Benefit–Risk Profile of Sphingosine-1-Phosphate
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Therapeutic Use of a Selective S1P1 Receptor Modulator Ponesimod in Autoimmune Diabetes
Therapeutic Use of a Selective S1P1 Receptor Modulator Ponesimod in Autoimmune Diabetes Sylvaine You1,2, Luca Piali3, Chantal Kuhn1,2, Beat Steiner3, Virginia Sauvaget1,2, Fabrice Valette1,2, Martine Clozel3, Jean-Franc¸ois Bach1,2, Lucienne Chatenoud1,2* 1 Universite´ Paris Descartes, Sorbonne Paris Cite´, Paris, France, 2 Institut National de la Sante´ et de la Recherche Me´dicale, Unite´ 1013, Paris, France, 3 Actelion Pharmaceuticals Ltd, Allschwil, Switzerland Abstract In the present study, we investigated the therapeutic potential of a selective S1P1 receptor modulator, ponesimod, to protect and reverse autoimmune diabetes in non-obese diabetic (NOD) mice. Ponesimod was administered orally to NOD mice starting at 6, 10, 13 and 16 weeks of age up to 35 weeks of age or to NOD mice showing recent onset diabetes. Peripheral blood and spleen B and T cell counts were significantly reduced after ponesimod administration. In pancreatic lymph nodes, B lymphocytes were increased and expressed a transitional 1-like phenotype. Chronic oral ponesimod treatment efficiently prevented autoimmune diabetes in 6, 10 and 16 week-old pre-diabetic NOD mice. Treatment withdrawal led to synchronized disease relapse. Ponesimod did not inhibit the differentiation of autoreactive T cells as assessed by adoptive transfer of lymphocytes from treated disease-free NOD mice. In addition, it did not affect the migration, proliferation and activation of transgenic BDC2.5 cells into the target tissue. However, ponesimod inhibited spreading of the T cell responses to islet-specific glucose-6-phosphatase catalytic subunit-related protein (IGRP). Treatment of diabetic NOD mice with ponesimod induced disease remission. However, here again, upon treatment cessation, the disease rapidly recurred. -

The Change of Fingolimod Patient Profiles Over Time
Journal of Personalized Medicine Article The Change of Fingolimod Patient Profiles over Time: A Descriptive Analysis of Two Non-Interventional Studies PANGAEA and PANGAEA 2.0 Tjalf Ziemssen 1,* and Ulf Schulze-Topphoff 2 1 Zentrum für Klinische Neurowissenschaften, Universitätsklinikum Carl Gustav Carus, D-01307 Dresden, Germany 2 Novartis Pharma GmbH, D-90429 Nuremberg, Germany; [email protected] * Correspondence: [email protected] Abstract: (1) Background: Fingolimod (Gilenya®) was the first oral treatment for patients with relapsing-remitting multiple sclerosis (RRMS). Since its approval, the treatment landscape has changed enormously. (2) Methods: Data of PANGAEA and PANGAEA 2.0, two German real-world studies, were descriptively analysed for possible evolution of patient profiles and treatment behavior. Both are prospective, multi-center, non-interventional, long-term studies on fingolimod use in RRMS in real life. Data of 4229 PANGAEA patients (recruited 2011–2013) and 2441 PANGAEA 2.0 patients (recruited 2015–2018) were available. Baseline data included demographics, RRMS characteristics and disease severity. (3) Results: The mean age of PANGAEA and PANGAEA 2.0 patients was similar (38.8 vs. 39.2 years). Patients in PANGAEA 2.0 had shorter disease duration (7.1 vs. 8.2 years) and fewer relapses in the year before baseline (1.2 vs. 1.6). Disease severity at baseline estimated by Citation: Ziemssen, T.; EDSS and SDMT was lower in PANGAEA 2.0 patients compared to PANGAEA (EDSS difference Schulze-Topphoff, U. The Change of 1.0 points; SDMT difference 3.3 points). (4) Conclusions: The results hint at an influence of changes in Fingolimod Patient Profiles over the treatment guidelines and the label on fingolimod patients profiles over time. -

