Hydromorphone Hydrochloride) C-II
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hydromorphone
Hydromorphone WHAT IS HYDROMORPHONE? sedation, and reduced anxiety. It may also cause Hydromorphone belongs to a class of drugs mental clouding, changes in mood, nervousness, called “opioids,” which includes morphine. It and restlessness. It works centrally (in the has an analgesic potency of two to eight times brain) to reduce pain and suppress cough. greater than that of morphine and has a rapid Hydromorphone use is associated with both onset of action. physiological and psychological dependence. WHAT IS ITS ORIGIN? What is its effect on the body? Hydromorphone is legally manufactured and Hydromorphone may cause: distributed in the United States. However, • Constipation, pupillary constriction, urinary retention, users can obtain hydromorphone from nausea, vomiting, respiratory depression, dizziness, forged prescriptions, “doctor-shopping,” impaired coordination, loss of appetite, rash, slow or theft from pharmacies, and from friends and rapid heartbeat, and changes in blood pressure acquaintances. What are its overdose effects? What are the street names? Acute overdose of hydromorphone can produce: Common street names include: Severe respiratory depression, drowsiness • D, Dillies, Dust, Footballs, Juice, and Smack progressing to stupor or coma, lack of skeletal muscle tone, cold and clammy skin, constricted What does it look like? pupils, and reduction in blood pressure and heart Hydromorphone comes in: rate • Tablets, capsules, oral solutions, and injectable Severe overdose may result in death due to formulations respiratory depression. How is it abused? Which drugs cause similar effects? Users may abuse hydromorphone tablets by Drugs that have similar effects include: ingesting them. Injectable solutions, as well as • Heroin, morphine, hydrocodone, fentanyl, and tablets that have been crushed and dissolved oxycodone in a solution may be injected as a substitute for heroin. -

(Oxycontin®) and Oxymorphone (Opana® ER) Reference Number: ERX.NSST.17 Effective Date: 06/15 Revision Log Last Review Date: 09/16
Clinical Policy: extended-release oxycodone (OxyContin®) and oxymorphone (Opana® ER) Reference Number: ERX.NSST.17 Effective Date: 06/15 Revision Log Last Review Date: 09/16 Clinical policies are intended to be reflective of current scientific research and clinical thinking. This policy is current at the time of approval, may be updated and therefore is subject to change. This Clinical Policy is not intended to dictate to providers how to practice medicine, nor does it constitute a contract or guarantee regarding payment or results. Providers are expected to exercise professional medical judgment in providing the most appropriate care, and are solely responsible for the medical advice and treatment of members. This policy is the property of Envolve Pharmacy Solutions. Unauthorized copying, use, and distribution of this Policy or any information contained herein is strictly prohibited. By accessing this policy, you agree to be bound by the foregoing terms and conditions, in addition to the Site Use Agreement for Health Plans associated with Envolve Pharmacy Solutions. Description The intent of the criteria is to ensure that patients follow selection elements established by Envolve Pharmacy Solutions for the use of extended-release oxycodone (OxyContin®) and oxymorphone (Opana® ER). Policy/Criteria It is the policy of health plans affiliated with Envolve Pharmacy Solutions® that extended-release oxycodone (OxyContin®) and oxymorphone (Opana® ER) are medically necessary for members meeting the following criteria: Initial Approval Criteria (must meet all): A. Failure of an immediate-release (short-acting) narcotic analgesic; B. Failure of fentanyl patch and morphine extended-release tablets/capsules in the past 6 months, unless intolerant or contraindicated; C. -

Veterinary Anesthetic and Analgesic Formulary 3Rd Edition, Version G
Veterinary Anesthetic and Analgesic Formulary 3rd Edition, Version G I. Introduction and Use of the UC‐Denver Veterinary Formulary II. Anesthetic and Analgesic Considerations III. Species Specific Veterinary Formulary 1. Mouse 2. Rat 3. Neonatal Rodent 4. Guinea Pig 5. Chinchilla 6. Gerbil 7. Rabbit 8. Dog 9. Pig 10. Sheep 11. Non‐Pharmaceutical Grade Anesthetics IV. References I. Introduction and Use of the UC‐Denver Formulary Basic Definitions: Anesthesia: central nervous system depression that provides amnesia, unconsciousness and immobility in response to a painful stimulation. Drugs that produce anesthesia may or may not provide analgesia (1, 2). Analgesia: The absence of pain in response to stimulation that would normally be painful. An analgesic drug can provide analgesia by acting at the level of the central nervous system or at the site of inflammation to diminish or block pain signals (1, 2). Sedation: A state of mental calmness, decreased response to environmental stimuli, and muscle relaxation. This state is characterized by suppression of spontaneous movement with maintenance of spinal reflexes (1). Animal anesthesia and analgesia are crucial components of an animal use protocol. This document is provided to aid in the design of an anesthetic and analgesic plan to prevent animal pain whenever possible. However, this document should not be perceived to replace consultation with the university’s veterinary staff. As required by law, the veterinary staff should be consulted to assist in the planning of procedures where anesthetics and analgesics will be used to avoid or minimize discomfort, distress and pain in animals (3, 4). Prior to administration, all use of anesthetics and analgesic are to be approved by the Institutional Animal Care and Use Committee (IACUC). -

