Edmonton Zone IHC Laboratory Breast Biomarker Requests
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

07/08 Annual Review Together for a Cancer- Free Future
07/08 Annual Review Together for a cancer- free future 07/08 Annual Review Together for a Cancer Free Future 01 Contents Message from Message from the Board Chair and CEO 01 the Board Chair Articles It computes: mining data for promising drugs 12 and CEO Walking beyond grief sends a message of hope 13 Sowing the seeds of a legacy 14 The first part of the team is our donors. Non-smoker tackles lung cancer head on 15 It has been our privilege Alberta Cancer Foundation donors make personal Stepping up to challenge of breast cancer 16 contributions, plan legacy gifts, purchase lottery again this year to connect the tickets, sponsor participants in our walks and put The art of healing 17 their own ingenuity to work as volunteers, planning Employees power powers innovation 18 work of two important groups and executing more than 300 fundraising events Special meaning to this year’s golf classic 19 each year. World’s longest hockey game 20 Their support is a tribute to the thousands of that form one team intent on Albertans diagnosed with cancer this year. It’s a Coping with the cost of cancer 21 message of hope for the nearly 16,000 expected Bridging the gap between research and practice 22 building a cancer-free future to be diagnosed next year. And their gifts honour Face off against cancer 23 the memory of more than 5,000 Albertans who New lab space key to attracting talent 24 for individual Albertans and lose their battle with cancer each year. -

2009 Program
ISOQOL 16th Annual Conference of the International Society for Quality of Life Research Integrating HRQOL in Health Care Policy, Research, and Practice October 28-31, 2009 New Orleans, Louisiana, USA Sheraton New Orleans Final Program Table of Contents Table of Contents Schedule-at-a-Glance ......................................................................................................................... 3 Welcome ................................................................................................................................................ 4 Scientific Program Committee .......................................................................................................... 5 ISOQOL Leadership ............................................................................................................................ 6 About ISOQOL/General Information .........................................................................................7 - 8 Program Schedule, Wednesday...................................................................................................9 - 14 Program Schedule, Thursday ................................................................................................... 15 - 21 Program Schedule, Friday ......................................................................................................... 22 - 28 Program Schedule, Saturday .................................................................................................... 29 - 35 Posters ......................................................................................................................................... -

Legislative Assembly of Alberta
April 23, 2001 Alberta Hansard 103 Legislative Assembly of Alberta ated from Montgomery, Bowness, Greenwood village, and Valley Ridge, but they’re also viewed from Scenic Acres, Tuscany, Arbour Title: Monday, April 23, 2001 8:00 p.m. Lake, Ranchlands, Silver Springs, Hawkwood, Varsity, Country Date: 01/04/23 Hills, Patterson Heights, Strathcona, and Artists View, and by all travelers entering or leaving Calgary by the main western access on [Mr. Shariff in the chair] the Trans-Canada highway. For all these tens of thousands of people Paskapoo Slopes is like head: Consideration of Her Honour a park in the sky enriching their everyday life, but Paskapoo Slopes the Lieutenant Governor’s Speech is also an accessible wilderness park right within the city. It’s a Mrs. Tarchuk moved that an humble address be presented to Her natural area that includes ravines, gullies, streams, springs, glades of Honour the Honourable the Lieutenant Governor as follows. aspen, balsam poplar, dogwood, and riverine tall shrub. The slopes To Her Honour the Honourable Lois E. Hole, CM, Lieutenant are also home to deer, small mammals, coyotes, and a large variety Governor of the province of Alberta: of migratory and breeding birds. Not only is it a home for the We, Her Majesty’s most dutiful and loyal subjects, the Legislative animals, but it’s also a key wildlife corridor within the city. Assembly, now assembled, beg leave to thank you, Your Honour, for Mr. Speaker, most of us members here are familiar with Head- the gracious speech Your Honour has been pleased to address to us Smashed-In Buffalo Jump, which is UNESCO’s world heritage site. -

Scientific Advisory Committee on Oncology Therapies (SAC-OT)
Scientific Advisory Committee on Oncology Therapies (SAC-OT) Membership List Core Members Alexander H.G. Paterson, MD, FRCP, FACP, MBChB (Chair) Medical Oncologist, Department of Medical Oncology, Tom Baker Cancer Centre Professor, Department of Medicine and Oncology, University of Calgary Calgary, Alberta Biography: Alexander Paterson graduated Medicine from Edinburgh University, United Kingdom in 1977 and subsequently trained at St. Bartholomew’s Hospital and Royal Marsden, London, England. He is a Fellow of the Royal College of Physicians and has been a Medical Oncologist at the Tom Baker Cancer Centre in Calgary since 1990. He is also Professor in the Departments of Medicine and Oncology at the University of Calgary, Alberta since 1995. He is a member of the Alberta Out-of-Province/Country Health Services Appeal Panel and the Board of Directors of the National Surgical Adjuvant Breast and Bowel Project (NSAPB). He is also Chair of the NSABP Protocol B-34 and the Alberta Breast Cancer Programme. He has published over 100 articles, authored over 15 book chapters, is the Editor of Fundamental Problems in Breast Cancer (I and II) and contributes a regular column to Alberta Doctors’ Digest. He has given over 200 invited lectures. Rick Abbott, BScPharm, RPEBC Pharmacy Manager, Provincial Systemic Therapy, Dr. H. Bliss Murphy Cancer Centre, Eastern Health St. John’s, Newfoundland and Labrador Biography: Rick Abbott graduated from the School of Pharmacy in 1990 at the Memorial University of Newfoundland, St. John’s, Newfoundland and Labrador. He has been the Pharmacy Manager of the Provincial Systemic Therapy since 2002 and is actively involved in the Pharmacy Profession. -

