13 Classical and Quantum Statistics
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Fermi- Dirac Statistics
Prof. Surajit Dhara Guest Teacher, Dept. Of Physics, Narajole Raj College GE2T (Thermal physics and Statistical Mechanics) , Topic :- Fermi- Dirac Statistics Fermi- Dirac Statistics Basic features: The basic features of the Fermi-Dirac statistics: 1. The particles are all identical and hence indistinguishable. 2. The fermions obey (a) the Heisenberg’s uncertainty relation and also (b) the exclusion principle of Pauli. 3. As a consequence of 2(a), there exists a number of quantum states for a given energy level and because of 2(b) , there is a definite a priori restriction on the number of fermions in a quantum state; there can be simultaneously no more than one particle in a quantum state which would either remain empty or can at best contain one fermion. If , again, the particles are isolated and non-interacting, the following two additional condition equations apply to the system: ∑ ; Thermodynamic probability: Consider an isolated system of N indistinguishable, non-interacting particles obeying Pauli’s exclusion principle. Let particles in the system have energies respectively and let denote the degeneracy . So the number of distinguishable arrangements of particles among eigenstates in the ith energy level is …..(1) Therefore, the thermodynamic probability W, that is, the total number of s eigenstates of the whole system is the product of ……(2) GE2T (Thermal physics and Statistical Mechanics), Topic :- Fermi- Dirac Statistics: Circulated by-Prof. Surajit Dhara, Dept. Of Physics, Narajole Raj College FD- distribution function : Most probable distribution : From eqn(2) above , taking logarithm and applying Stirling’s theorem, For this distribution to represent the most probable distribution , the entropy S or klnW must be maximum, i.e. -

Canonical Ensemble
ME346A Introduction to Statistical Mechanics { Wei Cai { Stanford University { Win 2011 Handout 8. Canonical Ensemble January 26, 2011 Contents Outline • In this chapter, we will establish the equilibrium statistical distribution for systems maintained at a constant temperature T , through thermal contact with a heat bath. • The resulting distribution is also called Boltzmann's distribution. • The canonical distribution also leads to definition of the partition function and an expression for Helmholtz free energy, analogous to Boltzmann's Entropy formula. • We will study energy fluctuation at constant temperature, and witness another fluctuation- dissipation theorem (FDT) and finally establish the equivalence of micro canonical ensemble and canonical ensemble in the thermodynamic limit. (We first met a mani- festation of FDT in diffusion as Einstein's relation.) Reading Assignment: Reif x6.1-6.7, x6.10 1 1 Temperature For an isolated system, with fixed N { number of particles, V { volume, E { total energy, it is most conveniently described by the microcanonical (NVE) ensemble, which is a uniform distribution between two constant energy surfaces. const E ≤ H(fq g; fp g) ≤ E + ∆E ρ (fq g; fp g) = i i (1) mc i i 0 otherwise Statistical mechanics also provides the expression for entropy S(N; V; E) = kB ln Ω. In thermodynamics, S(N; V; E) can be transformed to a more convenient form (by Legendre transform) of Helmholtz free energy A(N; V; T ), which correspond to a system with constant N; V and temperature T . Q: Does the transformation from N; V; E to N; V; T have a meaning in statistical mechanics? A: The ensemble of systems all at constant N; V; T is called the canonical NVT ensemble. -

