Hemoglobin Subunit Alpha Antibody
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

HBA2 Antibody Cat
HBA2 Antibody Cat. No.: 23-575 HBA2 Antibody Immunohistochemistry of paraffin-embedded mouse Immunohistochemistry of paraffin-embedded mouse heart using HBA2 antibody (23-575) at dilution of 1:100 brain using HBA2 antibody (23-575) at dilution of 1:100 (40x lens). (40x lens). Specifications HOST SPECIES: Rabbit SPECIES REACTIVITY: Mouse, Rat Recombinant fusion protein containing a sequence corresponding to amino acids 1-142 of IMMUNOGEN: human HBA2 (NP_000508.1). TESTED APPLICATIONS: IHC, WB WB: ,1:500 - 1:2000 APPLICATIONS: IHC: ,1:50 - 1:100 POSITIVE CONTROL: 1) Mouse liver September 30, 2021 1 https://www.prosci-inc.com/hba2-antibody-23-575.html PREDICTED MOLECULAR Observed: 13kDa WEIGHT: Properties PURIFICATION: Affinity purification CLONALITY: Polyclonal ISOTYPE: IgG CONJUGATE: Unconjugated PHYSICAL STATE: Liquid BUFFER: PBS with 0.02% sodium azide, 50% glycerol, pH7.3. STORAGE CONDITIONS: Store at -20˚C. Avoid freeze / thaw cycles. Additional Info OFFICIAL SYMBOL: HBA2 ALTERNATE NAMES: Hemoglobin subunit alpha, Alpha-globinHBH GENE ID: 3040 USER NOTE: Optimal dilutions for each application to be determined by the researcher. Background and References The human alpha globin gene cluster located on chromosome 16 spans about 30 kb and includes seven loci: 5'- zeta - pseudozeta - mu - pseudoalpha-1 - alpha-2 - alpha-1 - theta - 3'. The alpha-2 (HBA2) and alpha-1 (HBA1) coding sequences are identical. These genes differ slightly over the 5' untranslated regions and the introns, but they differ significantly over the 3' untranslated regions. Two alpha chains plus two beta chains constitute HbA, BACKGROUND: which in normal adult life comprises about 97% of the total hemoglobin; alpha chains combine with delta chains to constitute HbA-2, which with HbF (fetal hemoglobin) makes up the remaining 3% of adult hemoglobin. -

Additional File 3.Pdf
************ globins ************ >Pdu_Egb_A1a MNGITVFLILAMASASLADDCTQLDMIKVKHQWAEVYGVESNRQEFGLAVFKRFFVIHPD RSLFVNVHGDNVYSPEFQAHVARVLAGVDILISSMDQEAIFKAAQKHYADFHKSKFGEVPLVEFGTAMRDVLP KYVGLRNYDNDSWSRCYAYITSKVE >Pdu_Egb_A1b MKGLLVFLVLASVSASLASECSSLDKIKVKNQWA RIHGSPSNRKAFGTAVFKRFFEDHPDRSLFANVNGNDIYSADFQAHVQRVFGGLDILIVSLDQDDLFTAAKSH YSEFHKKLGDVPFAEFGVAFLDTLSDFLPLRDYNQDPWSRCYNYIIS >Pdu_Egb_A1c MNTVTVVLVLLG CIASAMTGDCNTLQRTKVKYQWSIVYGATDNRQAFGTLVWRDFFGLYPDRSLFSGVRGENIYSPEFRAHVVRV FAGFDILISLLDQEDILNSALAHYAAFHKQFPSIPFKEFGVVLLEALAKTIPEQFDQDAWSQCYAVIVAGVTA >Pdu_Egb_A1d_alpha MYQILSVAVLVLSCLALGTLGEEVCGPLERIKVQHQWVSVYGADHDRLKVSTL VWKDFFEHHPEERARFERVNSDNIFSGDFRAHMVRVFAGFDLLIGVLNEEEIFKSAMIHYTKMHNDLGVTTEI IKEFGKSIARVLPEFMDGKPDITAWRPCFNLIAAGVSE >Pdu_Egb_A1d_beta MYFSYFTAAASYLSVAVLVLSCLVQGILGEEVCGPLEKIKVQHQWASAYRGDHD RLKMSTLVWKDFFAHNPEERARFERVHSDDIYSGDFRAHMVRVFAGFDLLIGALNQEDIFRSAMIHYTKMHKK LGVTYEIGIEFGKSIGRVLPEFIDGKLDITAWRPCYKLIATGVDE >Pdu_Egb_A1d_gamma MYLSVAVLVLSCLALGTQGEEVCGPLEKIKVQHQWASAYRGDHDRLKMSTLVW