Isomer Overview the Following Table Summarizes the Various Types Of
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Introduction to Aromaticity
Introduction to Aromaticity Historical Timeline:1 Spotlight on Benzene:2 th • Early 19 century chemists derive benzene formula (C6H6) and molecular mass (78). • Carbon to hydrogen ratio of 1:1 suggests high reactivity and instability. • However, benzene is fairly inert and fails to undergo reactions that characterize normal alkenes. - Benzene remains inert at room temperature. - Benzene is more resistant to catalytic hydrogenation than other alkenes. Possible (but wrong) benzene structures:3 Dewar benzene Prismane Fulvene 2,4- Hexadiyne - Rearranges to benzene at - Rearranges to - Undergoes catalytic - Undergoes catalytic room temperature. Faraday’s benzene. hydrogenation easily. hydrogenation easily - Lots of ring strain. - Lots of ring strain. - Lots of ring strain. 1 Timeline is computer-generated, compiled with information from pg. 594 of Bruice, Organic Chemistry, 4th Edition, Ch. 15.2, and from Chemistry 14C Thinkbook by Dr. Steven Hardinger, Version 4, p. 26 2 Chemistry 14C Thinkbook, p. 26 3 Images of Dewar benzene, prismane, fulvene, and 2,4-Hexadiyne taken from Chemistry 14C Thinkbook, p. 26. Kekulé’s solution: - “snake bites its own tail” (4) Problems with Kekulé’s solution: • If Kekulé’s structure were to have two chloride substituents replacing two hydrogen atoms, there should be a pair of 1,2-dichlorobenzene isomers: one isomer with single bonds separating the Cl atoms, and another with double bonds separating the Cl atoms. • These isomers were never isolated or detected. • Rapid equilibrium proposed, where isomers interconvert so quickly that they cannot be isolated or detected. • Regardless, Kekulé’s structure has C=C’s and normal alkene reactions are still expected. - But the unusual stability of benzene still unexplained. -

II. Stereochemistry 5
B.Sc.(H) Chemistry Semester - II Core Course - III (CC-III) Organic Chemistry - I II. Stereochemistry 5. Physical and Chemical Properties of Stereoisomers Dr. Rajeev Ranjan University Department of Chemistry Dr. Shyama Prasad Mukherjee University, Ranchi 1 Syllabus & Coverage Syllabus II Stereochemistry: Fischer Projection, Newmann and Sawhorse Projection formulae and their interconversions. Geometrical isomerism: cis–trans and syn-anti isomerism, E/Z notations with Cahn Ingold and Prelog (CIP) rules for determining absolute configuration. Optical Isomerism: Optical Activity, Specific Rotation, Chirality/Asymmetry, Enantiomers, Molecules with two or more chiral-centres, Distereoisomers, Meso structures, Racemic mixture. Resolution of Racemic mixtures. Relative and absolute configuration: D/L and R/S designations. Coverage: 1. Types of Isomers : Comparing Structures 2. Optical Activity 3. Racemic Mixtures : Separation of Racemic Mixtures 4. Enantiomeric Excess and Optical Purity 5. Relative and Absolute Configuration 6. Physical and Chemical Properties of Stereoisomers 2 Stereochemistry Types of Isomers Dr. Rajeev Ranjan 3 Stereochemistry Determining the Relationship Between Two Non-Identical Molecules Dr. Rajeev Ranjan 4 Stereochemistry Comparing Structures: Are the structures connected the same? yes no Are they mirror images? Constitutional Isomers yes no Enantiomers Enantiomers Is there a plane of symmetry? All chiral centers will be opposite between them. yes no Meso Diastereomers superimposable Dr. Rajeev Ranjan 5 Stereochemistry Optical Activity: • The chemical and physical properties of two enantiomers are identical except in their interaction with chiral substances. • The physical property that differs is the behavior when subjected to plane-polarized light ( this physical property is often called an optical property). • Plane-polarized (polarized) light is light that has an electric vector that oscillates in a single plane. -