Cross Cancer Institute Public Parking
Cross Cancer Institute Public Parking New parking equipment has been installed to make paying for parking more convenient. New equipment is conveniently located at the locations marked above in yellow. Buy a permit by entering your license plate before paying by coin or credit card (no need to return to your vehicle to display permit). Weekly permits are not available from Pay by Plate equipment (please see next page for weekly permit purchases). Staying longer than expected? Before your time expires, add time to your permit without returning to your vehicle. Just enter your plate number at any pay station and follow prompts. Have a hard time remembering your license plate number? Take a picture with your smartphone. P1 Main Lot (West) P2 Main Lot (South) Gravel Lot (East) The Southfield Underground Parkade at the Cross Cancer Institute is operated by the University of Alberta, not by Alberta Health Services. For information on the Southfield Parkade please call (780) 492‐7275. See next page for more information. Valid until March 31, 2018 www.albertahealthservices.ca/parking Rates subject to change Cross Cancer Parking Office (located at Kaye Edmonton Clinic) Location: Room 1P019, Kaye Edmonton Clinic Business Hours: 8:00 a.m. – 4:00 p.m. Monday to Friday (closed on holidays) Phone Number: (780) 407‐5142 Email: [email protected] Public Parking Rates Rates apply 24 hours per day, and are in effect for all public parkers, including those with provincially issued placards for persons with disabilities. Public parking is GST exempt. Pay by Plate machines accept Canadian coins or credit card (Visa, Mastercard, American Express). -
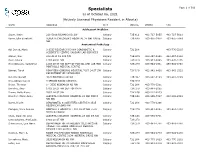
Specialists Page 1 of 509 As of October 06, 2021 (Actively Licensed Physicians Resident in Alberta)
Specialists Page 1 of 509 as of October 06, 2021 (Actively Licensed Physicians Resident in Alberta) NAME ADDRESS CITY POSTAL PHONE FAX Adolescent Medicine Soper, Katie 220-5010 RICHARD RD SW Calgary T3E 6L1 403-727-5055 403-727-5011 Vyver, Ellie Elizabeth ALBERTA CHILDREN'S HOSPITAL 28 OKI DRIVE Calgary T3B 6A8 403-955-2978 403-955-7649 NW Anatomical Pathology Abi Daoud, Marie 9-3535 RESEARCH RD NW DIAGNOSTIC & Calgary T2L 2K8 403-770-3295 SCIENTIFIC CENTRE CALGARY LAB SERVICES Alanen, Ken 242-4411 16 AVE NW Calgary T3B 0M3 403-457-1900 403-457-1904 Auer, Iwona 1403 29 ST NW Calgary T2N 2T9 403-944-8225 403-270-4135 Benediktsson, Hallgrimur 1403 29 ST NW DEPT OF PATHOL AND LAB MED Calgary T2N 2T9 403-944-1981 493-944-4748 FOOTHILLS MEDICAL CENTRE Bismar, Tarek ROKYVIEW GENERAL HOSPITAL 7007 14 ST SW Calgary T2V 1P9 403-943-8430 403-943-3333 DEPARTMENT OF PATHOLOGY Bol, Eric Gerald 4070 BOWNESS RD NW Calgary T3B 3R7 403-297-8123 403-297-3429 Box, Adrian Harold 3 SPRING RIDGE ESTATES Calgary T3Z 3M8 Brenn, Thomas 9 - 3535 RESEARCH RD NW Calgary T2L 2K8 403-770-3201 Bromley, Amy 1403 29 ST NW DEPT OF PATH Calgary T2N 2T9 403-944-5055 Brown, Holly Alexis 7007 14 ST SW Calgary T2V 1P9 403-212-8223 Brundler, Marie-Anne ALBERTA CHILDREN HOSPITAL 28 OKI DRIVE Calgary T3B 6A8 403-955-7387 403-955-2321 NW NW Bures, Nicole DIAGNOSTIC & SCIENTIFIC CENTRE 9 3535 Calgary T2L 2K8 403-770-3206 RESEARCH ROAD NW Caragea, Mara Andrea FOOTHILLS HOSPITAL 1403 29 ST NW 7576 Calgary T2N 2T9 403-944-6685 403-944-4748 MCCAIG TOWER Chan, Elaine So Ling ALBERTA CHILDREN HOSPITAL 28 OKI DR NW Calgary T3B 6A8 403-955-7761 Cota Schwarz, Ana Lucia 1403 29 ST NW Calgary T2N 2T9 DiFrancesco, Lisa Marie DEPARTMENT OF PATHOLOGY (CLS) MCCAIG Calgary T2N 2T9 403-944-4756 403-944-4748 TOWER 7TH FLOOR FOOTHILLS MEDICAL CENTRE 1403 29TH ST NW Duggan, Maire A. -