Sphingosine-1-Phosphate: Its Pharmacological Regulation and the Treatment of Multiple Sclerosis: a Review Article
biomedicines Review Sphingosine-1-Phosphate: Its Pharmacological Regulation and the Treatment of Multiple Sclerosis: A Review Article Stanley Cohan, Elisabeth Lucassen, Kyle Smoot, Justine Brink and Chiayi Chen * Providence Multiple Sclerosis Center, Providence Brain and Spine Institute, Providence St, Vincent Medical Center, Portland, OR 97225, USA; [email protected] (S.C.); [email protected] (E.L.); [email protected] (K.S.); [email protected] (J.B.) * Correspondence: [email protected]; Tel.: +1-503-216-1012 Received: 27 May 2020; Accepted: 15 July 2020; Published: 18 July 2020 Abstract: Sphingosine-1-phosphate (S1P), via its G-protein-coupled receptors, is a signaling molecule with important regulatory properties on numerous, widely varied cell types. Five S1P receptors (S1PR1-5) have been identified, each with effects determined by their unique G-protein-driven downstream pathways. The discovery that lymphocyte egress from peripheral lymphoid organs is promoted by S1P via S1PR-1 stimulation led to the development of pharmacological agents which are S1PR antagonists. These agents promote lymphocyte sequestration and reduce lymphocyte-driven inflammatory damage of the central nervous system (CNS) in animal models, encouraging their examination of efficacy in the treatment of multiple sclerosis (MS). Preclinical research has also demonstrated direct protective effects of S1PR antagonists within the CNS, by modulation of S1PRs, particularly S1PR-1 and S1PR-5, and possibly S1PR-2, independent of effects upon lymphocytes. Three of these agents, fingolimod, siponimod and ozanimod have been approved, and ponesimod has been submitted for regulatory approval. In patients with MS, these agents reduce relapse risk, sustained disability progression, magnetic resonance imaging markers of disease activity, and whole brain and/or cortical and deep gray matter atrophy. -

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 22-527 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number 22-527 Priority or Standard P Submit Date 12-21-2009 Received Date 12-21-2009 PDUFA Goal Date 9-21-2010 Division / Office FDA/ODE1 Reviewer Name Heather D. Fitter Review Completion Date August 26, 2010 Established Name Fingolimod (Proposed) Trade Name Gilenya Therapeutic Class S1P receptor modulator Applicant Novartis Formulation(s) Oral tablets Dosing Regimen 0.5 mg Indication(s) To reduce the frequency of relapses and delay the progression of disability in relapsing MS Intended Population(s) Relapsing Multiple Sclerosis Template Version: March 6, 2009 Clinical Review Heather D. Fitter, M.D. NDA 22-527 Fingolimod Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT......................................... 7 1.1 Recommendation on Regulatory Action ............................................................. 7 1.2 Risk Benefit Assessment.................................................................................... 8 2 INTRODUCTION AND REGULATORY BACKGROUND ........................................ 8 2.1 Product Information ............................................................................................ 9 2.2 Table of Currently Available Treatments for the Proposed Indication................. 9 2.3 Availability of Proposed Active Ingredient in the United States ........................ 12 2.4 Important Safety Issues with Consideration to Related Drugs......................... -