Heterodimerization of Μ and Δ Opioid Receptors: a Role in Opiate Synergy
The Journal of Neuroscience, 2000, Vol. 20 RC110 1of5 Heterodimerization of and ␦ Opioid Receptors: A Role in Opiate Synergy I. Gomes, B. A. Jordan, A. Gupta, N. Trapaidze, V. Nagy, and L. A. Devi Departments of Pharmacology and Anesthesiology, New York University School of Medicine, New York, New York 10016 Opiate analgesics are widely used in the treatment of severe -selective ligands results in a significant increase in the bind- pain. Because of their importance in therapy, different strate- ing of a ␦ receptor agonist. This robust increase is also seen in gies have been considered for making opiates more effective SKNSH cells that endogenously express both and ␦ recep- while curbing their liability to be abused. Although most opiates tors. Furthermore, we find that a ␦ receptor antagonist en- exert their analgesic effects primarily via opioid receptors, a hances both the potency and efficacy of the receptor signal- number of studies have shown that ␦ receptor-selective drugs ing; likewise a antagonist enhances the potency and efficacy can enhance their potency. The molecular basis for these find- of the ␦ receptor signaling. A combination of agonists ( and ␦ ings has not been elucidated previously. In the present study, receptor selective) also synergistically binds and potentiates we examined whether heterodimerization of and ␦ receptors signaling by activating the –␦ heterodimer. Taken together, could account for the cross-modulation previously observed these studies show that heterodimers exhibit distinct ligand between these two receptors. We find that co-expression of binding and signaling characteristics. These findings have im- and ␦ receptors in heterologous cells followed by selective portant clinical ramifications and may provide new foundations immunoprecipitation results in the isolation of –␦ het- for more effective therapies. -

1 Impact of Opioid Agonists on Mental Health in Substitution
Impact of opioid agonists on mental health in substitution treatment for opioid use disorder: A systematic review and Bayesian network meta-analysis of randomized clinical trials Supplementary Table 1_ Specific search strategy for each database The following general combination of search terms, Boolean operators, and search fields were used where “*” means that any extension of that word would be considered: Title field [opium OR opiate* OR opioid OR heroin OR medication assisted OR substitution treatment OR maintenance treatment OR methadone OR levomethadone OR buprenorphine OR suboxone OR (morphine AND slow) OR diamorphine OR diacetylmorphine OR dihydrocodeine OR hydromorphone OR opium tincture OR tincture of opium OR methadol OR methadyl OR levomethadyl] AND Title/Abstract field [trial* OR random* OR placebo] AND All fields [depress* OR anxiety OR mental] Wherever this exact combination was not possible, a more inclusive version of the search strategy was considered. Database Search Strategy Ovid for EBM Reviews - Cochrane Central Register of (opium or opiate$ or opioid or heroin or medication Controlled Trials August 2018; Embase 1974 to assisted or substitution treatment or maintenance September 07, 2018; MEDLINE(R) and Epub Ahead treatment or methadone or levomethadone or of Print, In-Process & Other Non-Indexed Citations buprenorphine or suboxone or (morphine and slow) or and Daily 1946 to September 07, 2018 diamorphine or diacetylmorphine or dihydrocodeine or hydromorphone or opium tincture or tincture of opium or methadol or methadyl -