KDFFAHHPEERARFERVHSDDIYSGDFRAHMVRVFAGFDLLIGVLNQDEIFKSAMIHYTKMHNDLGVKTEIVL EFGKSIARVLPDFIDGKPDITAWRPCFKLIAAGVSE >Pdu_Egb_A2 MNNLVILVGLLCLGLTSATKCGPL QRLKVKQQWAKAYGVGHERLELGIALWKSIFAQDPESRSIFTRVHGDDVRHPAFEAHIARVFNGFDRIISSLT DEDVLQAQLAHLKAQHIKLGISAHHFKLMRTGLSYVLPAQLGRCFDKEAWGSCWDEVIYPGIKSL >Pdu_Egb_B1 MLVLAVFVAALGLAAADQCCSIEDRNEVQALWQSIWSAENTGKRTIIGHQIFEELFDINP GTKDLFKRVNVEDTSSPEFEAHVLRVMNGLDTLIGVLDDPATGYSLITHLAEQHKAREGFKPSYFKDIGVALK RVLPQVASCFNPEAWDHCFNGFVEAITNKMNAL >Pdu_Egb_B2 MLVLVLSLAFLGSALAEDCCSAADRKTVLRDWQSVWSAEFTGRRVAIGTAIFEELFAIDA GAKDVFKNVAVDKPESAEWAAHVIRVINGLDLAINLLEDPRALKEELLHLAKQHRERDGVKAVYFDEIGRALL -

Anti-Hemoglobin Antibody (FITC) Product Number: AC15-0147-12
Anti-Hemoglobin Antibody (FITC) Product Number: AC15-0147-12 Overview Host Species: Goat Clonality: Polyclonal Purity: Hemoglobin Antibody (FITC) is affinity purified. The affinity purified antibody is then conjugated to the fluorescent dye FITC (fluorescein isothiocyanate). Conjugate: FITC Immunogen Type: Anti-hemoglobin antibody (FITC) was rasied against human hemoglobin. Physical Characteristics Amount: 0.05 mg Concentration: 1 mg/ml Volume: 0.05 ml Buffer: 10 mM Sodium Phosphate, 0.15 M NaCl, pH 7.2 with 0.05% (w/v) sodium azide. Storage: FITC conjugated antibody can be stored at 4?C for up to 18 months. For longer storage the conjugate can be stored at -20?C after adding 50% glycerol. Fluorescein conjugated antibodies should always be stored in the dark. Specificity Species Reactivity: Human Specificity: Human hemoglobin Target Information Gene ID Number(s): 3047 (human), 3039 (human), 3040 (human) Alternative Names: 3-prime alpha-globin gene; Alpha globin; alpha one globin; alpha-1 globin; Alpha-globin; Beta globin; CD113t C; CD31antibody (FITC); Erythremia, beta-globin type, included; Gamma 1 globin; Hb FAgamma; HBA 1; HBA 2; HBA; HBA_HUMAN; HBA1antibody (FITC); HBA2; HBB; Hbb-y; HBD; Hbe1; HBG 1antibody (FITC); HBG; HBG1; HBGA; HBGR; HBHantibody (FITC); Hemoglobin alpha 1; hemoglobin alpha 1 globin chain; Hemoglobinalpha chain; Hemoglobin alpha locus; Hemoglobin alpha locus 1antibody (FITC); hemoglobin alpha-1 chain; Hemoglobin beta; Hemoglobin beta chainantibody (FITC); Hemoglobin beta chain complex; Hemoglobin beta locus; Hemoglobingamma -
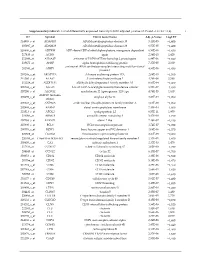
Supplementary Table S1. List of Differentially Expressed
Supplementary table S1. List of differentially expressed transcripts (FDR adjusted p‐value < 0.05 and −1.4 ≤ FC ≥1.4). 1 ID Symbol Entrez Gene Name Adj. p‐Value Log2 FC 214895_s_at ADAM10 ADAM metallopeptidase domain 10 3,11E‐05 −1,400 205997_at ADAM28 ADAM metallopeptidase domain 28 6,57E‐05 −1,400 220606_s_at ADPRM ADP‐ribose/CDP‐alcohol diphosphatase, manganese dependent 6,50E‐06 −1,430 217410_at AGRN agrin 2,34E‐10 1,420 212980_at AHSA2P activator of HSP90 ATPase homolog 2, pseudogene 6,44E‐06 −1,920 219672_at AHSP alpha hemoglobin stabilizing protein 7,27E‐05 2,330 aminoacyl tRNA synthetase complex interacting multifunctional 202541_at AIMP1 4,91E‐06 −1,830 protein 1 210269_s_at