Supplementary Figure 1. Tables of GC-MS Data for Each of the 82 Essential Oils Reported Herein
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Retention Indices for Frequently Reported Compounds of Plant Essential Oils
Retention Indices for Frequently Reported Compounds of Plant Essential Oils V. I. Babushok,a) P. J. Linstrom, and I. G. Zenkevichb) National Institute of Standards and Technology, Gaithersburg, Maryland 20899, USA (Received 1 August 2011; accepted 27 September 2011; published online 29 November 2011) Gas chromatographic retention indices were evaluated for 505 frequently reported plant essential oil components using a large retention index database. Retention data are presented for three types of commonly used stationary phases: dimethyl silicone (nonpolar), dimethyl sili- cone with 5% phenyl groups (slightly polar), and polyethylene glycol (polar) stationary phases. The evaluations are based on the treatment of multiple measurements with the number of data records ranging from about 5 to 800 per compound. Data analysis was limited to temperature programmed conditions. The data reported include the average and median values of retention index with standard deviations and confidence intervals. VC 2011 by the U.S. Secretary of Commerce on behalf of the United States. All rights reserved. [doi:10.1063/1.3653552] Key words: essential oils; gas chromatography; Kova´ts indices; linear indices; retention indices; identification; flavor; olfaction. CONTENTS 1. Introduction The practical applications of plant essential oils are very 1. Introduction................................ 1 diverse. They are used for the production of food, drugs, per- fumes, aromatherapy, and many other applications.1–4 The 2. Retention Indices ........................... 2 need for identification of essential oil components ranges 3. Retention Data Presentation and Discussion . 2 from product quality control to basic research. The identifi- 4. Summary.................................. 45 cation of unknown compounds remains a complex problem, in spite of great progress made in analytical techniques over 5. -

Effect of Enzymes on Strawberry Volatiles During Storage, at Different Ripeness
Effect of Enzymes on Strawberry Volatiles During Storage, at Different Ripeness Level, in Different Cultivars and During Eating Thesis Presented in Partial Fulfillment of the Requirements for the Degree Master of Science in the Graduate School of The Ohio State University By Gulsah Ozcan Graduate Program in Food Science and Technology The Ohio State University 2010 Thesis Committee: Sheryl Ann Barringer, Adviser W. James Harper John Litchfield 1 Copyright by Gülşah Özcan 2010 ii ABSTRACT Strawberry samples with enzyme activity and without enzyme activity (stannous chloride added) were measured for real time formation of lipoxygenase (LOX) derived aroma compounds after 5 min pureeing using selected ion flow tube mass spectrometry (SIFT-MS). The concentration of (Z)-3-hexenal and (E)-2-hexenal increased immediately after blending and gradually decreased over time while hexanal concentration increased for at least 5 min in ground strawberries. The formation of hexanal was slower than the formation of (Z)-3-hexenal and (E)-2-hexenal in the headspace of pureed strawberries. The concentration of LOX aldehydes and esters significantly increased during refrigerated storage. Damaging strawberries increased the concentration of LOX aldehydes but did not significantly affect the concentration of esters. The concentrations of many of the esters were strongly correlated to their corresponded acids and/or aldehydes. The concentration of LOX generated aldehydes decreased during ripening, while fruity esters increased. Different varieties had different aroma profiles and esters were the greatest percentage of the volatiles. The aroma release of some of the LOX derived aldehydes in the mouthspace in whole strawberries compared to chopped strawberries showed that these volatiles are formed in the mouth during chewing. -

The Composition of Strawberry Aroma Is Influenced by Cultivar, Maturity, and Storage Charles F
WORKSHOP The Composition of Strawberry Aroma Is Influenced by Cultivar, Maturity, and Storage Charles F. Forney1, Willy Kalt2, and Michael A. Jordan3 Agriculture and Agri-Food Canada, Atlantic Food and Horticulture Research Centre, 32 Main Street, Kentville, N.S., B4N 1J5, Canada Strawberry (Fragaria ×ananassa Duch.) fruit have a unique, they both may contribute to strawberry aroma (Dirinck et al., 1981; highly desirable flavor and are one of the most popular summer fruits. Schreier, 1980). Sugars, acids, and aroma volatiles contribute to the characteristic The volatile profile obtained from strawberry fruit is influenced by strawberry flavor, which is dependent on the proper balance of these the analytical methods used to characterize these compounds. Volatiles chemical constituents. While sugars and acids are responsible for the from whole, intact fruit can be collected using headspace techniques; sweetness and tartness of the fruit, aroma volatiles provide the unique, these samples can be analyzed directly or concentrated using adsor- fruity flavors that characterize a fresh strawberry. bent or cold traps. Volatiles are also collected from homogenized fruit The aroma of fresh strawberries is dependent on many factors. The or juice, using either headspace or solvent extraction techniques. large genetic variability in the nature of strawberry aroma results in Volatile samples are normally analyzed by gas liquid chromatography differences in flavor among cultivars. In addition, the aroma changes using a variety of methods of sample introduction, including liquid dramatically during fruit ripening after harvest; therefore, it is impor- injection, thermal desorption, and cold on-column injection. High tant to preserve and enhance the ripe fruit aroma during postharvest performance liquid chromatography (HPLC) has been used for some handling. -

