Mxaf Gene, a Gene Encoding Alpha Subunit of Methanol Dehydrogenase in and False Growth of Acetic Acid Bacteria on Methanol
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Asaia Bogorensis Gen. Nov., Sp. Nov., an Unusual Acetic Acid Bacterium in the Α-Proteobacteria
International Journal of Systematic and Evolutionary Microbiology (2000) 50, 823–829 Printed in Great Britain Asaia bogorensis gen. nov., sp. nov., an unusual acetic acid bacterium in the α-Proteobacteria Yuzo Yamada,1 Kazushige Katsura,1 Hiroko Kawasaki,2 Yantyati Widyastuti,3 Susono Saono,3 Tatsuji Seki,2 Tai Uchimura1 and Kazuo Komagata1 Author for correspondence: Yuzo Yamada. Tel\Fax: j81 54 635 2316. 1 Laboratory of General and Eight Gram-negative, aerobic, rod-shaped and peritrichously flagellated strains Applied Microbiology, were isolated from flowers of the orchid tree (Bauhinia purpurea) and of Department of Applied Biology and Chemistry, plumbago (Plumbago auriculata), and from fermented glutinous rice, all Faculty of Applied collected in Indonesia. The enrichment culture approach for acetic acid bacteria Bioscience, Tokyo was employed, involving use of sorbitol medium at pH 35. All isolates grew University of Agriculture, Sakuragaoka 1-1-1, well at pH 30 and 30 SC. They did not oxidize ethanol to acetic acid except for Setagaya-ku, one strain that oxidized ethanol weakly, and 035% acetic acid inhibited their Tokoyo 156-8502, Japan growth completely. However, they oxidized acetate and lactate to carbon 2 The International Center dioxide and water. The isolates grew well on mannitol agar and on glutamate for Biotechnology, Osaka agar, and assimilated ammonium sulfate for growth on vitamin-free glucose University, Yamadaoka 2-1, Suita, Osaka 565-0871, medium. The isolates produced acid from D-glucose, D-fructose, L-sorbose, Japan dulcitol and glycerol. The quinone system was Q-10. DNA base composition 3 Research and ranged from 593to610 mol% GMC. -
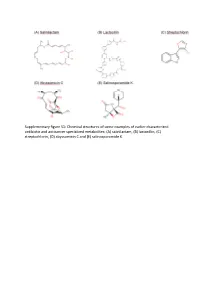
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

Developing a Genetic Manipulation System for the Antarctic Archaeon, Halorubrum Lacusprofundi: Investigating Acetamidase Gene Function
www.nature.com/scientificreports OPEN Developing a genetic manipulation system for the Antarctic archaeon, Halorubrum lacusprofundi: Received: 27 May 2016 Accepted: 16 September 2016 investigating acetamidase gene Published: 06 October 2016 function Y. Liao1, T. J. Williams1, J. C. Walsh2,3, M. Ji1, A. Poljak4, P. M. G. Curmi2, I. G. Duggin3 & R. Cavicchioli1 No systems have been reported for genetic manipulation of cold-adapted Archaea. Halorubrum lacusprofundi is an important member of Deep Lake, Antarctica (~10% of the population), and is amendable to laboratory cultivation. Here we report the development of a shuttle-vector and targeted gene-knockout system for this species. To investigate the function of acetamidase/formamidase genes, a class of genes not experimentally studied in Archaea, the acetamidase gene, amd3, was disrupted. The wild-type grew on acetamide as a sole source of carbon and nitrogen, but the mutant did not. Acetamidase/formamidase genes were found to form three distinct clades within a broad distribution of Archaea and Bacteria. Genes were present within lineages characterized by aerobic growth in low nutrient environments (e.g. haloarchaea, Starkeya) but absent from lineages containing anaerobes or facultative anaerobes (e.g. methanogens, Epsilonproteobacteria) or parasites of animals and plants (e.g. Chlamydiae). While acetamide is not a well characterized natural substrate, the build-up of plastic pollutants in the environment provides a potential source of introduced acetamide. In view of the extent and pattern of distribution of acetamidase/formamidase sequences within Archaea and Bacteria, we speculate that acetamide from plastics may promote the selection of amd/fmd genes in an increasing number of environmental microorganisms. -