Magnetism, Magnetic Properties, Magnetochemistry
Magnetism, Magnetic Properties, Magnetochemistry 1 Magnetism All matter is electronic Positive/negative charges - bound by Coulombic forces Result of electric field E between charges, electric dipole Electric and magnetic fields = the electromagnetic interaction (Oersted, Maxwell) Electric field = electric +/ charges, electric dipole Magnetic field ??No source?? No magnetic charges, N-S No magnetic monopole Magnetic field = motion of electric charges (electric current, atomic motions) Magnetic dipole – magnetic moment = i A [A m2] 2 Electromagnetic Fields 3 Magnetism Magnetic field = motion of electric charges • Macro - electric current • Micro - spin + orbital momentum Ampère 1822 Poisson model Magnetic dipole – magnetic (dipole) moment [A m2] i A 4 Ampere model Magnetism Microscopic explanation of source of magnetism = Fundamental quantum magnets Unpaired electrons = spins (Bohr 1913) Atomic building blocks (protons, neutrons and electrons = fermions) possess an intrinsic magnetic moment Relativistic quantum theory (P. Dirac 1928) SPIN (quantum property ~ rotation of charged particles) Spin (½ for all fermions) gives rise to a magnetic moment 5 Atomic Motions of Electric Charges The origins for the magnetic moment of a free atom Motions of Electric Charges: 1) The spins of the electrons S. Unpaired spins give a paramagnetic contribution. Paired spins give a diamagnetic contribution. 2) The orbital angular momentum L of the electrons about the nucleus, degenerate orbitals, paramagnetic contribution. The change in the orbital moment -

Spin Statistics, Partition Functions and Network Entropy
This is a repository copy of Spin Statistics, Partition Functions and Network Entropy. White Rose Research Online URL for this paper: https://eprints.whiterose.ac.uk/118694/ Version: Accepted Version Article: Wang, Jianjia, Wilson, Richard Charles orcid.org/0000-0001-7265-3033 and Hancock, Edwin R orcid.org/0000-0003-4496-2028 (2017) Spin Statistics, Partition Functions and Network Entropy. Journal of Complex Networks. pp. 1-25. ISSN 2051-1329 https://doi.org/10.1093/comnet/cnx017 Reuse Items deposited in White Rose Research Online are protected by copyright, with all rights reserved unless indicated otherwise. They may be downloaded and/or printed for private study, or other acts as permitted by national copyright laws. The publisher or other rights holders may allow further reproduction and re-use of the full text version. This is indicated by the licence information on the White Rose Research Online record for the item. Takedown If you consider content in White Rose Research Online to be in breach of UK law, please notify us by emailing [email protected] including the URL of the record and the reason for the withdrawal request. [email protected] https://eprints.whiterose.ac.uk/ IMA Journal of Complex Networks (2017)Page 1 of 25 doi:10.1093/comnet/xxx000 Spin Statistics, Partition Functions and Network Entropy JIANJIA WANG∗ Department of Computer Science,University of York, York, YO10 5DD, UK ∗[email protected] RICHARD C. WILSON Department of Computer Science,University of York, York, YO10 5DD, UK AND EDWIN R. HANCOCK Department of Computer Science,University of York, York, YO10 5DD, UK [Received on 2 May 2017] This paper explores the thermodynamic characterization of networks using the heat bath analogy when the energy states are occupied under different spin statistics, specified by a partition function. -

Entropy: from the Boltzmann Equation to the Maxwell Boltzmann Distribution
Entropy: From the Boltzmann equation to the Maxwell Boltzmann distribution A formula to relate entropy to probability Often it is a lot more useful to think about entropy in terms of the probability with which different states are occupied. Lets see if we can describe entropy as a function of the probability distribution between different states. N! WN particles = n1!n2!....nt! stirling (N e)N N N W = = N particles n1 n2 nt n1 n2 nt (n1 e) (n2 e) ....(nt e) n1 n2 ...nt with N pi = ni 1 W = N particles n1 n2 nt p1 p2 ...pt takeln t lnWN particles = "#ni ln pi i=1 divide N particles t lnW1particle = "# pi ln pi i=1 times k t k lnW1particle = "k# pi ln pi = S1particle i=1 and t t S = "N k p ln p = "R p ln p NA A # i # i i i=1 i=1 ! Think about how this equation behaves for a moment. If any one of the states has a probability of occurring equal to 1, then the ln pi of that state is 0 and the probability of all the other states has to be 0 (the sum over all probabilities has to be 1). So the entropy of such a system is 0. This is exactly what we would have expected. In our coin flipping case there was only one way to be all heads and the W of that configuration was 1. Also, and this is not so obvious, the maximal entropy will result if all states are equally populated (if you want to see a mathematical proof of this check out Ken Dill’s book on page 85). -

