Developing Perspectives on Molluscan Shells, Part 1: Introduction and Molecular Biology
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Impact Damage and Repair in Shells of the Limpet Patella Vulgata David Taylor
© 2016. Published by The Company of Biologists Ltd | Journal of Experimental Biology (2016) 219, 3927-3935 doi:10.1242/jeb.149880 RESEARCH ARTICLE Impact damage and repair in shells of the limpet Patella vulgata David Taylor ABSTRACT can be a major cause of lethal damage. Shanks and Wright (1986) Experiments and observations were carried out to investigate the demonstrated the destructive power of wave-borne missiles by response of the Patella vulgata limpet shell to impact. Dropped-weight setting up targets to record impacts in a given area. They studied four impact tests created damage that usually took the form of a hole in the limpet species, finding that they were much more likely to be lost in shell’s apex. Similar damage was found to occur naturally, a location where there were many movable rocks and pebbles presumably as a result of stones propelled by the sea during compared with a location consisting largely of solid rock mass. storms. Apex holes were usually fatal, but small holes were Examining the populations over a one year period, and making a sometimes repaired, and the repaired shell was as strong as the number of assumptions about size distributions and growth rates, original, undamaged shell. The impact strength (energy to failure) of they estimated that 47% of shells were destroyed in the former shells tested in situ was found to be 3.4-times higher than that of location, compared with 7% in the latter (Shanks and Wright, 1986). empty shells found on the beach. Surprisingly, strength was not Another study (Cadée, 1999) reported that impact damage by ice affected by removing the shell from its home location, or by removing blocks and stones was the major cause of damage to the limpet the limpet from the shell and allowing the shell to dry out. -

Some Aspects of the Biology of Three Northwestern Atlantic Chitons
University of New Hampshire University of New Hampshire Scholars' Repository Doctoral Dissertations Student Scholarship Spring 1978 SOME ASPECTS OF THE BIOLOGY OF THREE NORTHWESTERN ATLANTIC CHITONS: TONICELLA RUBRA, TONICELLA MARMOREA, AND ISCHNOCHITON ALBUS (MOLLUSCA: POLYPLACOPHORA) PAUL DAVID LANGER University of New Hampshire, Durham Follow this and additional works at: https://scholars.unh.edu/dissertation Recommended Citation LANGER, PAUL DAVID, "SOME ASPECTS OF THE BIOLOGY OF THREE NORTHWESTERN ATLANTIC CHITONS: TONICELLA RUBRA, TONICELLA MARMOREA, AND ISCHNOCHITON ALBUS (MOLLUSCA: POLYPLACOPHORA)" (1978). Doctoral Dissertations. 2329. https://scholars.unh.edu/dissertation/2329 This Dissertation is brought to you for free and open access by the Student Scholarship at University of New Hampshire Scholars' Repository. It has been accepted for inclusion in Doctoral Dissertations by an authorized administrator of University of New Hampshire Scholars' Repository. For more information, please contact [email protected]. INFORMATION TO USERS This material was produced from a microfilm copy of the original document. While the most advanced technological means to photograph and reproduce this document have been used, the quality is heavily dependent upon the quality of the original submitted. The following explanation of techniques is provided to help you understand markings or patterns which may appear on this reproduction. 1.The sign or "target" for pages apparently lacking from the document photographed is "Missing Page(s)". If it was possible to obtain the missing page(s) or section, they are spliced into the film along with adjacent pages. This may have necessitated cutting thru an image and duplicating adjacent pages to insure you complete continuity. 2. When an image on the film is obliterated with a large round black mark, it is an indication that the photographer suspected that the copy may have moved during exposure and thus cause a blurred image. -
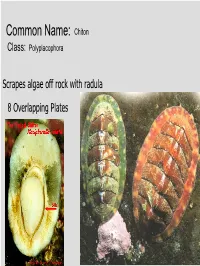
Common Name: Chiton Class: Polyplacophora
Common Name: Chiton Class: Polyplacophora Scrapes algae off rock with radula 8 Overlapping Plates Phylum? Mollusca Class? Gastropoda Common name? Brown sea hare Class? Scaphopoda Common name? Tooth shell or tusk shell Mud Tentacle Foot Class? Gastropoda Common name? Limpet Phylum? Mollusca Class? Bivalvia Class? Gastropoda Common name? Brown sea hare Phylum? Mollusca Class? Gastropoda Common name? Nudibranch Class? Cephalopoda Cuttlefish Octopus Squid Nautilus Phylum? Mollusca Class? Gastropoda Most Bivalves are Filter Feeders A B E D C • A: Mantle • B: Gill • C: Mantle • D: Foot • E: Posterior adductor muscle I.D. Green: Foot I.D. Red Gills Three Body Regions 1. Head – Foot 2. Visceral Mass 3. Mantle A B C D • A: Radula • B: Mantle • C: Mouth • D: Foot What are these? Snail Radulas Dorsal HingeA Growth line UmboB (Anterior) Ventral ByssalC threads Mussel – View of Outer Shell • A: Hinge • B: Umbo • C: Byssal threads Internal Anatomy of the Bay Mussel A B C D • A: Labial palps • B: Mantle • C: Foot • D: Byssal threads NacreousB layer Posterior adductorC PeriostracumA muscle SiphonD Mantle Byssal threads E Internal Anatomy of the Bay Mussel • A: Periostracum • B: Nacreous layer • C: Posterior adductor muscle • D: Siphon • E: Mantle Byssal gland Mantle Gill Foot Labial palp Mantle Byssal threads Gill Byssal gland Mantle Foot Incurrent siphon Byssal Labial palp threads C D B A E • A: Foot • B: Gills • C: Posterior adductor muscle • D: Excurrent siphon • E: Incurrent siphon Heart G F H E D A B C • A: Foot • B: Gills • C: Mantle • D: Excurrent siphon • E: Incurrent siphon • F: Posterior adductor muscle • G: Labial palps • H: Anterior adductor muscle Siphon or 1. -