Isomer Distributions of Molecular Weight 247 and 273 Nitro-Pahs in Ambient Samples, NIST Diesel SRM, and from Radical-Initiated Chamber Reactions
Atmospheric Environment 55 (2012) 431e439 Contents lists available at SciVerse ScienceDirect Atmospheric Environment journal homepage: www.elsevier.com/locate/atmosenv Isomer distributions of molecular weight 247 and 273 nitro-PAHs in ambient samples, NIST diesel SRM, and from radical-initiated chamber reactions Kathryn Zimmermann a,1, Roger Atkinson a,1,2,3, Janet Arey a,1,2,*, Yuki Kojima b,4, Koji Inazu b,5 a Air Pollution Research Center, University of California, Riverside, CA 92521, USA b Tokyo Institute of Technology, 4259 Nagatsuta, Midori-ku, Yokohama 226-8502, Japan article info abstract Article history: Molecular weight (mw) 247 nitrofluoranthenes and nitropyrenes and mw 273 nitrotriphenylenes (NTPs), Received 27 December 2011 nitrobenz[a]anthracenes, and nitrochrysenes were quantified in ambient particles collected in Riverside, Received in revised form CA, Tokyo, Japan, and Mexico City, Mexico. 2-Nitrofluoranthene (2-NFL) was the most abundant nitro- 28 February 2012 polycyclic aromatic hydrocarbon (nitro-PAH) in Riverside and Mexico City, and the mw 273 nitro-PAHs Accepted 5 March 2012 were observed in lower concentrations. However, in Tokyo concentrations of 1- þ 2-NTP were more similar to that of 2-NFL. NIST SRM 1975 diesel extract standard reference material was also analyzed to Keywords: examine nitro-PAH isomer distributions, and 12-nitrobenz[a]anthracene was identified for the first time. Nitro-PAH fl Atmospheric reactions The atmospheric formation pathways of nitro-PAHs were studied from chamber reactions of uo- Ambient particles ranthene, pyrene, triphenylene, benz[a]anthracene, and chrysene with OH and NO3 radicals at room Nitrotriphenylenes temperature and atmospheric pressure, with the PAH concentrations being controlled by their vapor pressures. -

Questions & Answers for the New Chemicals Program
Note: Effective January 19, 2016, PMNs must be submitted electronically. Learn more about the new e-PMN requirements. Questions & Answers for the New Chemicals Program (Q&A) U.S. Environmental Protection Agency Office of Pollution Prevention and Toxics Washington, DC 20460 2004 -1- TABLE OF CONTENTS Page 1. GENERAL PROGRAM INFORMATION 100. General ............................................................................................................ 1-1 101. Guidance for Completion of §5 Submission Form ......................................... 1-6 102. Inventory Searches/Bona Fides ....................................................................... 1-17 103. Chemical Identification ................................................................................... 1-22 104. Nomenclature .................................................................................................. 1-26 105. Inventory Issues ................................................................................................ 1-31 106. Review Process ............................................................................................... 1-31 107. Notice of Commencement .............................................................................. 1-33 108. User Fee .......................................................................................................... 1-35 109. Consolidated Notices ...................................................................................... 1-39 110. Joint Submissions .......................................................................................... -
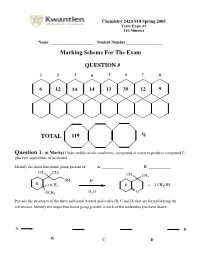
Marking Scheme for the Exam
Chemistry 2420 S10 Spring 2005 Term Exam #1 110 Minutes Name: ______________________ Student Number___________________ Marking Scheme For The Exam QUESTION # 1 2 3 4 5 6 7 8 6 12 14 14 13 39 12 9 TOTAL 119 % Question 1. (6 Marks) Under mildly acidic conditions, compound A reacts to produce compound E plus two equivalents of methanol. Identify the main functional group present in: A: ___________ E: ___________ CH3 CH3 CH 3 CH3 + NH2 H A OCH3 E + 2 CH3OH H O OCH3 2 N Provide the structures of the three additional neutral molecules (B, C and D) that are formed during the conversion. Identify the major functional group present in each of the molecules you have drawn. A E B C D Question 2. (12 Marks) Circle the structure that will best satisfy the given information. - the compound able to undergo an intramolecular cyclization to form a hemiacetal H H HO H HO OH O O O - the compound able to undergo an intramolecular cyclization to form an enamine H H H Me2N MeHN H2N O O O + - the produc t formed from treatment of 2-butanone with NaBD4 followed by a H3O work-up DO H HO D DO D HO H - the compound that would most likely be coloured O H O O - the compound capable of undergoing mutarotation O O O OH OCH3 OH CH3 H CH 3 - the compound that would not react with MeMgBr O O O OH OCH3 OH CH3 H CH3 - the compound bes t described as “anti-aromatic”: - the most stable carbocation + + + Cl Cl Cl OCH 3 H NO2 H H H Question 3. -

