The Role of Complement Receptors and Fc Gamma Receptor Iib in Collagen-Induced Arthritis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Role of IL-4 Receptor &Alpha
Role of IL-4 receptor a–positive CD41 T cells in chronic airway hyperresponsiveness Frank Kirstein, PhD, Natalie E. Nieuwenhuizen, PhD,* Jaisubash Jayakumar, PhD, William G. C. Horsnell, PhD, and Frank Brombacher, PhD Cape Town, South Africa, and Berlin, Germany Background: TH2 cells and their cytokines are associated with IL-17–producing T cells and, consequently, increased airway allergic asthma in human subjects and with mouse models of neutrophilia. allergic airway disease. IL-4 signaling through the IL-4 receptor Conclusion: IL-4–responsive T helper cells are dispensable for 1 a (IL-4Ra) chain on CD4 T cells leads to TH2 cell acute OVA-induced airway disease but crucial in maintaining differentiation in vitro, implying that IL-4Ra–responsive CD41 chronic asthmatic pathology. (J Allergy Clin Immunol T cells are critical for the induction of allergic asthma. However, 2016;137:1852-62.) mechanisms regulating acute and chronic allergen-specific T 2 H Key words: responses in vivo remain incompletely understood. TH2 cell, acute allergic airway disease, chronic asthma, Objective: This study defines the requirements for IL-4Ra– cytokine receptors, IL-4, IL-13, gene-deficient mice responsive CD41 T cells and the IL-4Ra ligands IL-4 and IL-13 in the development of allergen-specific TH2 responses during the Allergic asthma is a chronic inflammatory disease of the airways onset and chronic phase of experimental allergic airway disease. characterized by an inappropriate immune response to harmless Methods: Development of acute and chronic ovalbumin environmental antigens. T 2 cells regulate adaptive immune 1 H (OVA)–induced allergic asthma was assessed weekly in CD4 T responses to allergens, and their presence correlates with disease 2 cell–specific IL-4Ra–deficient BALB/c mice (LckcreIL-4Ra /lox) symptoms in human subjects and mice.1 IL-4 plays a crucial role and respective control mice in the presence or absence of IL-4 in the in vitro and in vivo differentiation of TH2 cells, suggesting or IL-13. -
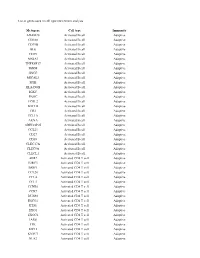
List of Genes Used in Cell Type Enrichment Analysis
List of genes used in cell type enrichment analysis Metagene Cell type Immunity ADAM28 Activated B cell Adaptive CD180 Activated B cell Adaptive CD79B Activated B cell Adaptive BLK Activated B cell Adaptive CD19 Activated B cell Adaptive MS4A1 Activated B cell Adaptive TNFRSF17 Activated B cell Adaptive IGHM Activated B cell Adaptive GNG7 Activated B cell Adaptive MICAL3 Activated B cell Adaptive SPIB Activated B cell Adaptive HLA-DOB Activated B cell Adaptive IGKC Activated B cell Adaptive PNOC Activated B cell Adaptive FCRL2 Activated B cell Adaptive BACH2 Activated B cell Adaptive CR2 Activated B cell Adaptive TCL1A Activated B cell Adaptive AKNA Activated B cell Adaptive ARHGAP25 Activated B cell Adaptive CCL21 Activated B cell Adaptive CD27 Activated B cell Adaptive CD38 Activated B cell Adaptive CLEC17A Activated B cell Adaptive CLEC9A Activated B cell Adaptive CLECL1 Activated B cell Adaptive AIM2 Activated CD4 T cell Adaptive BIRC3 Activated CD4 T cell Adaptive BRIP1 Activated CD4 T cell Adaptive CCL20 Activated CD4 T cell Adaptive CCL4 Activated CD4 T cell Adaptive CCL5 Activated CD4 T cell Adaptive CCNB1 Activated CD4 T cell Adaptive CCR7 Activated CD4 T cell Adaptive DUSP2 Activated CD4 T cell Adaptive ESCO2 Activated CD4 T cell Adaptive ETS1 Activated CD4 T cell Adaptive EXO1 Activated CD4 T cell Adaptive EXOC6 Activated CD4 T cell Adaptive IARS Activated CD4 T cell Adaptive ITK Activated CD4 T cell Adaptive KIF11 Activated CD4 T cell Adaptive KNTC1 Activated CD4 T cell Adaptive NUF2 Activated CD4 T cell Adaptive PRC1 Activated -