Haliotis Asinina) in Coastal Waters of Thailand Determined Using Microsatellite Markers
Mar. Biotechnol. 6, 604–611, 2004 DOI: 10.1007/s10126-004-2300-5 Ó 2005 Springer Science+Business Media, Inc. Population Structure of Tropical Abalone (Haliotis asinina) in Coastal Waters of Thailand Determined Using Microsatellite Markers S. Tang,1 A. Tassanakajon,1 S. Klinbunga,2 P. Jarayabhand,3,4 and P. Menasveta2,4 1Department of Biochemistry, Faculty of Science, Chulalongkorn University, Bangkok 10330, Thailand 2Marine Biotechnology Research Unit, National Center for Genetic Engineering and Biotechnology (BIOTEC), National Science and Technology Development Agency, Pathumthani 12120, Thailand 3Aquatic Resources Research Institute, Chulalongkorn University, Bangkok 10330, Thailand 4Department of Marine Science, Faculty of Science, Chulalongkorn University, Bangkok 10330, Thailand Abstract: Three partial genomic libraries were constructed from genomic DNA of the tropical abalone (Haliotis asinina) that was digested with AluI, vortexed/sonicated, and digested with mixed enzyme (AluI, HincII, and RsaI). The libraries yielded 0.02%, 0.42%, and 1.46% positive microsatellite-containing clones, respectively. Eleven clones each of perfect, imperfect, and compound microsatellites were isolated. Ten primer pairs (CU- Has1–CUHas10) were analyzed to evaluate their polymorphic level. The numbers of alleles per locus, observed heterozygosity (H0), and expected heterozygosity (He) ranged from 3 to 26 alleles, and varied between 0.27 and 0.85 and between 0.24 and 0.93, respectively. Three microsatellite loci (CUHas2, CUHas3, and CUHas8) were further used for examination of genetic diversity and differentiation of natural H. asinina in coastal waters of Thailand. Genetic variabilities in terms of the effective number of alleles (ne), H0, and He were higher in 2 samples from the Gulf of Thailand (ne = 9.37, 7.66; H0 = 0.62, 0.78; and He = 0.87, 0.86) than those of one sample (ne = 6.04; H0 = 0.58; and He = 0.62) derived from the Andaman Sea. -

Marine Invertebrate Field Guide
Marine Invertebrate Field Guide Contents ANEMONES ....................................................................................................................................................................................... 2 AGGREGATING ANEMONE (ANTHOPLEURA ELEGANTISSIMA) ............................................................................................................................... 2 BROODING ANEMONE (EPIACTIS PROLIFERA) ................................................................................................................................................... 2 CHRISTMAS ANEMONE (URTICINA CRASSICORNIS) ............................................................................................................................................ 3 PLUMOSE ANEMONE (METRIDIUM SENILE) ..................................................................................................................................................... 3 BARNACLES ....................................................................................................................................................................................... 4 ACORN BARNACLE (BALANUS GLANDULA) ....................................................................................................................................................... 4 HAYSTACK BARNACLE (SEMIBALANUS CARIOSUS) .............................................................................................................................................. 4 CHITONS ........................................................................................................................................................................................... -