Introduction to Organic Chemistry 2018 More
Introduction to Organic Chemistry 25 Introduction to Organic Chemistry Handout 2 - Stereochemistry OH O O OH enantiomers Me OH HO Me A B NH2 NH2 diastereomers diastereomers diastereomers OH O O OH Me OH HO Me C D NH2 enantiomers NH2 http://burton.chem.ox.ac.uk/teaching.html ◼ Organic Chemistry J. Clayden, N. Greeves, S. Warren ◼ Stereochemistry at a Glance J. Eames & J. M. Peach ◼ The majority of organic chemistry text books have good chapters on the topics covered by these lectures ◼ Eliel Stereochemistry of Organic Compounds (advanced reference text) Introduction to Organic Chemistry 26 ◼ representations of formulae in organic chemistry ◼ skeletal representations are far less cluttered and as a result are much clearer than drawing all carbon and hydrogen atoms explicitly, they also give a much better representation of the likely bond angles and hence hybridisation states of the carbon atoms ◼ skeletal representations allow functional groups (sites of reactivity) to be clearly seen ◼ guidelines for drawing skeletal structures i) draw chains of atoms as zig-zags ii) do not draw C atoms unless there is good reason to draw them iii) do not draw C-H bonds unless there is good reason to draw then iv) do not draw Hs attached to carbon atoms unless there is good reason to draw them v) make drawings realistic Introduction to Organic Chemistry 27 ◼ representing structures in three dimensions ◼ a wedged bond indicates the bond is projecting out in front of the plane of the paper ◼ a dashed bond indicates the bond is projecting behind the plane -

Electrophilic Mercuration and Thallation of Benzene and Substituted Benzenes in Trifluoroacetic Acid Solution* (Electrophilic Substitution/Selectivity) GEORGE A
Proc. Natl. Acad. Sci. USA Vol. 74, No. 10, pp. 4121-4125, October 1977 Chemistry Electrophilic mercuration and thallation of benzene and substituted benzenes in trifluoroacetic acid solution* (electrophilic substitution/selectivity) GEORGE A. OLAH, IWAO HASHIMOTOt, AND HENRY C. LINtt Institute of Hydrocarbon Chemistry, Department of Chemistry, University of Southern California, Los Angeles, California 90007; and the Department of Chemistry, Case Western Reserve University, Cleveland, Ohio 44101 Contributed by George A. Olah, July 18, 1977 ABSTRACT The mercuration and thallation of benzene and iments at 250 and quenched the mixtures after 5 min, whereas substituted benzenes was studied with mercuric and thallic Brown and Nelson carried out the reaction for 6.5 hr. In sub- trifluoroacetate, respectively, in trifluoroacetic acid. With the in view of the importance of both the mechanistic shortest reaction time (1 sec) at 00, the relative rate of mercu- sequent work, ration of toluene compared to that of benzene was 17.5, with and practical implications of the orientation-rate correlation, the isomer distribution in toluene of: ortho, 17.4%; meta, 5.9%; Brown and McGary (7), carried out a more detailed study of and para, 76.7%. The isomer distribution in toluene varied with the mercuration reaction. They concluded: "A redetermination the reaction time, significantly more at 25° than at 00. The of the isomer distributions and relative rates indicates excellent competitive thallation of benzene and toluene with thallic tri- agreement with the linear relationship of orientation and rel- fluoroacetate in trifluoroacetic acid at 150 showed the relative ative however, that the isomer distri- rate, toluene/benzene, to be 33, with the isomer distribution in rate." They recognized, toluene of: ortho, 9.5%; meta, 5.5%; and para, 85.0%. -