Physical Biology
Physical Biology Kevin Cahill Department of Physics and Astronomy University of New Mexico, Albuquerque, NM 87131 i For Ginette, Mike, Sean, Peter, Mia, James, Dylan, and Christopher Contents Preface page v 1 Atoms and Molecules 1 1.1 Atoms 1 1.2 Bonds between atoms 1 1.3 Oxidation and reduction 5 1.4 Molecules in cells 6 1.5 Sugars 6 1.6 Hydrocarbons, fatty acids, and fats 8 1.7 Nucleotides 10 1.8 Amino acids 15 1.9 Proteins 19 Exercises 20 2 Metabolism 23 2.1 Glycolysis 23 2.2 The citric-acid cycle 23 3 Molecular devices 27 3.1 Transport 27 4 Probability and statistics 28 4.1 Mean and variance 28 4.2 The binomial distribution 29 4.3 Random walks 32 4.4 Diffusion as a random walk 33 4.5 Continuous diffusion 34 4.6 The Poisson distribution 36 4.7 The gaussian distribution 37 4.8 The error function erf 39 4.9 The Maxwell-Boltzmann distribution 40 Contents iii 4.10 Characteristic functions 41 4.11 Central limit theorem 42 Exercises 45 5 Statistical Mechanics 47 5.1 Probability and the Boltzmann distribution 47 5.2 Equilibrium and the Boltzmann distribution 49 5.3 Entropy 53 5.4 Temperature and entropy 54 5.5 The partition function 55 5.6 The Helmholtz free energy 57 5.7 The Gibbs free energy 58 Exercises 59 6 Chemical Reactions 61 7 Liquids 65 7.1 Diffusion 65 7.2 Langevin's Theory of Brownian Motion 67 7.3 The Einstein-Nernst relation 71 7.4 The Poisson-Boltzmann equation 72 7.5 Reynolds number 73 7.6 Fluid mechanics 76 8 Cell structure 77 8.1 Organelles 77 8.2 Membranes 78 8.3 Membrane potentials 80 8.4 Potassium leak channels 82 8.5 The Debye-H¨uckel equation 83 8.6 The Poisson-Boltzmann equation in one dimension 87 8.7 Neurons 87 Exercises 89 9 Polymers 90 9.1 Freely jointed polymers 90 9.2 Entropic force 91 10 Mechanics 93 11 Some Experimental Methods 94 11.1 Time-dependent perturbation theory 94 11.2 FRET 96 11.3 Fluorescence 103 11.4 Some mathematics 107 iv Contents References 109 Index 111 Preface Most books on biophysics hide the physics so as to appeal to a wide audi- ence of students. -
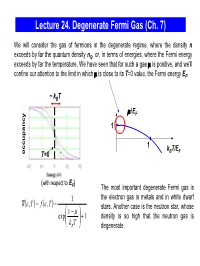
Lecture 24. Degenerate Fermi Gas (Ch
Lecture 24. Degenerate Fermi Gas (Ch. 7) We will consider the gas of fermions in the degenerate regime, where the density n exceeds by far the quantum density nQ, or, in terms of energies, where the Fermi energy exceeds by far the temperature. We have seen that for such a gas μ is positive, and we’ll confine our attention to the limit in which μ is close to its T=0 value, the Fermi energy EF. ~ kBT μ/EF 1 1 kBT/EF occupancy T=0 (with respect to E ) F The most important degenerate Fermi gas is 1 the electron gas in metals and in white dwarf nε()(),, T= f ε T = stars. Another case is the neutron star, whose ε⎛ − μ⎞ exp⎜ ⎟ +1 density is so high that the neutron gas is ⎝kB T⎠ degenerate. Degenerate Fermi Gas in Metals empty states ε We consider the mobile electrons in the conduction EF conduction band which can participate in the charge transport. The band energy is measured from the bottom of the conduction 0 band. When the metal atoms are brought together, valence their outer electrons break away and can move freely band through the solid. In good metals with the concentration ~ 1 electron/ion, the density of electrons in the electron states electron states conduction band n ~ 1 electron per (0.2 nm)3 ~ 1029 in an isolated in metal electrons/m3 . atom The electrons are prevented from escaping from the metal by the net Coulomb attraction to the positive ions; the energy required for an electron to escape (the work function) is typically a few eV. -