Genetic Susceptibility to Chronic Wasting Disease in Free-Ranging White-Tailed Deer: Complement Component C1q and Prnp Polymorphisms Julie A
Natural Resource Ecology and Management Natural Resource Ecology and Management Publications 12-2009 Genetic susceptibility to chronic wasting disease in free-ranging white-tailed deer: Complement component C1q and Prnp polymorphisms Julie A. Blanchong Iowa State University, [email protected] Dennis M. Heisey United States Geological Survey Kim T. Scribner Michigan State University Scot V. Libants Michigan State University Chad Johnson UFonilvloerwsit ythi of sW aiscondn asiddn - itMionadisoaln works at: http://lib.dr.iastate.edu/nrem_pubs Part of the Animal Diseases Commons, Genetics Commons, Natural Resources Management See next page for additional authors and Policy Commons, Veterinary Infectious Diseases Commons, and the Zoology Commons The ompc lete bibliographic information for this item can be found at http://lib.dr.iastate.edu/ nrem_pubs/84. For information on how to cite this item, please visit http://lib.dr.iastate.edu/ howtocite.html. This Article is brought to you for free and open access by the Natural Resource Ecology and Management at Iowa State University Digital Repository. It has been accepted for inclusion in Natural Resource Ecology and Management Publications by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. Genetic susceptibility to chronic wasting disease in free-ranging white- tailed deer: Complement component C1q and Prnp polymorphisms Abstract The eg netic basis of susceptibility to chronic wasting disease (CWD) in free-ranging cervids is of great interest. Association studies of disease susceptibility in free-ranging populations, however, face considerable challenges including: the need for large sample sizes when disease is rare, animals of unknown pedigree create a risk of spurious results due to population admixture, and the inability to control disease exposure or dose. -

Neutrophil Chemoattractant Receptors in Health and Disease: Double-Edged Swords
Cellular & Molecular Immunology www.nature.com/cmi REVIEW ARTICLE Neutrophil chemoattractant receptors in health and disease: double-edged swords Mieke Metzemaekers1, Mieke Gouwy1 and Paul Proost 1 Neutrophils are frontline cells of the innate immune system. These effector leukocytes are equipped with intriguing antimicrobial machinery and consequently display high cytotoxic potential. Accurate neutrophil recruitment is essential to combat microbes and to restore homeostasis, for inflammation modulation and resolution, wound healing and tissue repair. After fulfilling the appropriate effector functions, however, dampening neutrophil activation and infiltration is crucial to prevent damage to the host. In humans, chemoattractant molecules can be categorized into four biochemical families, i.e., chemotactic lipids, formyl peptides, complement anaphylatoxins and chemokines. They are critically involved in the tight regulation of neutrophil bone marrow storage and egress and in spatial and temporal neutrophil trafficking between organs. Chemoattractants function by activating dedicated heptahelical G protein-coupled receptors (GPCRs). In addition, emerging evidence suggests an important role for atypical chemoattractant receptors (ACKRs) that do not couple to G proteins in fine-tuning neutrophil migratory and functional responses. The expression levels of chemoattractant receptors are dependent on the level of neutrophil maturation and state of activation, with a pivotal modulatory role for the (inflammatory) environment. Here, we provide an overview -

An Anticomplement Agent That Homes to the Damaged Brain and Promotes Recovery After Traumatic Brain Injury in Mice
An anticomplement agent that homes to the damaged brain and promotes recovery after traumatic brain injury in mice Marieta M. Rusevaa,1,2, Valeria Ramagliab,1, B. Paul Morgana, and Claire L. Harrisa,3 aInstitute of Infection and Immunity, School of Medicine, Cardiff University, Cardiff CF14 4XN, United Kingdom; and bDepartment of Genome Analysis, Academic Medical Center, Amsterdam 1105 AZ, The Netherlands Edited by Douglas T. Fearon, Cornell University, Cambridge, United Kingdom, and approved September 29, 2015 (received for review July 15, 2015) Activation of complement is a key determinant of neuropathology to rapidly and specifically inhibit MAC at sites of complement and disability after traumatic brain injury (TBI), and inhibition is activation, and test its therapeutic potential in experimental TBI. neuroprotective. However, systemic complement is essential to The construct, termed CD59-2a-CRIg, comprises CD59a linked fight infections, a critical complication of TBI. We describe a to CRIg via the murine IgG2a hinge. CD59a prevents assembly targeted complement inhibitor, comprising complement receptor of MAC in cell membranes (16), whereas CRIg binds C3b/iC3b of the Ig superfamily (CRIg) fused with complement regulator CD59a, deposited at sites of complement activation (17). The IgG2a designed to inhibit membrane attack complex (MAC) assembly at hinge promotes dimerization to increase ligand avidity. CD59- sites of C3b/iC3b deposition. CRIg and CD59a were linked via the 2a-CRIg protected in the TBI model, demonstrating that site- IgG2a hinge, yielding CD59-2a-CRIg dimer with increased iC3b/C3b targeted anti-MAC therapeutics may be effective in prevention binding avidity and MAC inhibitory activity. CD59-2a-CRIg inhibited of secondary neuropathology and improve neurologic recovery MAC formation and prevented complement-mediated lysis in vitro. -