Differences in Volatile Ester Composition Between Fragaria Ananassa and F
Scientia Horticulturae 150 (2013) 47–53 Contents lists available at SciVerse ScienceDirect Scientia Horticulturae journa l homepage: www.elsevier.com/locate/scihorti × Differences in volatile ester composition between Fragaria ananassa and F. ଝ vesca and implications for strawberry aroma patterns a,b b c b a,∗ Jing Dong , Yuntao Zhang , Xiaowei Tang , Wanmei Jin , Zhenhai Han a Institute for Horticultural Plants, College of Agronomy and Biotechnology, China Agricultural University, No. 2 Yuanmingyuan West Road, Haidian District, Beijing 100193, PR China b Institute of Forestry and Pomology, Beijing Academy of Agriculture and Forestry Sciences, No. 12 Ruiwangfen, Xiangshan, Haidian District, Beijing 100093, PR China c Beijing Vegetable Research Center, Beijing Academy of Agriculture and Forestry Sciences, Banjin, Haidian District, Beijing 100097, PR China a r t i c l e i n f o a b s t r a c t Article history: Esters are a very important component of strawberry (Fragaria sp.) aroma. In this study, fruit volatiles Received 8 April 2012 were isolated by solid-phase microextraction and analyzed by gas chromatography–mass spectrometry Received in revised form 1 November 2012 (SPME/GC–MS). It was found that F. × ananassa had more esters than F. vesca, but there was no significant Accepted 1 November 2012 difference in average relative content of esters. Twenty five esters were selected as predominant esters, 16 of which were prevalent in F. × ananassa and another 16 in F. vesca. Among them, hexyl acetate, Keywords: octyl acetate, ethyl butyrate, ethyl hexanoate, ethyl octanoate, methyl decanoate, and ethyl decanoate Volatile ester composition were found in both F. -

Expanding the Modular Ester Fermentative Pathways for Combinatorial Biosynthesis of Esters from Volatile Organic Acids
ARTICLE Expanding the Modular Ester Fermentative Pathways for Combinatorial Biosynthesis of Esters From Volatile Organic Acids Donovan S. Layton,1,2 Cong T. Trinh1,2,3 1 Department of Chemical and Biomolecular Engineering, University of Tennessee, Knoxville, Tennessee 2 BioEnergy Science Center (BESC), Oak Ridge National Laboratory, Oak Ridge, Tennessee 3 Bredesen Center for Interdisciplinary Research and Graduate Education, University of Tennessee, Knoxville, Tennessee; telephone: þ865-974-8121; fax: 865-974-7076; e-mail: [email protected] Biotechnol. Bioeng. 2016;113: 1764–1776. ABSTRACT: Volatile organic acids are byproducts of fermentative ß 2016 Wiley Periodicals, Inc. metabolism, for example, anaerobic digestion of lignocellulosic KEYWORDS: modular chassis cell; carboxylate; ester; acyl acetate; biomass or organic wastes, and are often times undesired inhibiting acyl acylate; ester fermentative pathway cell growth and reducing directed formation of the desired products. Here, we devised a general framework for upgrading these volatile organic acids to high-value esters that can be used as flavors, fragrances, solvents, and biofuels. This framework employs the acid-to-ester modules, consisting of an AAT (alcohol Introduction acyltransferase) plus ACT (acyl CoA transferase) submodule and an alcohol submodule, for co-fermentation of sugars and organic Harnessing renewable or waste feedstocks (e.g., switchgrass, corn acids to acyl CoAs and alcohols to form a combinatorial library of stover, agricultural residue, or municipal solid waste) -

