistra Angelovska. Development and Validation of a Method for the Simultaneous Determination of 20 Organophosphorus Pesticide Residues in Corn by Accelerated Solvent Extraction and Gas Chromatography with Nitrogen Phosphorus Detection. American Journal of Applied Chemistry. Vol. 2, No. 4, 2014, pp. 46-54. doi: 10.11648/j.ajac.20140204.11 Abstract: The method for simultaneous determination of 20 organophosphorus pesticide residues in corn samples has been developed and validated. For the extraction of organophosporus pesticide residues from the samples, the accelerated solvent technique with the mixture of dichloromethane: acetone (1:1, V/V ) was used. Clean up was done using liquid – liquid extraction with n – hexane, followed by solid phase extraction on primary secondary amine adsorbent, and elution with the mixture of acetone: toluene (65:35). The determination of the pesticides was carried out by gas chromatography with nitrogen phosphorus detection. -

The Effect of Processing on 14 C-Chlofenvinphos Residues In
EG0800342 The Effect of Processing on 14C- Chlorfenvinphos Residues in Maize Oil and Bioavailability of its Cake Residues on Rats F. Mahdy, S. El-Maghraby National Research Centre, Dokki, Giza, Egypt ABSTRACT Maize seed obtained from 14C-chlorfenvinphos treated plants contained 0.12 % of the applied dose. The insecticide residues in crude oil, methanol and coke amounted to 10 %, 6 % and 69 %, respectively of original residues inside the seeds.The 14C-activity in the crude oil could be a gradual reduced by the refining processes. The alkali treatment and bleaching steps are more effective steps in the refining processes remove about (63 %). The refined oil contained only about 17 % of the 14C-residues originally present. The major residues in processed oil contain parent compound, in addition to five metabolites of the insecticide. When rats fed the extracted seeds (cake), the bound residues were found to be considerably bioavailable. After feeding rats for 5 days with the cake, a substantial amount of 14C-residues was eliminated in the urine (59.5 %), while about 20 % was excreted in the feces. About 15 % of the radioactivity was distribution among various organs. Keywords: 14C-chlorfenvinphos, maize oil, refining processes, residues, bioavailability. INTRODUCTION Pesticides are of interest as residues because of their widespread use in a variety of crops for field and post-harvest protection and because with their degradation products, they could be a potential health hazard. Past investigations suggest that most organochlorine and organophosphorus pesticide in edible oils can be reduced considerably by a chemical refining process, but pyrethroid pesticides remain to a certain extent (1-6). -

Development of a Heterologous Enzyme-Linked Immunosorbent Assay for Organophosphorus Pesticides with Phage-Borne Peptide
HHS Public Access Author manuscript Author Manuscript Author ManuscriptRSC Adv Author Manuscript. Author manuscript; Author Manuscript available in PMC 2015 August 17. Published in final edited form as: RSC Adv. 2014 January 1; 4(80): 42445–42453. doi:10.1039/C4RA07059C. Development of a heterologous enzyme-linked immunosorbent assay for organophosphorus pesticides with phage-borne peptide Xiude Huaa,b, Xiaofeng Liua,b, Haiyan Shia,b, Yanru Wangc, Hee Joo Kimc, Shirley J. Geec, Minghua Wanga,b,*, Fengquan Liud,*, and Bruce D. Hammockc aCollege of Plant Protection (State & Local Joint Engineering Research Center of Green Pesticide Invention and Application), Nanjing Agricultural University, Nanjing 210095, China bKey Laboratory of Integrated Management of Crop Diseases and Pests (Nanjing Agricultural University), Ministry of Education cDepartment of Entomology and UCD Cancer Center, University of California, Davis, California 95616, United States dInstitute of Plant Protection, Jiangsu Academy of Agricultural Science, Nanjing 210014, China Abstract An enzyme-linked immunosorbent assay (ELISA) was developed to detect organophosphorus pesticides using a phage-borne peptide that was isolated from a cyclic 8-residue peptide phage −1 library. The IC50 values of the phage ELISA ranged from 1.4 to 92.1 μg L for eight organophosphorus pesticides (parathion-methyl, parathion, fenitrothion, cyanophos, EPN, paraoxon-methyl, paraoxon, fenitrooxon). The sensitivity was improved 120- and 2-fold compared to conventional homologous and heterologous ELISA, respectively. The selectivity of the phage ELISA was evaluated by measuring its cross-reactivity with 23 organophosphorus pesticides, among which eight were the main cross-reactants. The spike recoveries were between 66.1% and 101.6% for the detection of single pesticide residues of parathion-methyl, parathion and fenitrothion in Chinese cabbage, apple and greengrocery, and all of the coefficient of variation were less than or equal to 15.9%. -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