Single-Cell RNA Sequencing Demonstrates the Molecular and Cellular Reprogramming of Metastatic Lung Adenocarcinoma
ARTICLE https://doi.org/10.1038/s41467-020-16164-1 OPEN Single-cell RNA sequencing demonstrates the molecular and cellular reprogramming of metastatic lung adenocarcinoma Nayoung Kim 1,2,3,13, Hong Kwan Kim4,13, Kyungjong Lee 5,13, Yourae Hong 1,6, Jong Ho Cho4, Jung Won Choi7, Jung-Il Lee7, Yeon-Lim Suh8,BoMiKu9, Hye Hyeon Eum 1,2,3, Soyean Choi 1, Yoon-La Choi6,10,11, Je-Gun Joung1, Woong-Yang Park 1,2,6, Hyun Ae Jung12, Jong-Mu Sun12, Se-Hoon Lee12, ✉ ✉ Jin Seok Ahn12, Keunchil Park12, Myung-Ju Ahn 12 & Hae-Ock Lee 1,2,3,6 1234567890():,; Advanced metastatic cancer poses utmost clinical challenges and may present molecular and cellular features distinct from an early-stage cancer. Herein, we present single-cell tran- scriptome profiling of metastatic lung adenocarcinoma, the most prevalent histological lung cancer type diagnosed at stage IV in over 40% of all cases. From 208,506 cells populating the normal tissues or early to metastatic stage cancer in 44 patients, we identify a cancer cell subtype deviating from the normal differentiation trajectory and dominating the metastatic stage. In all stages, the stromal and immune cell dynamics reveal ontological and functional changes that create a pro-tumoral and immunosuppressive microenvironment. Normal resident myeloid cell populations are gradually replaced with monocyte-derived macrophages and dendritic cells, along with T-cell exhaustion. This extensive single-cell analysis enhances our understanding of molecular and cellular dynamics in metastatic lung cancer and reveals potential diagnostic and therapeutic targets in cancer-microenvironment interactions. 1 Samsung Genome Institute, Samsung Medical Center, Seoul 06351, Korea. -

B Cell Checkpoints in Autoimmune Rheumatic Diseases
REVIEWS B cell checkpoints in autoimmune rheumatic diseases Samuel J. S. Rubin1,2,3, Michelle S. Bloom1,2,3 and William H. Robinson1,2,3* Abstract | B cells have important functions in the pathogenesis of autoimmune diseases, including autoimmune rheumatic diseases. In addition to producing autoantibodies, B cells contribute to autoimmunity by serving as professional antigen- presenting cells (APCs), producing cytokines, and through additional mechanisms. B cell activation and effector functions are regulated by immune checkpoints, including both activating and inhibitory checkpoint receptors that contribute to the regulation of B cell tolerance, activation, antigen presentation, T cell help, class switching, antibody production and cytokine production. The various activating checkpoint receptors include B cell activating receptors that engage with cognate receptors on T cells or other cells, as well as Toll-like receptors that can provide dual stimulation to B cells via co- engagement with the B cell receptor. Furthermore, various inhibitory checkpoint receptors, including B cell inhibitory receptors, have important functions in regulating B cell development, activation and effector functions. Therapeutically targeting B cell checkpoints represents a promising strategy for the treatment of a variety of autoimmune rheumatic diseases. Antibody- dependent B cells are multifunctional lymphocytes that contribute that serve as precursors to and thereby give rise to acti- cell- mediated cytotoxicity to the pathogenesis of autoimmune diseases -