Hydromorphone and Morphine Are Not the Same Drug!
HYDROmorphone and Morphine are not the same drug! HYDROmorphone and morphine are deemed high risk/high alert medications. An Independent Double Check (IDC) must be done to avoid errors when administering all high risk drugs. Please refer to Clinical Practice Standard 1-20-6-3-260. HYDROmorphone and morphine need to knows: HYDROmorphone is 5 times more potent than morphine! (e.g., 1 mg of HYDROmorphone PO is equivalent to 5 mg of morphine PO) When administered via the subcutaneous route, these drugs are twice as strong! Therefore when converting between oral and subcutaneous routes half the dose. (10 mg of oral morphine = 5 mg of subcutaneous morphine) HYDROmorphone and morphine: The name game. Both drugs are used in the management of moderate to severe pain. HYDROmorphone and morphine come in different brand names and formulations. HYDROmorphone = Dilaudid (Injectable), Jurnista (SR), HYDROmorph Contin (SR) Morphine = Morphine Sulphate (Injectable), MS-IR, M.O.S. (Liquid/Tablet/SR), M-Eslon (SR), Statex (Liquid/Tablet/Supp.), Doloral (Liquid), MS Contin (SR), Kadian (SR) (SR=Slow Release CAP) *As per NH, use the generic names (NOT THE BRAND NAME), of these drugs in your documentation. Common side effects: 3 ‘B’s of opioid prescribing: Common: Nausea, Vomiting, Constipation, Sedation, Syncope, Xerostomia (Dry Mouth), If your client is on an opioid: Delirium, Restlessness, Urinary Retention. Do they have a Breakthrough? Less Common: Respiratory Depression, Do they have something for Barfing? Opioid-Induced Neurotoxicity, Myoclonus, Do they have something for their Bowels? Pruritis (itching). Opioid induced neurotoxicity: What does it look like? Opioid induced neurotoxicity is the result of a build up of toxic opioid metabolites in the blood stream. -

Modulation of Opioid Transport at the Blood-Brain Barrier by Altered ATP-Binding Cassette (ABC) Transporter Expression and Activity
pharmaceutics Review Modulation of Opioid Transport at the Blood-Brain Barrier by Altered ATP-Binding Cassette (ABC) Transporter Expression and Activity Junzhi Yang 1, Bianca G. Reilly 2, Thomas P. Davis 1,2 and Patrick T. Ronaldson 1,2,* 1 Department of Pharmacology and Toxicology, College of Pharmacy, University of Arizona, 1295 N. Martin St., P.O. Box 210207, Tucson, AZ 85721, USA; [email protected] (J.Y.); [email protected] (T.P.D.) 2 Department of Pharmacology, College of Medicine, University of Arizona, 1501 N. Campbell Ave, P.O. Box 245050, Tucson, AZ 85724-5050, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-520-626-2173 Received: 19 September 2018; Accepted: 16 October 2018; Published: 18 October 2018 Abstract: Opioids are highly effective analgesics that have a serious potential for adverse drug reactions and for development of addiction and tolerance. Since the use of opioids has escalated in recent years, it is increasingly important to understand biological mechanisms that can increase the probability of opioid-associated adverse events occurring in patient populations. This is emphasized by the current opioid epidemic in the United States where opioid analgesics are frequently abused and misused. It has been established that the effectiveness of opioids is maximized when these drugs readily access opioid receptors in the central nervous system (CNS). Indeed, opioid delivery to the brain is significantly influenced by the blood-brain barrier (BBB). In particular, ATP-binding cassette (ABC) transporters that are endogenously expressed at the BBB are critical determinants of CNS opioid penetration. In this review, we will discuss current knowledge on the transport of opioid analgesic drugs by ABC transporters at the BBB. -

Do You Know... Methadone
Do You Know... Street names: juice, meth (also used to refer to methamphetamines) What is it? Methadone belongs to the opioid family of Methadone drugs. It is used most commonly to treat addiction to other opioid drugs such as heroin, oxycodone (e.g., Percodan, Percocet), fentanyl (e.g., Duragesic, Sublimaze) and hydromorphone (e.g., Dilaudid). Methadone is a synthetic opioid, which means that it is made from chemicals in a lab. Methadone was developed in Germany during the Second World War and was first used to provide pain relief. Methadone maintenance treatment, which prevents opioid withdrawal and reduces or eliminates drug cravings, was first developed in the 1960s. For many years, Canadian reg- ulations around the prescription of methadone were so restrictive that few doctors offered the treatment. People who wanted methadone treatment often had to wait months or years. 1/4 © 2003, 2011, 2012 CAMH | www.camh.ca In the 1990s, the need to reduce the harm of drug use with medical care, improves the chances of having a was more clearly recognized, and changes were made healthy baby. There are no known long-term effects of to make it easier for doctors to provide methadone methadone on the baby. treatment. People who inject opioid drugs regularly, and who are Methadone maintenance is not a “cure”: it is a treatment. HIV- or hepatitis C–positive, are enrolled in methadone Through treatment, people who are addicted to opioids treatment to help protect their health. Methadone receive the medical and social support they need to treatment also helps to prevent these infections from stabilize and improve their lives. -