Oral MS Disease-Modifying Therapies C21142-A
Drug and Biologic Coverage Criteria Effective Date: 05/01/2019 Last P&T Approval/Version: 07/28/2021 Next Review Due By: 08/2022 Policy Number: C21142-A Oral MS Disease-Modifying Therapies PRODUCTS AFFECTED Mayzent (siponimod), Aubagio (teriflunomide), Gilenya (fingolimod), Mavenclad (cladribine), Tecfidera (dimethyl fumarate), Vumerity (diroximel fumarate), Bafiertam (monomethyl fumarate),dimethyl fumarate, Zeposia (ozanimod), Ponvory (ponesimod) COVERAGE POLICY Coverage for services, procedures, medical devices, and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Coverage Guideline must be read in its entirety to determine coverage eligibility, if any. This Coverage Guideline provides information related to coverage determinations only and does not imply that a service or treatment is clinically appropriate or inappropriate. The provider and the member are responsible for all decisions regarding the appropriateness of care. Providers should provide Molina Healthcare complete medical rationale when requesting any exceptions to these guidelines Documentation Requirements: Molina Healthcare reserves the right to require that additional documentation be made available as part of its coverage determination; quality improvement; and fraud; waste and abuse prevention processes. Documentation required may include, but is not limited to, patient records, test results and credentials of the provider ordering or performing a drug or service. Molina Healthcare may deny reimbursement or take additional appropriate action if the documentation provided does not support the initial determination that the drugs or services were medically necessary, not investigational or experimental, and otherwise within the scope of benefits afforded to the member, and/or the documentation demonstrates a pattern of billing or other practice that is inappropriate or excessive DIAGNOSIS: Multiple Sclerosis REQUIRED MEDICAL INFORMATION: A. -
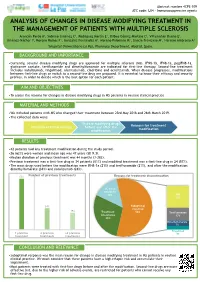
Analysis of Changes in Disease Modifying Treatment in The
Abstract number 4CPS-109 ATC code: L04 - Immunosuppressive agents ANALYSIS OF CHANGES IN DISEASE MODIFYING TREATMENT IN THE MANAGEMENT OF PATIENTS WITH MULTIPLE SCLEROSIS Arancón Pardo A1, Sobrino Jiménez C1, Rodríguez Martín E1, Bilbao Gómez-Martino C1, Villamañán Bueno E1, Jiménez-Nácher I1, Moreno Ramos F1, González Fernández A1, Moreno Palomino M1, García-Trevijano M1, Herrero Ambrosio A1 1Hospital Universitario La Paz, Pharmacy Department, Madrid, Spain. BACKGROUND AND IMPORTANCE •Currently, several disease modifying drugs are approved for multiple sclerosis (MS). IFNβ-1b, IFNβ-1a, pegIFNβ-1a, glatiramer acetate, teriflunomide and dimethylfumarate are indicated for first-line therapy. Second-line treatment includes natalizumab, fingolimod, alemtuzumab, cladribine and ocrelizumab. When disease progresses, modifications between first-line drugs or switch to a second-line drug are proposed. It is essential to know their efficacy and security profiles, in order to decide which is the best option for each patient. AIM AND OBJECTIVES •To assess the reasons for changes in disease modifying drugs in MS patients in routine clinical practice. MATERIAL AND METHODS •We included patients with MS who changed their treatment between 23rd May 2018 and 26th March 2019. •The collected data were: Disease modifying drugs Reasons for treatment Duration of initial therapy before and after the modification modification RESULTS •42 patients had any treatment modification during the study period. •26 (62%) were women and mean age was 47 years (SD 9.3). •Median duration of previous treatment was 44 months (3-282). •Previous treatment was a first-line drug in 34 patients (81%) and modified treatment was a first-line drug in 24 (57%). -

Patient Focused Disease State and Assistance Programs
Patient Focused Disease State and Assistance Programs Medication Medication Toll-free Brand (Generic) Website number Additional Resources Allergy/Asthma Xolair (omalizumab) xolair.com 1-866-4-XOLAIR lung.org Cardiovascular Pradaxa (dabigatran) pradaxa.com 877-481-5332 heart.org Praluent (alirocumab) praluent.com 844-PRALUENT thefhfoundation.org Repatha (evolocumab) repatha.com 844-REPATHA Tikosyn (dofetilide) tikosyn.com 800-879-3477 Crohn’s Disease Cimzia (certolizumab pegol) cimzia.com 866-4-CIMZIA crohnsandcolitis.com Humira (adalimumab) humira.com 800-4-HUMIRA crohnsforum.com Stelara (ustekinumab) stelarainfo.com 877-STELARA Dermatology Cosentyx (secukinumab) cosentyx.com 844-COSENTYX psoriasis.org Dupixent (dupilumab) dupixent.com 844-DUPIXENT nationaleczema.org Enbrel (etanercept) enbrel.com 888-4-ENBREL Humira (adalimumab) humira.com 800-4-HUMIRA Otezla (apremilast) otezla.com 844-4-OTEZLA Stelara (ustekinumab) stelarainfo.com 877-STELARA Taltz (ixekizumab) taltz.com 800-545-5979 Hematology Aranesp (darbepoetin alfa) aranesp.com 805-447-1000 chemocare.com Granix (filgrastim) granixrx.com 888-4-TEVARX hematology.org Jadenu (deferasirox) jadenu.com 888-282-7630 Neulasta (pegfilgrastim) neulasta.com 800-77-AMGEN Neupogen (filgrastim) neupogen.com 800-77-AMGEN Nivestym (filgrastim) nivestym.com 800-879-3477 Zarxio (filgrastim) zarxio.com 800-525-8747 Zytiga (abiraterone) zytiga.com 800-JANSSEN Hepatitis B Baraclude (entecavir) baraclude.com 800-321-1335 cdc.gov Viread (tenofovir disoproxil viread.com 800-GILEAD-5 hepb.org fumarate) -