Status and Protection of Globally Threatened Species in the Caucasus
STATUS AND PROTECTION OF GLOBALLY THREATENED SPECIES IN THE CAUCASUS CEPF Biodiversity Investments in the Caucasus Hotspot 2004-2009 Edited by Nugzar Zazanashvili and David Mallon Tbilisi 2009 The contents of this book do not necessarily reflect the views or policies of CEPF, WWF, or their sponsoring organizations. Neither the CEPF, WWF nor any other entities thereof, assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, product or process disclosed in this book. Citation: Zazanashvili, N. and Mallon, D. (Editors) 2009. Status and Protection of Globally Threatened Species in the Caucasus. Tbilisi: CEPF, WWF. Contour Ltd., 232 pp. ISBN 978-9941-0-2203-6 Design and printing Contour Ltd. 8, Kargareteli st., 0164 Tbilisi, Georgia December 2009 The Critical Ecosystem Partnership Fund (CEPF) is a joint initiative of l’Agence Française de Développement, Conservation International, the Global Environment Facility, the Government of Japan, the MacArthur Foundation and the World Bank. This book shows the effort of the Caucasus NGOs, experts, scientific institutions and governmental agencies for conserving globally threatened species in the Caucasus: CEPF investments in the region made it possible for the first time to carry out simultaneous assessments of species’ populations at national and regional scales, setting up strategies and developing action plans for their survival, as well as implementation of some urgent conservation measures. Contents Foreword 7 Acknowledgments 8 Introduction CEPF Investment in the Caucasus Hotspot A. W. Tordoff, N. Zazanashvili, M. Bitsadze, K. Manvelyan, E. Askerov, V. Krever, S. Kalem, B. Avcioglu, S. Galstyan and R. Mnatsekanov 9 The Caucasus Hotspot N. -

Os Nomes Galegos Dos Moluscos
A Chave Os nomes galegos dos moluscos 2017 Citación recomendada / Recommended citation: A Chave (2017): Nomes galegos dos moluscos recomendados pola Chave. http://www.achave.gal/wp-content/uploads/achave_osnomesgalegosdos_moluscos.pdf 1 Notas introdutorias O que contén este documento Neste documento fornécense denominacións para as especies de moluscos galegos (e) ou europeos, e tamén para algunhas das especies exóticas máis coñecidas (xeralmente no ámbito divulgativo, por causa do seu interese científico ou económico, ou por seren moi comúns noutras áreas xeográficas). En total, achéganse nomes galegos para 534 especies de moluscos. A estrutura En primeiro lugar preséntase unha clasificación taxonómica que considera as clases, ordes, superfamilias e familias de moluscos. Aquí apúntase, de maneira xeral, os nomes dos moluscos que hai en cada familia. A seguir vén o corpo do documento, onde se indica, especie por especie, alén do nome científico, os nomes galegos e ingleses de cada molusco (nalgún caso, tamén, o nome xenérico para un grupo deles). Ao final inclúese unha listaxe de referencias bibliográficas que foron utilizadas para a elaboración do presente documento. Nalgunhas desas referencias recolléronse ou propuxéronse nomes galegos para os moluscos, quer xenéricos quer específicos. Outras referencias achegan nomes para os moluscos noutras linguas, que tamén foron tidos en conta. Alén diso, inclúense algunhas fontes básicas a respecto da metodoloxía e dos criterios terminolóxicos empregados. 2 Tratamento terminolóxico De modo moi resumido, traballouse nas seguintes liñas e cos seguintes criterios: En primeiro lugar, aprofundouse no acervo lingüístico galego. A respecto dos nomes dos moluscos, a lingua galega é riquísima e dispomos dunha chea de nomes, tanto específicos (que designan un único animal) como xenéricos (que designan varios animais parecidos). -