AKAP17A A‐kinase anchoring protein 17A 2,64E‐10 −1,560 211560_s_at ALAS2 5ʹ‐aminolevulinate synthase 2 4,28E‐06 3,560 212224_at ALDH1A1 aldehyde dehydrogenase 1 family member A1 8,93E‐04 −1,400 205583_s_at ALG13 ALG13 UDP‐N‐acetylglucosaminyltransferase subunit 9,50E‐07 −1,430 207206_s_at ALOX12 arachidonate 12‐lipoxygenase, 12S type 4,76E‐05 1,630 AMY1C (includes 208498_s_at amylase alpha 1C 3,83E‐05 −1,700 others) 201043_s_at ANP32A acidic nuclear phosphoprotein 32 family member A 5,61E‐09 −1,760 202888_s_at ANPEP alanyl aminopeptidase, membrane 7,40E‐04 −1,600 221013_s_at APOL2 apolipoprotein L2 6,57E‐11 1,600 219094_at ARMC8 armadillo repeat containing 8 3,47E‐08 −1,710 207798_s_at ATXN2L ataxin 2 like 2,16E‐07 −1,410 215990_s_at BCL6 BCL6 transcription repressor 1,74E‐07 −1,700 200776_s_at BZW1 basic leucine zipper and W2 domains 1 1,09E‐06 −1,570 222309_at -

Research Article Association Between HBA Locus Copy Number Gains And
INTERNATIONAL JOURNAL OF MEDICAL BIOCHEMISTRY DOI: 10.14744/ijmb.2021.65477 Int J Med Biochem 2021;4(2):91-6 Research Article Association between HBA locus copy number gains and pathogenic HBB gene variants Guven Toksoy1, Nergis Akay2, Agharza Aghayev1, Volkan Karaman1, Sahin Avci1, Tugba Kalayci1, Umut Altunoglu1, Zeynep Karakas2, Zehra Oya Uyguner1 1Department of Medical Genetics, Istanbul University Istanbul Faculty of Medicine, Istanbul, Turkey 2Department of Pediatric Hematology-Oncology, Istanbul University Istanbul Faculty of Medicine, Istanbul, Turkey Abstract Objectives: Alpha (α) and beta (β) thalassemia are the most prevalent genetic hematological disorders. The co-occur- rence of silent β-thalassemia with excess α-globin gene copies is associated with the thalassemia intermedia pheno- type. This study was an investigation of the α-globulin gene dosage and sequence variations in thalassemia patients. Methods: Multiplex ligation-dependent probe amplification and Sanger sequencing were used to identify the hemo- globin subunit alpha 1 (HBA1) and HBA2 gene alterations in 32 patients. Deletion, duplication, and other findings were analyzed in the index cases and family members. Results: Four of the 32 cases (12.5%) were found to have gross duplications. Two cases demonstrated α-globin triplica- tion, and 2 had a quadruplicated HBA1/2 genes. Affected family members revealed genotype-phenotype correlation. In 1 patient, it was observed that quadruplicated HBA genes co-occurrence with hemoglobin subunit beta (HBB) mu- tation was inherited from his mother. Notably, the mother did not demonstrate any thalassemia phenotype. Further investigation showed that the mother was carrying a single copy HBA gene deletion in the trans allele that explained her clinical condition. -

For Molecular Biosystems. This Journal Is © the Royal Society of Chemistry 2014