Comprehensive Mapping of Volatile Organic Compounds in Fruits
International PhD Program in Biomolecular Sciences XXVII Cycle Comprehensive Mapping of Volatile Organic Compounds in Fruits Tutor Dr. Fulvio Mattivi Department of Food Quality and Nutrition, Fondazione Edmund Mach Advisor Prof. Vladimir Shulaev Department of Biological Sciences, University of North Texas Ph.D. Thesis of Manoj Shahaji Ghaste Department of Food Quality and Nutrition Fondazione Edmund Mach 2013-2014 This thesis is lovingly dedicated to my Mother. Her support, encouragement, belief and constant love have sustained me throughout my life. Declaration I, Manoj Shahaji Ghaste confirm that this is my own work and the use of all material from other sources has been properly and fully acknowledged. Thesis abstract Volatile organic compounds (VOCs) are the key aroma producers in fruits and sensory quality of fruits is widely determined by qualitative and quantitative composition of VOCs. The aroma of grape is a complex of hundreds of VOCs belonging to different chemical classes like alcohols, esters, acids, terpenes, aldehydes, furanones, pyrazines, isoprenoids and many more. VOCs play important role as they determine the flavor of grapes and wine made from it. The objective of this thesis is to study of VOCs through development of different mass spectrometry based analytical methodologies and its applications for the comprehensive investigation and construction of database of the VOCs in grapes. First part of the study was dedicated to generation of a database of grape VOCs through the screening of multiple grape varieties (n=124) representing different species, color and origin. The experiment was carried out using headspace solid-phase microextraction (HS-SPME) and gas chromatography mass spectrometry (GC-MS) based approach and according to metabolomics protocols. -

Effect of Thickeners and Sweeteners on the Release of Blended Aroma
Effect of thickeners and sweeteners on the release of blended aroma compounds in fat-free stirred yoghurt during shear conditions Nicolas Decourcelle, Samuel Lubbers, Nadine Vallet, Pascale Rondeau, Elisabeth Guichard To cite this version: Nicolas Decourcelle, Samuel Lubbers, Nadine Vallet, Pascale Rondeau, Elisabeth Guichard. Effect of thickeners and sweeteners on the release of blended aroma compounds in fat-free stirred yo- ghurt during shear conditions. International Dairy Journal, Elsevier, 2004, 14 (9), pp.783 - 789. 10.1016/j.idairyj.2004.02.007. hal-01413574 HAL Id: hal-01413574 https://hal.archives-ouvertes.fr/hal-01413574 Submitted on 9 Dec 2016 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. ARTICLE IN PRESS International Dairy Journal 14 (2004) 783–789 Effect of thickeners and sweeteners on the release of blended aroma compounds in fat-free stirred yoghurt during shear conditions Nicolas Decourcellea, Samuel Lubbersa,*, Nadine Valletb, Pascale Rondeaub, Elisabeth Guicharda a Unite Mixte de Recharche sur les Aromes, ENESAD-INRA, 17, rue de Sully Dijon Cedex F-21065, France b UMA Danone Vitapole, RD128, Palaiseau Cedex 92767, France Received 3 July 2003; accepted 18 February 2004 Abstract The present study was conducted on aromatised fat-free stirred yoghurts and dealt with the influence of some thickeners and sweeteners on aroma compounds release and rheology. -

Stability of Volatile Profile and Sensory Properties of Passion Fruit Juice During Storage in Glass Bottles
Ciência e Tecnologia de Alimentos ISSN 0101-2061 Stability of volatile profile and sensory properties of passion fruit juice during storage in glass bottles Original Estabilidade do perfil de voláteis e propriedades sensoriais de suco de maracujá durante estocagem em garrafas de vidro Vitória Matos de FREitas1, Deborah dos Santos GARRUTI2*, Manoel Alves SOUZA NETO2, Heliofábia Virgínia de Vasconcelos FacUNDO1, José Maria CORREia1 Abstract This study aimed at assessing the stability of passion fruit juice in glass bottles during a 120-day storage period, regarding its volatile compounds profile and sensory properties (aroma and flavor). Samples were obtained from a Brazilian tropical juice industry (Fortaleza, Brazil) and submitted to sensory and chromatographic analyses. The characteristic aroma and flavor of passion fruit were evaluated by a trained panel with a non-structured scale of 9 cm. The headspace volatile compounds were isolated from the product by suction and trapped in Porapak Q, analyzed through high-resolution gas chromatography and identified through gas chromatography-mass spectrometry (GC-MS). Twelve odoriferous compounds were monitored: ethyl butanoate, ethyl propanoate, 3-methyl-1-butanol, 3-methyl-2-butenol, (E)-3-hexenol, (Z)- 3-hexenol, 3-methylbutyl acetate, benzaldehyde, ethyl hexanoate, hexyl acetate, limonene and furfural. The slight variations observed in the volatile profile were not enough to provoke significant changes in the characteristic aroma and flavor of the passion fruit juice. Keywords: Passiflora; tropical juice; packaging material; volatile compounds; sensory analysis; storage. Resumo O objetivo deste estudo foi avaliar a estabilidade do suco de maracujá em garrafas de vidro durante 120 dias de estocagem, em relação ao perfil de compostos voláteis e características sensoriais (aroma e sabor). -