The Malacological Society of London
ACKNOWLEDGMENTS This meeting was made possible due to generous contributions from the following individuals and organizations: Unitas Malacologica The program committee: The American Malacological Society Lynn Bonomo, Samantha Donohoo, The Western Society of Malacologists Kelly Larkin, Emily Otstott, Lisa Paggeot David and Dixie Lindberg California Academy of Sciences Andrew Jepsen, Nick Colin The Company of Biologists. Robert Sussman, Allan Tina The American Genetics Association. Meg Burke, Katherine Piatek The Malacological Society of London The organizing committee: Pat Krug, David Lindberg, Julia Sigwart and Ellen Strong THE MALACOLOGICAL SOCIETY OF LONDON 1 SCHEDULE SUNDAY 11 AUGUST, 2019 (Asilomar Conference Center, Pacific Grove, CA) 2:00-6:00 pm Registration - Merrill Hall 10:30 am-12:00 pm Unitas Malacologica Council Meeting - Merrill Hall 1:30-3:30 pm Western Society of Malacologists Council Meeting Merrill Hall 3:30-5:30 American Malacological Society Council Meeting Merrill Hall MONDAY 12 AUGUST, 2019 (Asilomar Conference Center, Pacific Grove, CA) 7:30-8:30 am Breakfast - Crocker Dining Hall 8:30-11:30 Registration - Merrill Hall 8:30 am Welcome and Opening Session –Terry Gosliner - Merrill Hall Plenary Session: The Future of Molluscan Research - Merrill Hall 9:00 am - Genomics and the Future of Tropical Marine Ecosystems - Mónica Medina, Pennsylvania State University 9:45 am - Our New Understanding of Dead-shell Assemblages: A Powerful Tool for Deciphering Human Impacts - Sue Kidwell, University of Chicago 2 10:30-10:45 -

Comparative Neuroanatomy of Mollusks and Nemerteans in the Context of Deep Metazoan Phylogeny
Comparative Neuroanatomy of Mollusks and Nemerteans in the Context of Deep Metazoan Phylogeny Von der Fakultät für Mathematik, Informatik und Naturwissenschaften der RWTH Aachen University zur Erlangung des akademischen Grades einer Doktorin der Naturwissenschaften genehmigte Dissertation vorgelegt von Diplom-Biologin Simone Faller aus Frankfurt am Main Berichter: Privatdozent Dr. Rudolf Loesel Universitätsprofessor Dr. Peter Bräunig Tag der mündlichen Prüfung: 09. März 2012 Diese Dissertation ist auf den Internetseiten der Hochschulbibliothek online verfügbar. Contents 1 General Introduction 1 Deep Metazoan Phylogeny 1 Neurophylogeny 2 Mollusca 5 Nemertea 6 Aim of the thesis 7 2 Neuroanatomy of Minor Mollusca 9 Introduction 9 Material and Methods 10 Results 12 Caudofoveata 12 Scutopus ventrolineatus 12 Falcidens crossotus 16 Solenogastres 16 Dorymenia sarsii 16 Polyplacophora 20 Lepidochitona cinerea 20 Acanthochitona crinita 20 Scaphopoda 22 Antalis entalis 22 Entalina quinquangularis 24 Discussion 25 Structure of the brain and nerve cords 25 Caudofoveata 25 Solenogastres 26 Polyplacophora 27 Scaphopoda 27 i CONTENTS Evolutionary considerations 28 Relationship among non-conchiferan molluscan taxa 28 Position of the Scaphopoda within Conchifera 29 Position of Mollusca within Protostomia 30 3 Neuroanatomy of Nemertea 33 Introduction 33 Material and Methods 34 Results 35 Brain 35 Cerebral organ 38 Nerve cords and peripheral nervous system 38 Discussion 38 Peripheral nervous system 40 Central nervous system 40 In search for the urbilaterian brain 42 4 General Discussion 45 Evolution of higher brain centers 46 Neuroanatomical glossary and data matrix – Essential steps toward a cladistic analysis of neuroanatomical data 49 5 Summary 53 6 Zusammenfassung 57 7 References 61 Danksagung 75 Lebenslauf 79 ii iii 1 General Introduction Deep Metazoan Phylogeny The concept of phylogeny follows directly from the theory of evolution as published by Charles Darwin in The origin of species (1859). -