Cd4/Cd8 Panel, Blood
Lab Dept: Flow and Immunology Test Name: CD4/CD8 PANEL, BLOOD General Information Lab Order Codes: C48P Synonyms: Helper/Suppressor Ratio; T- cells; T-cell subsets; T-cell phenotyping See also: Immune Status Panel and Comprehensive Immune Status Panel CPT Codes: 86359 – T cells, total count 86360 – T cells; absolute CD4 and CD8 count, including ratio Test Includes: CD4(CD3+) and CD8(CD3+) relative percentages, absolute values and a calculated Helper/Suppressor ratio. Logistics Test Indications: This is a minimal antibody panel to monitor immune status. Lab Testing Sections: Flow Cytometry Phone Numbers: MIN Lab: 612-813-6280 STP Lab: 651-220-6550 Test Availability: 3 times weekly determined by volume. Transport collected specimen immediately to Flow Cytometry. Routine testing is not available on weekends or holidays. Therefore, specimens cannot be used if drawn the day before a 3 day weekend such as Memorial Day, Labor Day or major holiday that falls on a Monday or Friday. Turnaround Time: 1 – 3 days Special Instructions: See Test Availability Specimen Specimen Type: Whole blood Container: Lavender top (EDTA) tube Draw Volume: 2 mL blood in a 2 mL Lavender (EDTA) tube Minimum volume: 0.5 mL in an EDTA microtainer Collection: Routine venipuncture Special Processing: Keep sample at room temperature and forward promptly to the laboratory. Do not centrifuge, refrigerate, or freeze sample. Patient Preparation: None Sample Rejection: Specimens will not be processed that are clotted; hemolyzed; greater than 72 hours old; collected in the wrong tube type (0.5 mL in a 2 mL tube), or that have been held or handled at a temperature other than room temperature Interpretive Reference Range: Age-dependant reference ranges provided with results. -

In Vitro Selection for Adhesion of Plasmodium Falciparum-Infected Erythrocytes to ABO Antigens Does Not Affect Pfemp1 and RIFIN
www.nature.com/scientificreports OPEN In vitro selection for adhesion of Plasmodium falciparum‑infected erythrocytes to ABO antigens does not afect PfEMP1 and RIFIN expression William van der Puije1,2, Christian W. Wang 4, Srinidhi Sudharson 2, Casper Hempel 2, Rebecca W. Olsen 4, Nanna Dalgaard 4, Michael F. Ofori 1, Lars Hviid 3,4, Jørgen A. L. Kurtzhals 2,4 & Trine Staalsoe 2,4* Plasmodium falciparum causes the most severe form of malaria in humans. The adhesion of the infected erythrocytes (IEs) to endothelial receptors (sequestration) and to uninfected erythrocytes (rosetting) are considered major elements in the pathogenesis of the disease. Both sequestration and rosetting appear to involve particular members of several IE variant surface antigens (VSAs) as ligands, interacting with multiple vascular host receptors, including the ABO blood group antigens. In this study, we subjected genetically distinct P. falciparum parasites to in vitro selection for increased IE adhesion to ABO antigens in the absence of potentially confounding receptors. The selection resulted in IEs that adhered stronger to pure ABO antigens, to erythrocytes, and to various human cell lines than their unselected counterparts. However, selection did not result in marked qualitative changes in transcript levels of the genes encoding the best-described VSA families, PfEMP1 and RIFIN. Rather, overall transcription of both gene families tended to decline following selection. Furthermore, selection-induced increases in the adhesion to ABO occurred in the absence of marked changes in immune IgG recognition of IE surface antigens, generally assumed to target mainly VSAs. Our study sheds new light on our understanding of the processes and molecules involved in IE sequestration and rosetting. -

Rational Design of a CD4 Mimic That Inhibits HIV-1 Entry and Exposes Cryptic Neutralization Epitopes
RESEARCH ARTICLE Rational design of a CD4 mimic that inhibits HIV-1 entry and exposes cryptic neutralization epitopes Loïc Martin1, François Stricher1, Dorothée Missé2, Francesca Sironi3, Martine Pugnière4, Philippe Barthe5, Rafael Prado-Gotor5, Isabelle Freulon1, Xavier Magne2, Christian Roumestand5, André Ménez1, Paolo Lusso3, Francisco Veas2, and Claudio Vita1* Published online 16 December 2002; doi:10.1038/nbt768 The conserved surfaces of the human immunodeficiency virus (HIV)-1 envelope involved in receptor binding represent potential targets for the development of entry inhibitors and neutralizing antibodies. Using structural information on a CD4-gp120-17b antibody complex, we have designed a 27-amino acid CD4 mimic, CD4M33, that presents optimal interactions with gp120 and binds to viral particles and diverse HIV-1 envelopes with CD4-like affinity.This mini-CD4 inhibits infection of both immortalized and primary cells by HIV-1, including pri- http://www.nature.com/naturebiotechnology mary patient isolates that are generally resistant to inhibition by soluble CD4. Furthermore, CD4M33 pos- sesses functional properties of CD4, including the ability to unmask conserved neutralization epitopes of gp120 that are cryptic on the unbound glycoprotein. CD4M33 is a prototype of inhibitors of HIV-1 entry and, in complex with envelope proteins, a potential component of vaccine formulations, or a molecular target in phage display technology to develop broad-spectrum neutralizing antibodies. HIV-1 avoids the host immune surveillance by exposing -