Risk Assessment and Risk Mitigation Review(S)
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 213498Orig1s000 RISK ASSESSMENT and RISK MITIGATION REVIEW(S) Division of Risk Management (DRISK) Office of Medication Error Prevention and Risk Management (OMEPRM) Office of Surveillance and Epidemiology (OSE) Center for Drug Evaluation and Research (CDER) Application Type NOA Application Number 213498 POU FA Goal Date March 18, 2021 OSE RCM # 2020-534 Reviewer Name(s) Carlisha Gentles, PharmD, BCPS Team Leader Jacqueline Sheppard, PharmD Deputy Director Doris Auth, PharmD Review Completion Date March 2, 2021 Subject Eva luation of Need for a REMS Established Name Ponesimod Trade Name Ponvory Name of Applicant Janssen Pharmaceuticals Inc Therapeutic Class Sphingosine 1-phosphate (SlP) receptor modulator Formulation(s) Film-coated tablets: 2 mg, 3 mg, 4 mg, 5 mg, 6 mg, 7 mg, 8 mg, 9 mg, 10 mg, 20 mg Dosing Regimen Treatment init iation with a 14-day titration (see table), followed by a maintenance regimen of 20 mg orally once daily Treatment init iation titration: Titration Day Daily Dose Day 1 a nd 2 2mg Day 3 a nd 4 3mg Day 5 a nd 6 4mg Day7 Smg Dav8 6mg Day9 7mg Day10 8mg Day ll 9mg Day 12, 13, and 14 10mg 1 Reference ID 4755518 Table of Contents EXECUTIVE SUMMARY ......................................................................................................................................................... 3 1 Introduction .................................................................................................................................................................... -

COMPARISON of the WHO ATC CLASSIFICATION & Ephmra/Intellus Worldwide ANATOMICAL CLASSIFICATION
COMPARISON OF THE WHO ATC CLASSIFICATION & EphMRA/Intellus Worldwide ANATOMICAL CLASSIFICATION: VERSION June 2019 2 Comparison of the WHO ATC Classification and EphMRA / Intellus Worldwide Anatomical Classification The following booklet is designed to improve the understanding of the two classification systems. The development of the two systems had previously taken place separately. EphMRA and WHO are now working together to ensure that there is a convergence of the 2 systems rather than a divergence. In order to better understand the two classification systems, we should pay attention to the way in which substances/products are classified. WHO mainly classifies substances according to the therapeutic or pharmaceutical aspects and in one class only (particular formulations or strengths can be given separate codes, e.g. clonidine in C02A as antihypertensive agent, N02C as anti-migraine product and S01E as ophthalmic product). EphMRA classifies products, mainly according to their indications and use. Therefore, it is possible to find the same compound in several classes, depending on the product, e.g., NAPROXEN tablets can be classified in M1A (antirheumatic), N2B (analgesic) and G2C if indicated for gynaecological conditions only. The purposes of classification are also different: The main purpose of the WHO classification is for international drug utilisation research and for adverse drug reaction monitoring. This classification is recommended by the WHO for use in international drug utilisation research. The EphMRA/Intellus Worldwide classification has a primary objective to satisfy the marketing needs of the pharmaceutical companies. Therefore, a direct comparison is sometimes difficult due to the different nature and purpose of the two systems. -