Lifetime Organophosphorous Insecticide Use Among Private Pesticide Applicators in the Agricultural Health Study
Journal of Exposure Science and Environmental Epidemiology (2012) 22, 584 -- 592 & 2012 Nature America, Inc. All rights reserved 1559-0631/12 www.nature.com/jes ORIGINAL ARTICLE Lifetime organophosphorous insecticide use among private pesticide applicators in the Agricultural Health Study Jane A. Hoppin1, Stuart Long2, David M. Umbach3, Jay H. Lubin4, Sarah E. Starks5, Fred Gerr5, Kent Thomas6, Cynthia J. Hines7, Scott Weichenthal8, Freya Kamel1, Stella Koutros9, Michael Alavanja9, Laura E. Beane Freeman9 and Dale P. Sandler1 Organophosphorous insecticides (OPs) are the most commonly used insecticides in US agriculture, but little information is available regarding specific OP use by individual farmers. We describe OP use for licensed private pesticide applicators from Iowa and North Carolina in the Agricultural Health Study (AHS) using lifetime pesticide use data from 701 randomly selected male participants collected at three time periods. Of 27 OPs studied, 20 were used by 41%. Overall, 95% had ever applied at least one OP. The median number of different OPs used was 4 (maximum ¼ 13). Malathion was the most commonly used OP (74%) followed by chlorpyrifos (54%). OP use declined over time. At the first interview (1993--1997), 68% of participants had applied OPs in the past year; by the last interview (2005--2007), only 42% had. Similarly, median annual application days of OPs declined from 13.5 to 6 days. Although OP use was common, the specific OPs used varied by state, time period, and individual. Much of the variability in OP use was associated with the choice of OP, rather than the frequency or duration of application. -

Managing Insect and Mite Pests of Texas Corn Patrick Porter Associate Professor and Extension Entomologist Gregory B
E-400 5/06 Managing Insect and Mite Pests of Texas Corn Patrick Porter Associate Professor and Extension Entomologist Gregory B. Cronholm Extension Agent—Pest Management Roy D. Parker Professor and Extension Entomologist; Noel Troxclair Assistant Professor and Extension Entomologist Carl D. Patrick Professor and Extension Entomologist Stephen P. Biles Extension Agent—Pest Management and Pat Morrison Associate Department Head (Retired) This publication may be used in conjunction with B-6177,Texas Corn Production Emphasizing Pest Management and Irrigation (2005), which is available from Texas Cooperative Extension (http://tcebookstore.org/). B-6177 provides details on each corn pest and discusses scouting and economic thresholds. Also, there are instructional videos and pest identification guides at http://lubbock.tamu.edu/cornIPM/. Contents Introduction . 4 Policy Statement for Making Pest Management Suggestions 4 . Endangered Species Regulations . 4 Worker Protection Standard . 5 Biological Control . 5 Transgenic Corn . 5 Preemergence Insect Control . 7 Soil-Inhabiting Pests . 7 Seed Treatment . 7 Soil Treatment . 7 White Grubs and Cutworms . 8 Wireworms, Seedcorn Maggots and Seedcorn Beetles . 8 Mexican and Western Corn Rootworm . 9 Southern Corn Rootworm . 10 Seedling to Tassel Stage Insect Control . 11 Corn Leaf Aphid . 11 Soil Cutworms . 11 Southwestern Corn Borer . 11 European Corn Borer . 12 Lesser Cornstalk Borer . 12 Other Borers . 12 Corn Earworm and Fall Armyworm . 13 Flea Beetles . 13 Chinch Bug . 14 Tassel to Hard Dough Stage Insect Control . 15 Corn Earworm . 15 Fall Armyworm . 15 Southwestern Corn Borer . 15 European Corn Borer . 16 Spider Mites . 17 Adult Rootworm Beetle . 19 True Armyworm . 20 Western Bean Cutworm . 20 Grasshoppers . 21 Sap Beetles . -
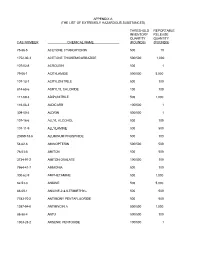
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

Pesticide Toxicity Index for Freshwater Aquatic Organisms, 2Nd Edition
Pesticide Toxicity Index for Freshwater Aquatic Organisms, 2nd Edition By Mark D. Munn, Robert J. Gilliom, Patrick W. Moran, and Lisa H. Nowell National Water-Quality Assessment Program Scientific Investigations Report 2006-5148 U.S. Department of the Interior U.S. Geological Survey U.S. Department of the Interior Dirk Kempthorne, Secretary U.S. Geological Survey P. Patrick Leahy, Acting Director U.S. Geological Survey, Reston, Virginia: 2006 Revised and reprinted: 2006 For sale by U.S. Geological Survey, Information Services Box 25286, Denver Federal Center Denver, CO 80225 For more information about the USGS and its products: Telephone: 1-888-ASK-USGS World Wide Web: http://www.usgs.gov/ Any use of trade, product, or firm names in this publication is for descriptive purposes only and does not imply endorsement by the U.S. Government. Although this report is in the public domain, permission must be secured from the individual copyright owners to reproduce any copyrighted materials contained within this report. Suggested reference: Munn, M.D., Gilliom, R.J., Moran, P.W., and Nowell, L.H., 2006, Pesticide toxicity index for freshwater aquatic organisms, 2nd Edition: U.S. Geological Survey Scientific Investigations Report 2006-5148, 81 p. iii FOREWORD The U.S. Geological Survey (USGS) is committed to providing the Nation with accurate and timely scien- tific information that helps enhance and protect the overall quality of life and that facilitates effective management of water, biological, energy, and mineral resources (http://www.usgs.gov/). Information on the quality of the Nation’s water resources is critical to assuring the long-term availability of water that is safe for drinking and recreation and suitable for industry, irrigation, and habitat for fish and wildlife.