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is © The Royal Society of Chemistry 2014 Table 6: Identified proteins in exosomes from activated VSMCs Paragon Biological Score %Cov(95) Accession Entry Name Name Peptides(95%) Processes Cellular localizatio SecretomeP Exocarta 1,48 6,627 P10668 COF1_PIG Cofilin-1 2 CA Cyt/Nucleus/Membr Sec nc 1,48 6,627 Q5G6V9 COF2_PIG Cofilin-2 1 CA Cyt/Nucleus Sec nc Exo 2 2,256 Q9GLP0 ITB1_PIG Integrin beta-1 1 CA Membr NP Exo 4,01 3,743 A5A8V7 HS71L_PIG Heat shock 70 kDa protein 1-like 1 CH Cyt NP Exo 4,03 3,683 O02705 HS90A_PIG Heat shock protein HSP 90-alpha 5 CH Cyt NP Exo 2,04 5,928 P34930 HS71A_PIG Heat shock 70 kDa protein 1A 1 CH Cyt NP Exo 8 43,9 P62936 PPIA_PIG Peptidyl-prolyl cis-trans isomerase A 5 CH Sec NP Exo 2 8,212 Q5S1U1 HSPB1_PIG Heat shock protein beta-1 1 CH Cyt/Nucleus Sec nc Exo 4,06 7,955 Q6S4N2 HS71B_PIG Heat shock 70 kDa protein 1B 6 CH Cyt NP Exo Signal transducer and activator of 2 1,039 Q19S50 STAT3_PIG transcription 3 1 CS Cyt/Nucleus Sec nc 2,29 6,222 P02543 VIME_PIG Vimentin 1 Cyt Org Cyt Sec nc Exo 4,21 7,981 P02550 TBA1A_PIG Tubulin alpha-1A chain 8 Cyt Org Cyt NP Exo 6,14 22,02 P02554 TBB_PIG Tubulin beta chain 1 Cyt Org Cyt Sec nc Exo 2 3,497 Q29221 CAZA2_PIG F-actin-capping protein subunit alpha-2 1 Cyt Org Cyt Sec nc 6,26 10,85 Q2XVP4 TBA1B_PIG Tubulin alpha-1B chain 9 Cyt Org Cyt NP Exo 16,06 23,99 Q6QAQ1 ACTB_PIG Actin, cytoplasmic 1 19 Cyt Org Cyt NP Exo 7,01 18,69 Q767L7 TBB5_PIG Tubulin beta chain 11 Cyt Org Cyt Sec nc Exo Inter-alpha-trypsin -

Identification of MAZ As a Novel Transcription Factor Regulating Erythropoiesis
Identification of MAZ as a novel transcription factor regulating erythropoiesis Darya Deen1, Falk Butter2, Michelle L. Holland3, Vasiliki Samara4, Jacqueline A. Sloane-Stanley4, Helena Ayyub4, Matthias Mann5, David Garrick4,6,7 and Douglas Vernimmen1,7 1 The Roslin Institute and Royal (Dick) School of Veterinary Studies, University of Edinburgh, Easter Bush, Midlothian EH25 9RG, United Kingdom. 2 Institute of Molecular Biology (IMB), 55128 Mainz, Germany 3 Department of Medical and Molecular Genetics, School of Basic and Medical Biosciences, King's College London, London SE1 9RT, UK 4 MRC Molecular Haematology Unit, Weatherall Institute for Molecular Medicine, University of Oxford, John Radcliffe Hospital, Oxford OX3 9DS, United Kingdom. 5 Department of Proteomics and Signal Transduction, Max Planck Institute of Biochemistry, 82152 Martinsried, Germany. 6 Current address : INSERM UMRS-976, Institut de Recherche Saint Louis, Université de Paris, 75010 Paris, France. 7 These authors contributed equally Correspondence: [email protected], [email protected] ADDITIONAL MATERIAL Suppl Fig 1. Erythroid-specific hypersensitivity regions DNAse-Seq. (A) Structure of the α-globin locus. Chromosomal position (hg38 genome build) is shown above the genes. The locus consists of the embryonic ζ gene (HBZ), the duplicated foetal/adult α genes (HBA2 and HBA1) together with two flanking pseudogenes (HBM and HBQ1). The upstream, widely-expressed gene, NPRL3 is transcribed from the opposite strand to that of the HBA genes and contains the remote regulatory elements of the α-globin locus (MCS -R1 to -R3) (indicated by red dots). Below is shown the ATAC-seq signal in human erythroblasts (taken from 8) indicating open chromatin structure at promoter and distal regulatory elements (shaded regions). -