Tethering IL2 to Its Receptor Il2rb Enhances Antitumor Activity and Expansion of Natural Killer NK92 Cells Youssef Jounaidi, Joseph F
Published OnlineFirst September 15, 2017; DOI: 10.1158/0008-5472.CAN-17-1007 Cancer Therapeutics, Targets, and Chemical Biology Research Tethering IL2 to Its Receptor IL2Rb Enhances Antitumor Activity and Expansion of Natural Killer NK92 Cells Youssef Jounaidi, Joseph F. Cotten, Keith W. Miller, and Stuart A. Forman Abstract IL2 is an immunostimulatory cytokine for key immune cells of IL2 and its receptor IL2Rb joined via a peptide linker (CIRB). including T cells and natural killer (NK) cells. Systemic IL2 NK92 cells expressing CIRB (NK92CIRB) were highly activated and supplementation could enhance NK-mediated immunity in a expanded indefinitely without exogenous IL2. When compared variety of diseases ranging from neoplasms to viral infection. with an IL2-secreting NK92 cell line, NK92CIRB were more acti- However, its systemic use is restricted by its serious side effects and vated, cytotoxic, and resistant to growth inhibition. Direct contact limited efficacy due to activation of T regulatory cells (Tregs). IL2 with cancer cells enhanced the cytotoxic character of NK92CIRB signaling is mediated through interactions with a multi-subunit cells, which displayed superior in vivo antitumor effects in mice. receptor complex containing IL2Ra, IL2Rb, and IL2Rg. Adult Overall, our results showed how tethering IL2 to its receptor natural killer (NK) cells express only IL2Rb and IL2Rg subunits IL2Rb eliminates the need for IL2Ra and IL2Rb, offering a and are therefore relatively insensitive to IL2. To overcome these new tool to selectively activate and empower immune therapy. limitations, we created a novel chimeric IL2-IL2Rb fusion protein Cancer Res; 77(21); 5938–51. Ó2017 AACR. Introduction in order for allogeneic NK cells to be effective, pretransfer lymphodepletion is required to reduce competition for growth Natural killer (NK) cells are lymphocytes endowed with the factors and cytokines (14, 15). -

Structural Insight on the Recognition of Surface-Bound Opsonins by the Integrin I Domain of Complement Receptor 3
Structural insight on the recognition of surface-bound opsonins by the integrin I domain of complement receptor 3 Goran Bajica, Laure Yatimea, Robert B. Simb, Thomas Vorup-Jensenc,1, and Gregers R. Andersena,1 Departments of aMolecular Biology and Genetics and cBiomedicine, Aarhus University, DK-8000 Aarhus, Denmark; and bDepartment of Pharmacology, University of Oxford, Oxford OX1 3QT, United Kingdom Edited by Douglas T. Fearon, University of Cambridge School of Clinical Medicine, Cambridge, United Kingdom, and approved August 28, 2013 (received for review June 13, 2013) Complement receptors (CRs), expressed notably on myeloid and degranulation, and changes in leukocyte cytokine production (2, lymphoid cells, play an essential function in the elimination of 5–7). CR3, and to a lesser degree CR4, are essential for the complement-opsonized pathogens and apoptotic/necrotic cells. In phagocytosis of complement-opsonized particles or complexes addition, these receptors are crucial for the cross-talk between the (6, 8, 9). Complement-opsonized immune complexes are cap- innate andadaptive branches ofthe immune system. CR3 (also known tured in the lymph nodes by CR3-positive subcapsular sinus as Mac-1, integrin α β , or CD11b/CD18) is expressed on all macro- macrophages (SSMs) and conveyed directly to naïve B cells or M 2 γ phages and recognizes iC3b on complement-opsonized objects, en- through follicular dendritic cells (10) using CR1, CR2, and Fc abling their phagocytosis. We demonstrate that the C3d moiety of receptors for antigen capture (11, 12). Hence, antigen-presenting iC3b harbors the binding site for the CR3 αI domain, and our structure cells such as SSMs may act as antigen storage and provide B of the C3d:αI domain complex rationalizes the CR3 selectivity for iC3b.