Chapter 13 Ideal Fermi
Chapter 13 Ideal Fermi gas The properties of an ideal Fermi gas are strongly determined by the Pauli principle. We shall consider the limit: k T µ,βµ 1, B � � which defines the degenerate Fermi gas. In this limit, the quantum mechanical nature of the system becomes especially important, and the system has little to do with the classical ideal gas. Since this chapter is devoted to fermions, we shall omit in the following the subscript ( ) that we used for the fermionic statistical quantities in the previous chapter. − 13.1 Equation of state Consider a gas ofN non-interacting fermions, e.g., electrons, whose one-particle wave- functionsϕ r(�r) are plane-waves. In this case, a complete set of quantum numbersr is given, for instance, by the three cartesian components of the wave vector �k and thez spin projectionm s of an electron: r (k , k , k , m ). ≡ x y z s Spin-independent Hamiltonians. We will consider only spin independent Hamiltonian operator of the type ˆ 3 H= �k ck† ck + d r V(r)c r†cr , �k � where thefirst and the second terms are respectively the kinetic and th potential energy. The summation over the statesr (whenever it has to be performed) can then be reduced to the summation over states with different wavevectork(p=¯hk): ... (2s + 1) ..., ⇒ r � �k where the summation over the spin quantum numberm s = s, s+1, . , s has been taken into account by the prefactor (2s + 1). − − 159 160 CHAPTER 13. IDEAL FERMI GAS Wavefunctions in a box. We as- sume that the electrons are in a vol- ume defined by a cube with sidesL x, Ly,L z and volumeV=L xLyLz. -

Recitation 8 Notes
3.024 Electrical, Optical, and Magnetic Properties of Materials Spring 2012 Recitation 8 Notes Overview 1. Electronic Band Diagram Review 2. Spin Review 3. Density of States 4. Fermi-Dirac Distribution 1. Electronic Band Diagram Review Considering 1D crystals with periodic potentials of the form: ( ) ∑ n a Here where is an integer, is the crystal lattice constant, and is the reciprocal space lattice constant. The Schrodinger equation for these systems has a general solution of the form: ( ) ∑ Upon substitution of this solution into Schrodinger’s equation, the following Central Equation is found: ( ) ∑ This is an eigenvalue/eigenvector equation where the eigenvalues are given by E and the eigenvectors by the coefficients Ck. By rewriting the above expression as a matrix equation, one can computationally solve for the energy eigenvalues for given values of k and produce a band diagram. ( ) ( ) ( ) ( ) [ ] [ ] The steps to solving this equation for just the energy eigenvalues is as follows: 1. Set values of equal to values of coefficients of the periodic potential of interest. 2. Truncate the infinite matrix to an matrix approximation suitable for the number of energy bands that is required for the problem to be investigated. If N is odd, set the center term to the Ck matrix coefficient. If N is even, an asymmetry will be introduced in the top band depending on whether the Ck matrix coefficient is placed to the left diagonal of the matrix center or the right diagonal of the matrix center. This asymmetry can be avoided by only using odd matrices or by solving both even matrices and superimposing the highest band energy solutions. -

Chapter 6. the Ising Model on a Square Lattice
Chapter 6. The Ising model on a square lattice 6.1 The basic Ising model The Ising model is a minimal model to capture the spontaneous emergence of macroscopic ferromagnetism from the interaction of individual microscopic magnetic dipole moments. Consider a two-dimensional system of spin 1=2 magnetic dipoles arrayed on a square L L lattice. At each lattice point .i; j /, the dipole can assume only one of two spin states, i;j 1. The dipoles interact with an externally applied constant magnetic field B, and the energyD˙ of a dipole interacting with the external field, Uext, is given by U .i;j / Hi;j ext D where H is a non-dimensionalized version of B. The energy of each spin (dipole) is lower when it is aligned with the external magnetic field. The dipoles also interact with their nearest neighbors. Due to quantum effects the energy of two spins in a ferromagnetic material is lower when they are aligned with each other1. As a model of this, it is assumed in the Ising model that the interaction energy for the dipole at .i; j / is given by Uint.i;j / Ji;j .i 1;j i 1;j i;j 1 i;j 1/ D C C C C C where J is a non-dimensionalized coupling strength. Summing over all sites, we get the total energy for a configuration given by E L L J L L U./ H i;j i;j .i 1;j i 1;j i;j 1 i;j 1/: E D 2 C C C C C i 1 j 1 i 1 j 1 XD XD XD XD The second sum is divided by two to account for each interaction being counted twice. -