Identification of Volatile Compounds Associated with the Aroma of White
Research Article Received: 11 December 2012 Revised: 12 September 2013 Accepted article published: 20 September 2013 Published online in Wiley Online Library: 21 October 2013 (wileyonlinelibrary.com) DOI 10.1002/jsfa.6412 Identification of volatile compounds associated with the aroma of white strawberries (Fragaria chiloensis) Loreto Prat,a Mar´ıa Ines´ Espinoza,b Eduardo Agosinb,c and Herman Silvaa∗ Abstract BACKGROUND: Fragaria chiloensis (L.) Mill spp. chiloensis form chiloensis, is a strawberry that produces white fruits with unique aromas. This species, endemic to Chile, is one of the progenitors of Fragaria x ananassa Duch. In order to identify the volatile compounds that might be responsible for aroma, these were extracted, and analyzed by gas chromatography–mass spectrometry (GC-MS), gas chromatography–olfactometry (GC-O) and compared with sensory analyses. RESULTS: Three methods of extraction were used: solvent-assisted evaporation (SAFE), headspace solid phase micro-extraction (HS-SPME) and liquid–liquid extraction (LLE). Ninety-nine volatile compounds were identified by GC-MS, of which 75 showed odor activity using GC-O. Based on the highest dilution factor (FD = 1000) and GC-O intensity ≥2, we determined 20 major compounds in white strawberry fruit that contribute to its aroma. We chose 51 compounds to be tested against their commercial standards. The identities were confirmed by comparison of their linear retention indices against the commercial standards. The aroma of white strawberry fruits was reconstituted with a synthetic mixture of most of these compounds. CONCLUSION: The volatile profile of white strawberry fruit described as fruity, green–fresh, floral, caramel, sweet, nutty and woody will be a useful reference for future strawberry breeding programs. -