The Real-World Patient Experience of Fingolimod and Dimethyl Fumarate
Wicks et al. BMC Res Notes (2016) 9:434 DOI 10.1186/s13104-016-2243-8 BMC Research Notes RESEARCH ARTICLE Open Access The real‑world patient experience of fingolimod and dimethyl fumarate for multiple sclerosis Paul Wicks1* , Lawrence Rasouliyan2, Bo Katic1, Beenish Nafees3, Emuella Flood3 and Rahul Sasané4 Abstract Background: Oral disease-modifying therapies offer equivalent or superior efficacy and greater convenience versus injectable options. Objectives: To compare patient-reported experiences of fingolimod and dimethyl fumarate. Methods: Adult relapsing-remitting multiple sclerosis patients treated with fingolimod or dimethyl fumarate were recruited from an online patient community and completed an online survey about treatment side effects, discon- tinuation, and satisfaction. Results: 281 patients in four groups completed the survey: currently receiving fingolimod (CF, N 61), currently receiving dimethyl fumarate (CDMF, N 129), discontinued fingolimod (DF, N 32) and discontinued= dimethyl fuma- rate (DDMF, N 59). Reasons for treatment= switch were to take oral treatment =(CF: 63.3 %, CDMF: 61.8 %), side effects of prior medication= (CF: 67.3 %, CDMF: 44.1 %) and lack of effectiveness of prior medication (CF: 38.8 %, CDMF: 31.4 %). Main reasons for discontinuation were side effects (DF: 46.9 %, DDMF: 67.8 %) and lack of effectiveness (DF: 25.0 %, DDMF: 15.3 %). CDMF patients had an increased risk of abdominal pain, flushing, diarrhea, and nausea. Treatment satisfaction was highest among CF patients followed by CDMF, DF, and then DDMF patients. Conclusions: Discontinuation was driven by experience of side effects. Patients currently taking dimethyl fumarate were more likely to experience a side effect versus patients currently taking fingolimod. -

Fingolimod (Gilenya)
Fingolimod (Gilenya) This factsheet is about fingolimod, a Whether you’ll be offered this or any other DMT disease modifying therapy (DMT) for depends on whether you qualify for it based relapsing multiple sclerosis (MS). on guidelines used by your neurologist. These come from NICE and the Association of British At the end of this factsheet you’ll find out where Neurologists and are based on a drug’s Europe- you can get more information on this drug, other wide licence. drugs for MS and the benefits of early treatment. In England there are also rules from NHS England about who can have the different DMTs and when. This factsheet doesn’t cover everything about Scotland, Wales and Northern Ireland also have this drug and shouldn’t be used in place of their own guidelines for many DMTs. advice from your MS specialist team. For more information speak to them and read the online information from the drug’s makers (see the Whether you can have a drug also depends on section More information and support). if the NHS where you live will pay for it. NHS England guidelines on this tend to follow what What is fingolimod? NICE says. Fingolimod is a drug that was first given a licence These people can have fingolimod: to be used against relapsing MS in the UK in 2011. In England and Northern Ireland: In 2012 the National Institute for Health and Care Excellence (NICE) gave the go ahead for it to be • People with ‘highly active relapsing remitting used on the NHS. -

Multiple Sclerosis
© Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 | Fax 503-947-2596 Drug Class Update with New Drug Evaluations: Multiple Sclerosis Date of Review: June 2021 Date of Last Review: August 2020 Dates of Literature Search: 02/03/2020 – 03/01/2021 Generic Name: Ofatumumab Ponesimod Brand Name (Manufacturer): Kesimpta (Novartis) Ponvory (Janssen) Dossier Received: yes, for ofatumumab Current Status of PDL Class: See Appendix 1. Purpose for Class Update: Evidence for the comparative effectiveness of disease modifying drugs (DMDs) for multiple sclerosis (MS) was last reviewed by the Oregon Pharmacy & Therapeutic Committee (P&T) in August 2020 as summarized in a Drug Effectiveness Review Project (DERP) report. This review examines new comparative evidence of DMDs for MS published since 2020 and summarizes the evidence for 2 new DMDs, ofatumumab and ponesimod, approved to treat relapsing forms of MS. Research Questions: 1. What is the comparative effectiveness and efficacy of DMDs for MS? 2. Do DMDs for MS differ in harms? 3. Are there subgroups of patients with MS based on demographics (age, racial or ethnic groups, and gender), socioeconomic status, concomitant medications, severity of disease, or co-morbidities for which one DMD is more effective or associated with fewer adverse events? 4. What is the evidence for efficacy and safety for use of ofatumumab in relapsing MS? 5. What is the evidence for efficacy and safety of ponesimod in relapsing MS ? Author: Deanna Moretz, PharmD, BCPS Conclusions: Multiple Sclerosis Disease-Modifying Drugs No new evidence of comparative efficacy or effectiveness of DMDs approved to treat MS has been published since the last MS class review.