Polyamine Profiles of Some Members of the Alpha Subclass of the Class Proteobacteria: Polyamine Analysis of Twenty Recently Described Genera
Microbiol. Cult. Coll. June 2003. p. 13 ─ 21 Vol. 19, No. 1 Polyamine Profiles of Some Members of the Alpha Subclass of the Class Proteobacteria: Polyamine Analysis of Twenty Recently Described Genera Koei Hamana1)*,Azusa Sakamoto1),Satomi Tachiyanagi1), Eri Terauchi1)and Mariko Takeuchi2) 1)Department of Laboratory Sciences, School of Health Sciences, Faculty of Medicine, Gunma University, 39 ─ 15 Showa-machi 3 ─ chome, Maebashi, Gunma 371 ─ 8514, Japan 2)Institute for Fermentation, Osaka, 17 ─ 85, Juso-honmachi 2 ─ chome, Yodogawa-ku, Osaka, 532 ─ 8686, Japan Cellular polyamines of 41 newly validated or reclassified alpha proteobacteria belonging to 20 genera were analyzed by HPLC. Acetic acid bacteria belonging to the new genus Asaia and the genera Gluconobacter, Gluconacetobacter, Acetobacter and Acidomonas of the alpha ─ 1 sub- group ubiquitously contained spermidine as the major polyamine. Aerobic bacteriochlorophyll a ─ containing Acidisphaera, Craurococcus and Paracraurococcus(alpha ─ 1)and Roseibium (alpha-2)contained spermidine and lacked homospermidine. New Rhizobium species, including some species transferred from the genera Agrobacterium and Allorhizobium, and new Sinorhizobium and Mesorhizobium species of the alpha ─ 2 subgroup contained homospermidine as a major polyamine. Homospermidine was the major polyamine in the genera Oligotropha, Carbophilus, Zavarzinia, Blastobacter, Starkeya and Rhodoblastus of the alpha ─ 2 subgroup. Rhodobaca bogoriensis of the alpha ─ 3 subgroup contained spermidine. Within the alpha ─ 4 sub- group, the genus Sphingomonas has been divided into four clusters, and species of the emended Sphingomonas(cluster I)contained homospermidine whereas those of the three newly described genera Sphingobium, Novosphingobium and Sphingopyxis(corresponding to clusters II, III and IV of the former Sphingomonas)ubiquitously contained spermidine. -

Oleomonas Sagaranensis Gen. Nov., Sp. Nov., Represents a Novel Genus in the K-Proteobacteria
FEMS Microbiology Letters 217 (2002) 255^261 www.fems-microbiology.org Oleomonas sagaranensis gen. nov., sp. nov., represents a novel genus in the K-Proteobacteria Takeshi Kanamori a, Naeem Rashid a, Masaaki Morikawa b, Haruyuki Atomi a, a;Ã Tadayuki Imanaka Downloaded from https://academic.oup.com/femsle/article/217/2/255/502948 by guest on 01 October 2021 a Department of Synthetic Chemistry and Biological Chemistry, Graduate School of Engineering, Kyoto University, Yoshida-Honmachi, Sakyo-ku, Kyoto 606-8501, Japan, and Core Research for Evolutional Science and Technology Program of Japan Science and Technology Corporation (CREST-JST), Kawaguchi, Saitama 332-0012, Japan b Department of Material and Life Science, Graduate School of Engineering, Osaka University, 2-1 Yamadaoka, Suita, Osaka 565-0871, Japan Received 13 July 2002; received in revised form 7 October 2002; accepted 21 October 2002 First published online 7 November 2002 Abstract A Gram-negative bacterium was previously isolated from an oil field in Shizuoka, Japan, and designated strain HD-1. Here we have performed detailed characterization of the strain, and have found that it represents a novel genus. The 16S rRNA sequence of strain HD-1 displayed highest similarity to various uncultured species (86.7V99.7%), along with 86.2V88.2% similarity to sequences from Azospirillum, Methylobacterium, Rhizobium, and Hyphomicrobium, all members of the K-Proteobacteria. Phylogeneticanalysis revealed that HD-1 represented a deep-branched lineage among the K-Proteobacteria. DNA^DNA hybridization analysis with Azospirillum lipoferum and Hyphomicrobium vulgare revealed low levels of similarity among the strains. We further examined the biochemical properties of the strain under aerobic conditions. -