Usbiological Datasheet
HBA1 (HBA2, Hemoglobin Subunit alpha, Alpha-globin, Hemoglobin alpha Chain, MGC126895, MGC126897) Catalog number 127733 Supplier United States Biological HBA2 located on chromosome 16 spans about 30kb and includes seven loci: 5'- zeta - pseudozeta - mu - pseudoalpha-1 - alpha-2 - alpha-1 - theta - 3'. The alpha-2 (HBA2) and alpha-1 (HBA1) coding sequences are identical. These genes differ slightly over the 5' untranslated regions and the introns, but they differ significantly over the 3' untranslated regions. Two alpha chains plus two beta chains constitute HbA, which in normal adult life comprises about 97% of the total hemoglobin; alpha chains combine with delta chains to constitute HbA-2, which with HbF (fetal hemoglobin) makes up the remaining 3% of adult hemoglobin. Alpha thalassemias result from deletions of each of the alpha genes as well as deletions of both HBA2 and HBA1; some nondeletion alpha thalassemias have also been reported. Applications Suitable for use in ELISA and Western Blot. Other applications not tested. Recommended Dilution Optimal dilutions to be determined by the researcher. AA Sequence MFLSFPTTKTYFPHFDLSHGSAQVKGHGKKVADALTNAVAHVDDMPNALSALSDLHAHKLRVDPVNFKLLSHCLLV TLAAHLPAEFTPAVHASLDKFLASVSTVLTSKYR Storage and Stability May be stored at 4°C for short-term only. Aliquot to avoid repeated freezing and thawing. Store at -20°C. Aliquots are stable for at least 12 months. For maximum recovery of product, centrifuge the original vial after thawing and prior to removing the cap. Immunogen Partial recombinant corresponding to aa33-142 from human HBA1 (NP_000549) with GST tag. MW of the GST tag alone is 26kD. Formulation Supplied as a liquid in PBS, pH 7.2. Purity Purified by Protein A affinity chromatography. -

Anti-Hemoglobin Alpha Antibody (ARG58926)
Product datasheet [email protected] ARG58926 Package: 100 μl anti-hemoglobin alpha antibody Store at: -20°C Summary Product Description Rabbit Polyclonal antibody recognizes hemoglobin alpha Tested Reactivity Hu, Ms Predict Reactivity Mk Tested Application FACS, IHC-P, WB Host Rabbit Clonality Polyclonal Isotype IgG Target Name hemoglobin alpha Antigen Species Human Immunogen KLH-conjugated synthetic peptide corresponding to aa. 100-128 of Human hemoglobin alpha. Conjugation Un-conjugated Alternate Names HBA-T3; Alpha-globin; HBH; Hemoglobin subunit alpha; Hemoglobin alpha chain Application Instructions Application table Application Dilution FACS 1:25 IHC-P 1:50 - 1:100 WB 1:1000 Application Note * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. Positive Control Human fetal heart Calculated Mw 15 kDa Properties Form Liquid Purification Purification with Protein A and immunogen peptide. Buffer PBS and 0.09% (W/V) Sodium azide. Preservative 0.09% (W/V) Sodium azide. Storage instruction For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C or below. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. www.arigobio.com 1/3 Note For laboratory research only, not for drug, diagnostic or other use. Bioinformation Gene Symbol HBA1 Gene Full Name hemoglobin, alpha 1 Background The human alpha globin gene cluster located on chromosome 16 spans about 30 kb and includes seven loci: 5'- zeta - pseudozeta - mu - pseudoalpha-1 - alpha-2 - alpha-1 - theta - 3'. -

Novel Protein Pathways in Development and Progression of Pulmonary Sarcoidosis Maneesh Bhargava1*, K
www.nature.com/scientificreports OPEN Novel protein pathways in development and progression of pulmonary sarcoidosis Maneesh Bhargava1*, K. J. Viken1, B. Barkes2, T. J. Grifn3, M. Gillespie2, P. D. Jagtap3, R. Sajulga3, E. J. Peterson4, H. E. Dincer1, L. Li2, C. I. Restrepo2, B. P. O’Connor5, T. E. Fingerlin5, D. M. Perlman1 & L. A. Maier2 Pulmonary involvement occurs in up to 95% of sarcoidosis cases. In this pilot study, we examine lung compartment-specifc protein expression to identify pathways linked to development and progression of pulmonary sarcoidosis. We characterized bronchoalveolar lavage (BAL) cells and fuid (BALF) proteins in recently diagnosed sarcoidosis cases. We identifed 4,306 proteins in BAL cells, of which 272 proteins were diferentially expressed in sarcoidosis compared to controls. These proteins map to novel pathways such as integrin-linked kinase and IL-8 signaling and previously implicated pathways in sarcoidosis, including phagosome maturation, clathrin-mediated endocytic signaling and redox balance. In the BALF, the diferentially expressed proteins map to several pathways identifed in the BAL cells. The diferentially expressed BALF proteins also map to aryl hydrocarbon signaling, communication between innate and adaptive immune response, integrin, PTEN and phospholipase C signaling, serotonin and tryptophan metabolism, autophagy, and B cell receptor signaling. Additional pathways that were diferent between progressive and non-progressive sarcoidosis in the BALF included CD28 signaling and PFKFB4 signaling. Our studies demonstrate the power of contemporary proteomics to reveal novel mechanisms operational in sarcoidosis. Application of our workfows in well-phenotyped large cohorts maybe benefcial to identify biomarkers for diagnosis and prognosis and therapeutically tenable molecular mechanisms. -

The Free Alpha-Hemoglobin: a Promising Biomarker for Β-Thalassemia
r Biomar ula ke c rs le o & M D f i a o g l Journal of Molecular Biomarkers n a o n Abdullah, et al., J Mol Biomark Diagn 2014, 5:5 r s i u s o J DOI: 10.4172/2155-9929.1000197 ISSN: 2155-9929 & Diagnosis Review Article Open Access The Free Alpha-Hemoglobin: A Promising Biomarker for β-Thalassemia Uday YH Abdullah1, Ahmed GF Al-Attraqchi1, Hishamshah M Ibrahim2, Zilfalil Bin Alwi3, Atif A Baig1, Iekhsan Othman4, Noraesah B Mahmud2, Rosline B Hassan3, Nor Hedayah