Evolution of Large Body Size in Abalones (Haliotis): Patterns and Implications
Paleobiology, 31(4), 2005, pp. 591±606 Evolution of large body size in abalones (Haliotis): patterns and implications James A. Estes, David R. Lindberg, and Charlie Wray Abstract.ÐKelps and other ¯eshy macroalgaeÐdominant reef-inhabiting organisms in cool seasÐ may have radiated extensively following late Cenozoic polar cooling, thus triggering a chain of evolutionary change in the trophic ecology of nearshore temperate ecosystems. We explore this hypothesis through an analysis of body size in the abalones (Gastropoda; Haliotidae), a widely distributed group in modern oceans that displays a broad range of body sizes and contains fossil representatives from the late Cretaceous (60±75 Ma). Geographic analysis of maximum shell length in living abalones showed that small-bodied species, while most common in the Tropics, have a cosmopolitan distribution, whereas large-bodied species occur exclusively in cold-water ecosys- tems dominated by kelps and other macroalgae. The phylogeography of body size evolution in extant abalones was assessed by constructing a molecular phylogeny in a mix of large and small species obtained from different regions of the world. This analysis demonstrates that small body size is the plesiomorphic state and largeness has likely arisen at least twice. Finally, we compiled data on shell length from the fossil record to determine how (slowly or suddenly) and when large body size arose in the abalones. These data indicate that large body size appears suddenly at the Miocene/Pliocene boundary. Our ®ndings support the view that ¯eshy-algal dominated ecosys- tems radiated rapidly in the coastal oceans with the onset of the most recent glacial age. -

Microgasterópodos Asociados Con El Banco Natural De La Pepitona Arca
Ciencias Marinas (2005), 31(1A): 119–124 http://dx.doi.org/10.7773/cm.v31i11.71 Nota de Investigación/Research Note Microgasterópodos asociados con el banco natural de la pepitona Arca zebra (Swainson, 1833; Mollusca: Bivalvia) ubicado en la localidad de Chacopata, Estado Sucre, Venezuela Microgastropods associated with the natural bank of Arca zebra (Swainson, 1833; Mollusca: Bivalvia) located in Chacopata, Sucre State, Venezuela Samuel Narciso1 Antulio Prieto-Arcas2 Vanessa Acosta-Balbás2* 1 Fundación para la Defensa de la Naturaleza (FUDENA) Estación de Chichiriviche Estado Falcón, Venezuela E-mail: [email protected] 2 Departamento de Biología, Escuela de Ciencias Universidad de Oriente Apartado 245 Cumaná, Estado Sucre, Venezuela * E-mail: [email protected] Recibido en febrero de 2004; aceptado en agosto de 2004 Resumen El objetivo de este estudio fue analizar la taxonomía de los microgasterópodos asociados con el bivalvo Arca zebra, un importante recurso pesquero del nororiente de Venezuela. Las muestras se obtuvieron con rastras metálicas en la localidad de Chacopata, Estado Sucre, Venezuela. Se recolectaron un total de 381 gasterópodos pertenecientes a 25 especies incluidas en 12 familias, de las cuales las más diversas, de acuerdo con el número de especies, fueron Marginellidae (4), Collumbelidae (4) y Fisurellidae (3). Del total de las especies recolectadas, siete (Marginella haematita, Lucapina sowerby, Cantharus cancellarius, Crassispira tampanensis, Pyrgocytara coxi, Monilispira leusosyma y Terebra nasulla) representan nuevos registros para Venezuela, aunque pueden catalogarse como típicas del Atlántico tropical occidental. Palabras clave: micromoluscos, bivalvos, costas de Venezuela. Abstract The objective of this study was to analyze the taxonomy of microgastropods associated with the bivalve Arca zebra, an important fishing resource of the northern coast of Venezuela. -

Download Preprint
1 Mobilising molluscan models and genomes in biology 2 Angus Davison1 and Maurine Neiman2 3 1. School of Life Sciences, University Park, University of Nottingham, NG7 2RD, UK 4 2. Department of Biology, University of Iowa, Iowa City, IA, USA and Department of Gender, 5 Women's, and Sexuality Studies, University of Iowa, Iowa, City, IA, USA 6 Abstract 7 Molluscs are amongst the most ancient, diverse, and important of all animal taxa. Even so, 8 no individual mollusc species has emerged as a broadly applied model system in biology. 9 We here make the case that both perceptual and methodological barriers have played a role 10 in the relative neglect of molluscs as research organisms. We then summarize the current 11 application and potential of molluscs and their genomes to address important questions in 12 animal biology, and the state of the field when it comes to the availability of resources such 13 as genome assemblies, cell lines, and other key elements necessary to mobilising the 14 development of molluscan model systems. We conclude by contending that a cohesive 15 research community that works together to elevate multiple molluscan systems to ‘model’ 16 status will create new opportunities in addressing basic and applied biological problems, 17 including general features of animal evolution. 18 Introduction 19 Molluscs are globally important as sources of food, calcium and pearls, and as vectors of 20 human disease. From an evolutionary perspective, molluscs are notable for their remarkable 21 diversity: originating over 500 million years ago, there are over 70,000 extant mollusc 22 species [1], with molluscs present in virtually every ecosystem.