Differential Analysis of Proteins Involved in Ester Metabolism in Two
microorganisms Article Differential Analysis of Proteins Involved in Ester Metabolism in two Saccharomyces cerevisiae Strains during the Second Fermentation in Sparkling Wine Elaboration Maria del Carmen González-Jiménez 1, Jaime Moreno-García 1 , Teresa García-Martínez 1,* , Juan José Moreno 2 , Anna Puig-Pujol 3, Fina Capdevilla 3 and Juan Carlos Mauricio 1 1 Department of Microbiology, University of Cordoba, 14014 Cordoba, Spain; [email protected] (M.d.C.G.-J.); [email protected] (J.M.-G.); [email protected] (J.C.M.) 2 Department of Agricultural Chemistry, University of Cordoba, 14014 Cordoba, Spain; [email protected] 3 Department of Enological Research, Institute of Agrifood Research and Technology-Catalan Institute of Vine and wine (IRTA-INCAVI), 08720 Barcelona, Spain; [email protected] (A.P.-P.); [email protected] (F.C.) * Correspondence: [email protected]; Tel.: +34 957218640; Fax: +34 957218650 Received: 29 January 2020; Accepted: 11 March 2020; Published: 13 March 2020 Abstract: The aromatic metabolites derived from yeast metabolism determine the characteristics of aroma and taste in wines, so they are considered of great industrial interest. Volatile esters represent the most important group and therefore, their presence is extremely important for the flavor profile of the wine. In this work, we use and compare two Saccharomyces cerevisiae yeast strains: P29, typical of sparkling wines resulting of second fermentation in a closed bottle; G1, a flor yeast responsible for the biological aging of Sherry wines. We aimed to analyze and compare the effect of endogenous CO2 overpressure on esters metabolism with the proteins related in these yeast strains, to understand the yeast fermentation process in sparkling wines. -
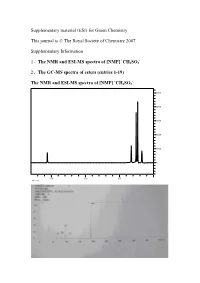
And the GC-MS
Supplementary material (ESI) for Green Chemistry This journal is © The Royal Society of Chemistry 2007 Supplementary Information + - 1、The NMR and ESI-MS spectra of [NMP] CH3SO3 2、The GC-MS spectra of esters (entries 1-19) + - The NMR and ESI-MS spectra of [NMP] CH3SO3 5000 4000 3000 2000 1000 0 15.0 10.0 5.0 ppm (t1) The GC-MS spectra of esters (entries 1-19) (1) The GC-MS spectrum of propyl acetate 0.69min, solvent of chloroform; 0.97min, propyl acetate The MS spectra of propyl acetate (0.97min) (2) The GC-MS spectrum of isopropyl acetate 1.98min, isopropyl acetate The MS spectrum of isopropyl acetate (1.98min) (3) The GC-MS spectrum of butyl acetate 2.90min, butyl acetate 4(ZHANG) #665 RT: 3.61 AV: 1 NL: 2.56E7 T: + c Full ms [ 14.00-400.00] 72.85 100 90 80 70 85.85 41.88 60 50 40 Relative Abundance 57.88 30 20 100.93 10 135.97154.12 207.58 253.45 280.90 355.76 0 50 100 150 200 250 300 350 400 m/z The MS spectrum of butyl acetate (2.90min) (4) The GC-MS spectrum of isopentyl acetate 2.20min, isopentyl acetate 3(ZHANG) #405 RT: 2.98 AV: 1 NL: 2.71E7 T: + c Full ms [ 14.00-400.00] 86.89 100 90 80 70 60 41.86 50 40 85.90 Relative Abundance Relative 57.85 30 20 69.89 10 87.91 88.94 121.16 158.00 207.11 226.90 280.98 314.33 356.16 0 50 100 150 200 250 300 350 400 m/z The MS spectrum of isopentyl acetate (2.2min) (5) The GC-MS spectrum of pentyl acetate 3.54min, pentyl acetate The MS spectrum of pentyl acetate (3.54min) (6) The GC-MS spectrum of hexyl acetate 4.15min, hexyl acetate 1 #920 RT: 3.08 AV: 1 NL: 5.24E7 T: + c Full ms [ 45.00-400.00] -

(12) United States Patent (10) Patent No.: US 9.260,817 B2 Williams Et Al
US0092.60817B2 (12) United States Patent (10) Patent No.: US 9.260,817 B2 Williams et al. (45) Date of Patent: *Feb. 16, 2016 (54) FRESHENING COMPOSITIONS A61L 9/14: D06M 13/005; D06M 15/61; COMPRISING MALODOR BINDING D06M 13/192: D06M 13/203: D06M 13/127; POLYMERS AND MALODOR D06M 13/148: D06M 13/256: D06M 13/402; COUNTERACTANTS D06M 23/06: D06M23/02: D06M 16/00 (75) Inventors: Kristin Rhedrick Williams, West USPC ...................................... 424/400, 76.1, 76.21 Chester, OH (US); Carla Jean Colina, See application file for complete search history. Cincinnati, OH (US); Cahit Eylem, West Chester, OH (US); Lon Montgomery (56) References Cited Gray, Florence, KY (US); Shih-Chuan U.S. PATENT DOCUMENTS Liou, Cincinnati, OH (US); Christine Marie Readnour, Fort Mitchell, KY 5,578,563 A 11/1996 Trinh et al. (US); Ricky Ah-Man Woo, Hamilton, 6,001,342 A 12/1999 Forestier et al. OH (US) (Continued) (73) Assignee: The Procter & Gamble Company, FOREIGN PATENT DOCUMENTS Cincinnati, OH (US) JP WO 82O1993 6, 1982 (*) Notice: Subject to any disclaimer, the term of this JP O3146.064 6, 1991 patent is extended or adjusted under 35 (Continued) U.S.C. 154(b) by 474 days. OTHER PUBLICATIONS This patent is Subject to a terminal dis “Methyl dihydrojasmonate’ CASEN 24851-98-7: http://www. claimer. chemicalbook.com/CASEN 24.851-98-7.htm. (21) Appl. No.: 12/562,534 (Continued) (22) Filed: Sep. 18, 2009 Primary Examiner — Sean Basquill (65) Prior Publication Data Assistant Examiner — Miriam A Levin (74) Attorney, Agent, or Firm — Abbey A.