A Bakar1 and Alawiyah B A Abd Rahman5 1University Sultan Zainal Abidin (UniSZA), Faculty of Medicine and Health Sciences, Kuala Terengganu, Malaysia 2Hospital Kuala Lumpur (HKL), Kuala Lumpur, Malaysia 3Univesiti Sains Malaysia (USM), School of Medical Science, Kelantan, Malaysia 4Monash University, Selangor, Malaysia 5Hospital Sultana Nor Zahirah (HSNZ), Kuala Terengganu, Malaysia *Corresponding author: Uday Younis Hussein Abdullah, University Sultan Zainal Abidin (UniSZA), Kampus Kota, Jalan Sultan Mahmud 20400, Kuala Terengganu, Malaysia, Tel: 609-625607; Fax: 60-6275771/5772; E-mail: [email protected] Rec date: Aug 28, 2014; Acc date: Sep 27, 2014; Pub date: Sep 29, 2014 Copyright: © 2014 Abdullah UYH, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Abstract The purpose is to review the current knowledge and methods of determination of the free alpha hemoglobin with emphasis on its potential implications in patients with beta-thalassemia (β-thal).The severity of β-thal correlates with the extent of imbalance between α- and non-α-globin chains and the amount of the free alpha hemoglobin (free α- Hb) pool in the erythrocytes. -

The Amniotic Fluid Proteome Differs Significantly Between Donor And
J Korean Med Sci. 2020 Mar 16;35(10):e73 https://doi.org/10.3346/jkms.2020.35.e73 eISSN 1598-6357·pISSN 1011-8934 Original Article The Amniotic Fluid Proteome Differs Obstetrics & Gynecology Significantly between Donor and Recipient Fetuses in Pregnancies Complicated by Twin-to-Twin Transfusion Syndrome Sun Min Kim ,1,2* Byoung-Kyu Cho ,3* Byoung Jae Kim ,1,2 Ha Yun Lee ,3 Errol R. Norwitz ,4 Min Jueng Kang ,3 Seung Mi Lee ,1 Chan-Wook Park ,1 Jong Kwan Jun ,1 Eugene C. Yi ,3 and Joong Shin Park 1 Received: Apr 7, 2019 1Department of Obstetrics & Gynecology, Seoul National University Hospital, Seoul National University Accepted: Jan 20, 2020 College of Medicine, Seoul, Korea 2Department of Obstetrics & Gynecology, Seoul Metropolitan Government Seoul National University Address for Correspondence: Boramae Medical Center, Seoul, Korea Joong Shin Park, MD, PhD 3Department of Molecular Medicine and Biopharmaceutical Sciences, Graduate School of Convergence Department of Obstetrics & Gynecology, Seoul Science and Technology and College of Medicine or College of Pharmacy, Seoul National University, Seoul, National University Hospital, Seoul National Korea University College of Medicine, 101 Daehak-ro, 4Department of Obstetrics & Gynecology, Tufts University School of Medicine, Boston, MA, USA Jongno-gu, Seoul 03080, Korea. E-mail: [email protected] Eugene C. Yi, PhD ABSTRACT Department of Molecular Medicine and Biopharmaceutical Sciences, Graduate School Background: Twin-to-twin transfusion syndrome (TTTS) is a serious complication of of Convergence Science and Technology and monochorionic twin pregnancies. It results from disproportionate blood supply to each College of Medicine or College of Pharmacy, fetus caused by abnormal vascular anastomosis within the placenta.