Advances in Applied Microbiology, Voume 49 (Advances in Applied
ADVANCES IN Applied Microbiology VOLUME 49 ThisPageIntentionallyLeftBlank ADVANCES IN Applied Microbiology Edited by ALLEN I. LASKIN JOAN W. BENNETT Somerset, New Jersey New Orleans, Louisiana GEOFFREY M. GADD Dundee, United Kingdom VOLUME 49 San Diego New York Boston London Sydney Tokyo Toronto This book is printed on acid-free paper. ∞ Copyright C 2001 by ACADEMIC PRESS All Rights Reserved. No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopy, recording, or any information storage and retrieval system, without permission in writing from the Publisher. The appearance of the code at the bottom of the first page of a chapter in this book indicates the Publisher’s consent that copies of the chapter may be made for personal or internal use of specific clients. This consent is given on the condition, however, that the copier pay the stated per copy fee through the Copyright Clearance Center, Inc. (222 Rosewood Drive, Danvers, Massachusetts 01923), for copying beyond that permitted by Sections 107 or 108 of the U.S. Copyright Law. This consent does not extend to other kinds of copying, such as copying for general distribution, for advertising or promotional purposes, for creating new collective works, or for resale. Copy fees for pre-2000 chapters are as shown on the title pages. If no fee code appears on the title page, the copy fee is the same as for current chapters. 0065-2164/01 $35.00 Academic Press A division of Harcourt, Inc. 525 B Street, Suite 1900, San Diego, California 92101-4495, USA http://www.academicpress.com Academic Press Harcourt Place, 32 Jamestown Road, London NW1 7BY, UK http://www.academicpress.com International Standard Serial Number: 0065-2164 International Standard Book Number: 0-12-002649-X PRINTED IN THE UNITED STATES OF AMERICA 010203040506MM987654321 CONTENTS Microbial Transformations of Explosives SUSAN J. -

Chemosynthetic Symbiont with a Drastically Reduced Genome Serves As Primary Energy Storage in the Marine Flatworm Paracatenula
Chemosynthetic symbiont with a drastically reduced genome serves as primary energy storage in the marine flatworm Paracatenula Oliver Jäcklea, Brandon K. B. Seaha, Målin Tietjena, Nikolaus Leischa, Manuel Liebekea, Manuel Kleinerb,c, Jasmine S. Berga,d, and Harald R. Gruber-Vodickaa,1 aMax Planck Institute for Marine Microbiology, 28359 Bremen, Germany; bDepartment of Geoscience, University of Calgary, AB T2N 1N4, Canada; cDepartment of Plant & Microbial Biology, North Carolina State University, Raleigh, NC 27695; and dInstitut de Minéralogie, Physique des Matériaux et Cosmochimie, Université Pierre et Marie Curie, 75252 Paris Cedex 05, France Edited by Margaret J. McFall-Ngai, University of Hawaii at Manoa, Honolulu, HI, and approved March 1, 2019 (received for review November 7, 2018) Hosts of chemoautotrophic bacteria typically have much higher thrive in both free-living environmental and symbiotic states, it is biomass than their symbionts and consume symbiont cells for difficult to attribute their genomic features to either functions nutrition. In contrast to this, chemoautotrophic Candidatus Riegeria they provide to their host, or traits that are necessary for envi- symbionts in mouthless Paracatenula flatworms comprise up to ronmental survival or to both. half of the biomass of the consortium. Each species of Paracate- The smallest genomes of chemoautotrophic symbionts have nula harbors a specific Ca. Riegeria, and the endosymbionts have been observed for the gammaproteobacterial symbionts of ves- been vertically transmitted for at least 500 million years. Such icomyid clams that are directly transmitted between host genera- prolonged strict vertical transmission leads to streamlining of sym- tions (13, 14). Such strict vertical transmission leads to substantial biont genomes, and the retained physiological capacities reveal and ongoing genome reduction. -