Hydromorphone Hydrochloride) CS-II WARNING: DILAUDID ORAL LIQUID and DILAUDID TABLETS CONTAIN HYDROMORPHONE, WHICH IS a POTENT SCHEDULE II CONTROLLED OPIOID AGONIST
DILAUDID® ORAL LIQUID and DILAUDID® TABLETS (hydromorphone hydrochloride) CS-II WARNING: DILAUDID ORAL LIQUID AND DILAUDID TABLETS CONTAIN HYDROMORPHONE, WHICH IS A POTENT SCHEDULE II CONTROLLED OPIOID AGONIST. SCHEDULE II OPIOID AGONISTS, INCLUDING MORPHINE, OXYMORPHONE, OXYCODONE, FENTANYL, AND METHADONE, HAVE THE HIGHEST POTENTIAL FOR ABUSE AND RISK OF PRODUCING RESPIRATORY DEPRESSION. ALCOHOL, OTHER OPIOIDS AND CENTRAL NERVOUS SYSTEM DEPRESSANTS (SEDATIVE-HYPNOTICS) POTENTIATE THE RESPIRATORY DEPRESSANT EFFECTS OF HYDROMORPHONE, INCREASING THE RISK OF RESPIRATORY DEPRESSION THAT MIGHT RESULT IN DEATH. DESCRIPTION Proprietary name: DILAUDID ORAL LIQUID Established name: hydromorphone hydrochloride Route of administration: ORAL (C38288) Active ingredients (moiety): hydromorphone hydrochloride (hydromorphone) # Strength Form Inactive ingredients 1 5 MILLIGRAM LIQUID purified water, methylparaben, propylparaben, sucrose, glycerin, sodium (C42953) metabisulfite Proprietary name: DILAUDID TABLETS Established name: hydromorphone hydrochloride Route of administration: ORAL (C38288) Active ingredients (moiety): hydromorphone hydrochloride (hydromorphone) # Strength Form Inactive ingredients 1 2 MILLIGRAM TABLET D&C red #30 Lake dye, D&C yellow #10 Lake dye, lactose, magnesium (C42998) stearate, sodium metabisulfite 2 4 MILLIGRAM TABLET D&C yellow #10 Lake dye, lactose, magnesium stearate, sodium metabisulfite (C42998) 3 8 MILLIGRAM TABLET lactose anhydrous, magnesium stearate, sodium metabisulfite (C42998) DILAUDID (hydromorphone hydrochloride), a hydrogenated ketone of morphine, is an opioid analgesic. The chemical name of DILAUDID (hydromorphone hydrochloride) is 4,5α-epoxy-3- hydroxy-17-methylmorphinan-6-one hydrochloride. The structural formula is: M.W. 321.8 Each 5 mL (1 teaspoon) of DILAUDID ORAL LIQUID contains 5 mg of hydromorphone hydrochloride. In addition, other ingredients include purified water, methylparaben, propylparaben, sucrose, and glycerin. DILAUDID ORAL LIQUID may contain traces of sodium metabisulfite. -
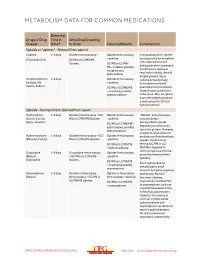
Metabolism Data for Common Medications
METABOLISM DATA FOR COMMON MEDICATIONS Detection Drugs or Drug Time in Urine Drug Screening Classes Urine* to Order Expected Results Consideration Opioids or “opiates” – Natural (from opium) Codeine 1–3 days Opiates Immunoassay + Opiates Immunoassay Immunoassays for “opiates” are responsive for morphine (Tylenol #2/3/4) GC/MS or LC/MS/MS – positive and codeine but do not Opiates GC/MS or LC/MS/ distinguish which is present. MS – codeine, possibly Confirmatory testing is morphine and required to reliably identify hydrocodone drug(s) present. Since Morphine (Avinza, 1–3 days Opiates Immunoassay codeine is metabolized Embeda, MS – positive to morphine and small Contin, Kadian) GC/MS or LC/MS/MS quantities to hydrocodone, – morphine, possibly these drugs may be found hydromorphone in the urine. Also, morphine may metabolize to produce a small amount (<10%) of hydromorphone. Opioids – Semisynthetic (derived from opium Hydrocodone 1–3 days Opiates Immunoassay + GC/ Opiates Immunoassay “Opiates” immunoassays (Lorcet, Lortab, MS or LC/MS/MS Opiates – positive may also detect Norco, Vicodin) semisynthetic opioids GC/MS or LC/MS/MS – depending on their cross- hydrocodone, possibly reactivity pattern. However, hydromorphone a negative result does not Hydromorphone 1–3 days Opiates Immunoassay + GC/ Opiates Immunoassay exclude use of semisynthetic (Dilaudid, Exalgo) MS or LC/MS/MS Opiates –positive opioids. Confirmatory GC/MS or LC/MS/MS testing (GC/MS or LC/ – hydromorphone MS/MS) is required to verify compliance with the Oxycodone 1–3 days Oxycodone Immunoassay Opiates Immunoassay prescribed semisynthetic (Roxicet, + GC/MS or LC/MS/MS –positive opioid(s). OxyContin) Opiates GC/MS or LC/MS/MS Since hydrocodone is – oxycodone possibly metabolized in small oxymorphone amounts to hydromorphone, Oxymorphone 1–3 days Opiates or Oxycodone Opiates or Oxycodone both may be found in (Opana) Immunoassay + GC/MS or Immunoassay – positive the urine. -