Cis-Trans Isomerism 2
Isomerism Isomerism • These compounds possess the same molecular formula but differ from each other in physical or chemical properties, and are called Isomers and the phenomenon is termed Isomerism • Since isomers have the same molecular formula, the difference in their properties must be due to different modes of combination or arrangement of atoms within the molecule. There are two main types of isomerism • (i) Structural Isomerism and (ii) Stereoisomerism. Structural Isomerism • When the isomerism is simply due to difference in the arrangement of atoms within the molecule without any reference to space, the phenomenon is termed Structural Isomerism. In other words, the structural isomers are compounds that have the same molecular formula but different structural formulas. Types of Structural Isomerism 1. Chain Isomerism 2. Position Isomerism 3. Functional Isomerism 4. Metamersim 5. Tautomerism Stereoisomerism • When isomerism is caused by the different arrangements of atoms or groups in space, the phenomenon is called Stereoisomerism (Greek, Stereos = occupying space). • The stereoisomers have the same structural formulas but differ in arrangement of atoms in space. In other words, stereoisomerism is exhibited by such compounds which have identical molecular structure but different configurations. Configuration refers to the 3-dimentional arrangement of atoms that characterizes a particular compound. Types of Stereoisomerism Stereoisomerism is of two types 1. Geometrical or Cis-Trans Isomerism 2. Optical Isomerism Structural Isomerism Chain Isomerism This type of isomerism arises from she difference in the structure of carbon chain which forms the nucleus of the molecule. It is, therefore, named as Chain or Nuclear Isomerism. Chain isomers have the same molecular formula but differ in the order in which the carbon atoms are bonded to each other For example, there are known two butanes which have the same molecular formula (C4H10) but differ in the structure of the carbon chains in their molecules. -

REFERENCE ONLY UNIVERSITY of LONDON THESIS This Thesis
REFERENCE ONLY UNIVERSITY OF LONDON THESIS Degree Year Name of Author 0" * COPYRIGHT This is a thesis accepted for a Higher Degree of the University of London. It is an unpublished typescript and the copyright is held by the author. All persons consulting the thesis must read and abide by the Copyright Declaration below. COPYRIGHT DECLARATION I recognise that the copyright of the above-described thesis rests with the author and that no quotation from it or information derived from it may be published without the prior written consent of the author. LOANS Theses may not be lent to individuals, but the Senate House Library may lend a copy to approved libraries within the United Kingdom, for consultation solely on the premises of those libraries. Application should be made to: Inter-Library Loans, Senate House Library, Senate House, Malet Street, London WC1E 7HU. REPRODUCTION University of London theses may not be reproduced without explicit written permission from the Senate House Library. Enquiries should be addressed to the Theses Section of the Library. Regulations concerning reproduction vary according to the date of acceptance of the thesis and are listed below as guidelines. A. Before 1962. Permission granted only upon the prior written consent of the author. (The Senate House Library will provide addresses where possible). B. 1962- 1974. In many cases the author has agreed to permit copying upon completion of a Copyright Declaration. C. 1975 - 1988. Most theses may be copied upon completion of a Copyright Declaration. D. 1989 onwards. Most theses may be copied. This thesis comes within category D. -

Enantiomeric Resolution and Absolute Configuration of a Chiral Δ-Lactam
molecules Article Enantiomeric Resolution and Absolute Configuration of a Chiral δ-Lactam, Useful Intermediate for the Synthesis of Bioactive Compounds 1, 1, 1 1 Roberta Listro y, Giacomo Rossino y, Serena Della Volpe , Rita Stabile , Massimo Boiocchi 2 , Lorenzo Malavasi 3, Daniela Rossi 1,* and Simona Collina 1 1 Department of Drug Sciences, University of Pavia, via Taramelli 12, 27100 Pavia, Italy; [email protected] (R.L.); [email protected] (G.R.); [email protected] (S.D.V.); [email protected] (R.S.); [email protected] (S.C.) 2 Centro Grandi Strumenti, University of Pavia, via Bassi 21, 27100 Pavia, Italy; [email protected] 3 Department of Chemistry, University of Pavia, via Taramelli 12, 27100 Pavia, Italy; [email protected] * Correspondence: [email protected] These authors contributed equally to this work. y Academic Editor: Józef Drabowicz Received: 2 December 2020; Accepted: 18 December 2020; Published: 19 December 2020 Abstract: During the past several years, the frequency of discovery of new molecular entities based on γ- or δ-lactam scaffolds has increased continuously. Most of them are characterized by the presence of at least one chiral center. Herein, we present the preparation, isolation and the absolute configuration assignment of enantiomeric 2-(4-bromophenyl)-1-isobutyl-6-oxopiperidin-3-carboxylic acid (trans-1). For the preparation of racemic trans-1, the Castagnoli-Cushman reaction was employed. (Semi)-preparative enantioselective HPLC allowed to obtain enantiomerically pure trans-1 whose absolute configuration was assigned by X-ray diffractometry. Compound (+)-(2R,3R)-1 represents a reference compound for the configurational study of structurally related lactams. -