Dyella Japonica Gen. Nov., Sp. Nov., a C-Proteobacterium Isolated from Soil
International Journal of Systematic and Evolutionary Microbiology (2005), 55, 753–756 DOI 10.1099/ijs.0.63377-0 Dyella japonica gen. nov., sp. nov., a c-proteobacterium isolated from soil Cheng-Hui Xie and Akira Yokota Correspondence Institute of Molecular and Cellular Biosciences, The University of Tokyo, Yayoi 1-1-1, Cheng-Hui Xie Bunkyo-Ku, Tokyo 113-0032, Japan [email protected] Three strains isolated from the soil of a garden in Tokyo, Japan, were characterized physiologically, biochemically and in terms of fatty acid profile, DNA–DNA relatedness and 16S rRNA gene sequence. The isolates were Gram-negative, aerobic, rod-shaped cells with polar flagellation. According to DNA–DNA similarity, the strains belonged to the same species. The bacteria grew at temperatures from 10 to 37 6C, with an optimum around 25–30 6C. Growth was observed at pH values from 5?6to8?0. The DNA G+C content ranged from 63?4to 64?0 mol%. Phylogenetic analyses of 16S rRNA gene sequences revealed a clear affiliation with members of the family ‘Xanthomonadaceae’. The closest relationship was seen with Fulvimonas soli and Frateuria aurantia, but, in terms of physiology and fatty acid profile, the bacteria described were rather distant from Fulvimonas and Frateuria. On the basis of phenotypic and phylogenetic distinctness, it is proposed that the isolates represent a novel species in a novel genus, namely Dyella japonica gen. nov., sp. nov. The type strain is XD53T (=IAM 15069T=DSM 16301T=ATCC BAA-939T). Three bacterial strains, XD10, XD22 and XD53T, were japonica gen. nov., sp. -

Methanol Consumption Drives the Bacterial Chloromethane Sink in a Forest Soil
The ISME Journal (2018) 12:2681–2693 https://doi.org/10.1038/s41396-018-0228-4 ARTICLE Methanol consumption drives the bacterial chloromethane sink in a forest soil 1,2 2 1,4 3 1 Pauline Chaignaud ● Mareen Morawe ● Ludovic Besaury ● Eileen Kröber ● Stéphane Vuilleumier ● 1 3 Françoise Bringel ● Steffen Kolb Received: 16 February 2018 / Revised: 1 June 2018 / Accepted: 15 June 2018 / Published online: 10 July 2018 © The Author(s) 2018. This article is published with open access Abstract Halogenated volatile organic compounds (VOCs) emitted by terrestrial ecosystems, such as chloromethane (CH3Cl), have pronounced effects on troposphere and stratosphere chemistry and climate. The magnitude of the global CH3Cl sink is uncertain since it involves a largely uncharacterized microbial sink. CH3Cl represents a growth substrate for some specialized methylotrophs, while methanol (CH3OH), formed in much larger amounts in terrestrial environments, may be more widely used by such microorganisms. Direct measurements of CH3Cl degradation rates in two field campaigns and in microcosms allowed the identification of top soil horizons (i.e., organic plus mineral A horizon) as the major biotic sink in a fi 1234567890();,: 1234567890();,: deciduous forest. Metabolically active members of Alphaproteobacteria and Actinobacteria were identi ed by taxonomic and functional gene biomarkers following stable isotope labeling (SIP) of microcosms with CH3Cl and CH3OH, added alone 13 or together as the [ C]-isotopologue. Well-studied reference CH3Cl degraders, such as Methylobacterium extorquens CM4, were not involved in the sink activity of the studied soil. Nonetheless, only sequences of the cmuA chloromethane dehalogenase gene highly similar to those of known strains were detected, suggesting the relevance of horizontal gene transfer for CH3Cl degradation in forest soil. -

A Microbiotic Survey of Lichen-Associated Bacteria Reveals a New Lineage from the Rhizobiales
SYMBIOSIS (2009) 49, 163–180 ©Springer Science+Business Media B.V. 2009 ISSN 0334-5114 A microbiotic survey of lichen-associated bacteria reveals a new lineage from the Rhizobiales Brendan P. Hodkinson* and François Lutzoni Department of Biology, Duke University, Box 90338, Durham, NC 27708, USA, Tel. +1-443-340-0917, Fax. +1-919-660-7293, Email. [email protected] (Received June 10, 2008; Accepted November 5, 2009) Abstract This study uses a set of PCR-based methods to examine the putative microbiota associated with lichen thalli. In initial experiments, generalized oligonucleotide-primers for the 16S rRNA gene resulted in amplicon pools populated almost exclusively with fragments derived from lichen photobionts (i.e., Cyanobacteria or chloroplasts of algae). This effectively masked the presence of other lichen-associated prokaryotes. In order to facilitate the study of the lichen microbiota, 16S ribosomal oligonucleotide-primers were developed to target Bacteria, but exclude sequences derived from chloroplasts and Cyanobacteria. A preliminary microbiotic survey of lichen thalli using these new primers has revealed the identity of several bacterial associates, including representatives of the extremophilic Acidobacteria, bacteria in the families Acetobacteraceae and Brucellaceae, strains belonging to the genus Methylobacterium, and members of an undescribed lineage in the Rhizobiales. This new lineage was investigated and characterized through molecular cloning, and was found to be present in all examined lichens that are associated with green algae. There is evidence to suggest that members of this lineage may both account for a large proportion of the lichen-associated bacterial community and assist in providing the lichen thallus with crucial nutrients such as fixed nitrogen. -