Summary of Synthetic Opioid Testing Within the Department of Defense
SUMMARY OF SYNTHETIC OPIOID TESTING WITHIN THE DEPARTMENT OF DEFENSE Tom Martin, Ph.D. Liaison to Director, DoD Drug Testing and Program Policy PERSONNEL AND READINESS PERSONNEL AND READINESS Drug Demand Reduction Program Mission and Scope Mission: Enable operational readiness, safety, and security of the Total Force by deterring illicit and prescription drug abuse through robust and dynamic drug testing; emerging drug threat surveillance; prevention, education, and outreach efforts; and development of new testing procedures. Scope: All DoD components and DoD civilians in testing designated positions (TDPs) Policies: DODI 1010.01 “Military Personnel Drug Abuse Testing Program (MPDATP)” DODI 1010.09 “DoD Civilian Employee Drug-Free Workplace Program” DODI 1010.16 “Technical Procedures for the Military Personnel Drug Abuse Testing Program (MPDATP)” PERSONNEL AND READINESS DDRP Driving Factors • Drug abuse in the general U.S. 18-25 year old male group is estimated to be 17-20%– the population from which the Service recruits their enlisted personnel • Before DoD instituted drug testing among Service personnel, drug use was a significant recurring problem – Vietnam (estimated over 5% of returning service members addicted to heroin) – 1981 CVN Nimitz aviation mishap – 14 killed, 48 injured, 7 aircraft destroyed, 11 aircraft damaged, $150M in damages, six deceased with detectable levels of marijuana • Notable increase in abuse/misuse of prescription pain medications • Personnel abusing illicit drugs or prescription medications are a safety hazard -

Summary Report of Benefit-Risk Assessment NALDEBAIN
Summary Report of Benefit-Risk Assessment NALDEBAIN EXTENDED RELEASE INJECTION 75 MG/ML NEW DRUG APPLICATION Active Ingredient(s) Dinalbuphine sebacate Product Registrant Intega Pte Ltd Product Registration Number SIN16058P Application Route Abridged evaluation Date of Approval 15 December 2020 Copyright © 2021 Health Sciences Authority of Singapore You may download, view, print and reproduce this summary report without modifications for non-commercial purposes only. Except as otherwise provided, the contents of this summary report may not be reproduced, republished, uploaded, posted, transmitted or otherwise distributed in any way without the prior written permission of the Health Sciences Authority. This summary report and its contents are made available on an “as is” basis and the Health Sciences Authority makes no warranty of any kind, whether express or implied. The information in the summary report is provided for general information only and the contents of the summary report do not constitute medical or other professional advice. If medical or other professional advice is required, services of a competent professional should be sought. Table of Contents A INTRODUCTION ............................................................................................................. 3 B ASSESSMENT OF PRODUCT QUALITY ....................................................................... 3 C ASSESSMENT OF CLINICAL EFFICACY ...................................................................... 4 D ASSESSMENT OF CLINICAL SAFETY .........................................................................