Part 1 in Our Series of Carbohydrate Lectures. in This Section, You Will Learn About Monosaccharide Structure
Welcome to Part 1 in our series of Carbohydrate lectures. In this section, you will learn about monosaccharide structure. The building blocks of larger carbohydrate polymers. 1 First, let’s review why learning about carbohydrates is important. Carbohydrates are used by biological systems as fuels and energy resources. Carbohydrates typically provide quick energy and are one of the primary energy storage forms in animals. Carbohydrates also provide the precursors to other major macromolecules within the body, including the deoxyribose and ribose required for nucleic acid biosynthesis. Carbohydrates can also provide structural support and cushioning/shock absorption, as well as cell‐cell communication, identification, and signaling. 2 Carbohydrates, as their name implies, are water hydrates of carbon, and they all have the same basic core formula (CH2O)n and are always found in the ratio of 1 carbon to 2 hydrogens to 1 oxygen (1:2:1) making them easy to identify from their molecular formula. 3 Carbohydrates can be divided into subcategories based on their complexity. The simplest carbohydrates are the monosaccharides which are the simple sugars required for the biosynthesis of all the other carbohydrate types. Disaccharides consist of two monosaccharides that have been joined together by a covalent bond called the glycosidic bond. Oligosaccharides are polymers that consist of a few monosaccharides covalently linked together, and Polysaccharides are large polymers that contain hundreds to thousands of monosaccharide units all joined together by glycosidic bonds. The remainder of this lecture will focus on monosaccharides 4 Monosaccharides all have alcohol functional groups associated with them. In addition they also have one additional functional group, either an aldehyde or a ketone. -

Lipids, Carbohydrates, Nucleobases & DNA
Chemistry 2050 “Introduction to Organic Chemistry” Fall Semester 2011 Dr. Rainer Glaser Examination #5: The Final “Lipids, Carbohydrates, Nucleobases & DNA.” Monday, December 12, 2011, 10 am – 12 pm. Name: Answer Key Question 1. Lipids and Detergents. 20 Question 2. Glyceraldehyde and Carbohydrates. 20 Question 3. Nucleobases of DNA and RNA. 20 Question 4. Ribose and Deoxyribose and Their Phosphate Esters. 20 Question 5. Single and Double Strands of DNA. 20 Total 100 — 1 — Question 1. Lipids and Detergents. (20 points) (a) Draw the line segment drawing of glyceryl distearoleate. This fat is the _TRI-ester formed between one molecule of the triol __GLYCEROL_, two molecules of the fatty acid stearic acid, H3C−(CH2)16−COOH, and one molecule of the unsaturated fatty acid oleic acid, H3C−(CH2)7−CH=CH−(CH2)7−COOH. In your drawing, attach the oleic acid to one of the primary alcohols of the triol. Clearly indicate whether the alkene in oleic acid is cis or trans. To convert this fat into soap, one would cook the fat with aqueous NaOH solution, a process that is called __SAPONIFICATION__. (12 points) (b) A simple micelle is shown schematically on the right. Each circle signifies a _________ (polar, nonpolar) head-group, and each wiggly line signifies a ________ (polar, nonpolar) alkyl chain. In the case of “normal” soap, the head- group is a ___CARBOXYLATE__ anion. Indicate where we would find the associated cations (i.e., sodium cations; draw a few in the scheme). Water is on the __________ (inside, outside) of this micelle and fats will accumulate on the _________ (inside, outside) of this micelle.