Identification of Frateuria Aurantia Strains Isolated from Indonesian Sources
Microbiol. Cult. Coll. Dec. 2003. p. 81 ― 90 Vol. 19, No. 2 Identification of Frateuria aurantia Strains Isolated from Indonesian Sources Puspita Lisdiyanti *, Yuzo Yamada, Tai Uchimura, and Kazuo Komagata Department of Applied Biology and Chemistry, Faculty of Applied Bioscience, Tokyo University of Agriculture, 1-1-1 Sakuragaoka, Setagaya-ku, Tokyo 156-8502, Japan In the course of the isolation of acetic acid bacteria from tropical countries, 16 water-soluble brown pigment-producing bacteria were isolated from fruits and flowers in Indonesia. Two rep- resentative isolates were clustered in the γ-Proteobacteria together with the genus Frateuria by 16S rRNA gene sequence analysis. All isolates were identified as Frateuria aurantia by DNA-DNA similarity. The strains were aerobic, Gram-negative rods and motile with a single polar flagellum. They grew at pH 3.5, produced acetic acid from ethanol and brown water-soluble pigment, oxidized lactate but not acetate, did not grow in the presence of 0.35 % acetic acid, and did not produce H2S. Further, they produced D-gluconate, 2-keto-D-gluconate, and 2, 5-diketo-D-gluconate from D-glucose but not 5-keto-D-gluconate. All isolates tested had Q-8 as the major ubiquinone and iso-branched acid of C15 : 0 as the major cellular fatty acid. The DNA base composition of the isolates ranged from 62 to 63 mol %. This study is the fourth case of the isolation of F. aurantia strains so far, and showed the distribution of this species not only in Japan but also in tropical regions. Key words: Frateuria aurantia, acetic acid bacteria INTRODUCTION The second case was reported by Ameyama and Kondo The genus Frateuria was validly established by (1)with three strains isolated from rose(Rose hybri- Swings et al. -

Swaminathania Salitolerans Gen. Nov., Sp. Nov., a Salt-Tolerant, Nitrogen-fixing and Phosphate-Solubilizing Bacterium from Wild Rice (Porteresia Coarctata Tateoka)
International Journal of Systematic and Evolutionary Microbiology (2004), 54, 1185–1190 DOI 10.1099/ijs.0.02817-0 Swaminathania salitolerans gen. nov., sp. nov., a salt-tolerant, nitrogen-fixing and phosphate-solubilizing bacterium from wild rice (Porteresia coarctata Tateoka) P. Loganathan and Sudha Nair Correspondence M. S. Swaminathan Research Foundation, 111 Cross St, Tharamani Institutional Area, Chennai, Sudha Nair Madras 600 113, India [email protected] A novel species, Swaminathania salitolerans gen. nov., sp. nov., was isolated from the rhizosphere, roots and stems of salt-tolerant, mangrove-associated wild rice (Porteresia coarctata Tateoka) using nitrogen-free, semi-solid LGI medium at pH 5?5. Strains were Gram-negative, rod-shaped and motile with peritrichous flagella. The strains grew well in the presence of 0?35 % acetic acid, 3 % NaCl and 1 % KNO3, and produced acid from L-arabinose, D-glucose, glycerol, ethanol, D-mannose, D-galactose and sorbitol. They oxidized ethanol and grew well on mannitol and glutamate agar. The fatty acids 18 : 1v7c/v9t/v12t and 19 : 0cyclo v8c constituted 30?41 and 11?80 % total fatty acids, respectively, whereas 13 : 1 AT 12–13 was found at 0?53 %. DNA G+C content was 57?6–59?9 mol% and the major quinone was Q-10. Phylogenetic analysis based on 16S rRNA gene sequences showed that these strains were related to the genera Acidomonas, Asaia, Acetobacter, Gluconacetobacter, Gluconobacter and Kozakia in the Acetobacteraceae. Isolates were able to fix nitrogen and solubilized phosphate in the presence of NaCl. Based on overall analysis of the tests and comparison with the characteristics of members of the Acetobacteraceae, a novel genus and species is proposed for these isolates, Swaminathania salitolerans gen.