The Physics of Electron Degenerate Matter in White Dwarf Stars
Western Michigan University ScholarWorks at WMU Master's Theses Graduate College 6-2008 The Physics of Electron Degenerate Matter in White Dwarf Stars Subramanian Vilayur Ganapathy Follow this and additional works at: https://scholarworks.wmich.edu/masters_theses Part of the Physics Commons Recommended Citation Ganapathy, Subramanian Vilayur, "The Physics of Electron Degenerate Matter in White Dwarf Stars" (2008). Master's Theses. 4251. https://scholarworks.wmich.edu/masters_theses/4251 This Masters Thesis-Open Access is brought to you for free and open access by the Graduate College at ScholarWorks at WMU. It has been accepted for inclusion in Master's Theses by an authorized administrator of ScholarWorks at WMU. For more information, please contact [email protected]. THE PHYSICS OF ELECTRON DEGENERATE MATTER IN WHITE DWARF STARS by Subramanian Vilayur Ganapathy A Thesis Submitted to the Faculty of The Graduate College in partial fulfillment of the requirements forthe Degree of Master of Arts Department of Physics Western MichiganUniversity Kalamazoo, Michigan June 2008 Copyright by Subramanian Vilayur Ganapathy 2008 ACKNOWLEDGMENTS I wish to express my infinite gratitude and sincere appreciation to my advisor, Professor Kirk Korista, for suggesting the topic of the thesis, and for all of his direction, encouragement and great help throughout the course of this research. I would also like to express my gratitude to the other members of my committee, Professor Dean Halderson and Professor Clement Bums for their valuable advice and help. Subramanian Vilayur Ganapathy 11 THE PHYSICS OF ELECTRON DEGENERATE MATTER IN WHITE DWARF STARS Subramanian Vilayur Ganapathy , M.A. Western Michigan University, 2008 White dwarfs are the remnant cores of medium and low mass stars with initial mass less than 8 times the mass of our sun. -

Lecture 7: Ensembles
Matthew Schwartz Statistical Mechanics, Spring 2019 Lecture 7: Ensembles 1 Introduction In statistical mechanics, we study the possible microstates of a system. We never know exactly which microstate the system is in. Nor do we care. We are interested only in the behavior of a system based on the possible microstates it could be, that share some macroscopic proporty (like volume V ; energy E, or number of particles N). The possible microstates a system could be in are known as the ensemble of states for a system. There are dierent kinds of ensembles. So far, we have been counting microstates with a xed number of particles N and a xed total energy E. We dened as the total number microstates for a system. That is (E; V ; N) = 1 (1) microstatesk withsaXmeN ;V ;E Then S = kBln is the entropy, and all other thermodynamic quantities follow from S. For an isolated system with N xed and E xed the ensemble is known as the microcanonical 1 @S ensemble. In the microcanonical ensemble, the temperature is a derived quantity, with T = @E . So far, we have only been using the microcanonical ensemble. 1 3 N For example, a gas of identical monatomic particles has (E; V ; N) V NE 2 . From this N! we computed the entropy S = kBln which at large N reduces to the Sackur-Tetrode formula. 1 @S 3 NkB 3 The temperature is T = @E = 2 E so that E = 2 NkBT . Also in the microcanonical ensemble we observed that the number of states for which the energy of one degree of freedom is xed to "i is (E "i) "i/k T (E " ).