Of Medicine and Dentistrynews Faculty Has Another Howard Hughes Scholar Page 3
VOLUME 8 • NUMBER 1 Faculty SEPTEMBER • 2 0 0 5 of Medicine and DentistryNews Faculty has another Howard Hughes Scholar Page 3 Uncovering another risk for type 2 diabetes Page 4 A practitioner of problem-based learning reflects on his career Page 7 Gold Medal 2005 The Canadian Council for the Advancement of Education UNIVERSITY OF ALBERTA Leading pediatric Preserving pancreatic islets for cardiologists join team Capital Health and the University of clinical transplantation Alberta have recruited two of Canada’s top pediatric cardiologists for the Diabetes Research Wellness Foundation donates $200,000 US Stollery Children’s Hospital. Lori West to support islet preservation and isolation techniques and Jeff Smallhorn, from Toronto’s Hospital for Sick Children, will move prominent American philan- associated with anoxia (severe their clinical and research activities to A thropic foundation has recently hypoxia) and cell death,” says Edmonton this month. awarded funding to a University Dr. Lakey. “Dr. West’s research and clinical prac- of Alberta professor to help solve He and his team will tice surrounding heart transplants in infants has revolutionized how these some of the problems associated use the support from the children are treated—not just here in with recovering and preserving Foundation to focus on three Canada but all around the world” says well-functioning islet cells used in areas: islet cell responses to Terry Klassen, regional clinical program the Edmonton Protocol. anoxia, hypoxia and hyperoxia director of Capital Health’s Child Health Program and chair of the University The Diabetes Research and with or without hypothermia; of Alberta’s Department of Pediatrics. -

Echoes in the Halls : an Unofficial History of the University of Alberta
An Unofficial History of the University of Alberta ASSOCIATION OF PROFESSORS EMERITI OF THE UNIVERSITY OF ALBERTA An Unofficial History of the University of Alberta ASSOCIATION OF PROFESSORS EMERITI OF THE UNIVERSITY OF ALBERTA □OA LES EDITIONS DUVAL The University of Alberta Press Published jointly by Duval House Publishing 18120 - 102 Avenue Edmonton, Alberta T5S 1S7 Telephone: (780) 488-1390 Fax: (780) 482-7213 e-mail: [email protected] website: www.duvalhouse.com University of Alberta Press □OA Ring House 2 Edmonton, Alberta T6G 2E2 Telephone: (780) 455-2200 Duval House Publishing and the University of Alberta Press gratefully Canada ac^now^e(^^e financial support of the Government of Canada through the Book Publishing Industry Development Program (BPIDP) for our publishing activities. © 1999 Association of Professors Emeriti of the University of Alberta All rights reserved. No part of this book may be reproduced or used in any form or by any means—graphic, electronic or mechanical—without prior written permission from the publishers. Printed in Canada. Canadian Cataloguing in Publication Data Main entry under title: Echoes in the halls ISBN 1-55220-074-4 1. University of Alberta-History-Anecdotes. I. Spencer, Mary, 1923- II. Dier, Kay, 1922- III. McIntosh, Gordon. LE3.A619E33 1999 378.7123’3 C99-911163-9 Cover photos: Front: Dr. Mark Arnfield adjusting the Argon-driven dye laser with the sixteen fibreoptic cables for interstitially applied PDT in Dunning R3327 rat prostate cancers (photo courtesy of Malcolm McPhee) Back: The trunk -
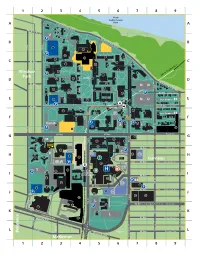
Northcampus Map 2009E 3.Ai
North Saskatchewan Saskatchewan Drive River 1 8 Edinboro Road 9 7 V 6 Saskatchewan Drive P 5 11 3 12 116 St 117 St P 25 32 35 13 21 92 Ave 34 37 26 30 33 22 48 29 36 23 Windsor P 55 52 LRT to City Centre > Park QUAD 40 42 51 54 44 49 Saskatchewan Drive 91 Ave 53 NN 71 62 63 45 50 69 73 P 90 Ave Brock Ave 65 72 66 64 67 East Campus 74 75 NU 89 61 P UNIVERSITY 89 Ave RESTRICTED ACCESS 89 Ave Legal Ave STATION 83 90 80 84 P 87 91 94 East Campus 88 82 88 97 110 St P 95 98 Varsity P 86 89 P 92 79 A Z 93 85 P 99 81 111 St 117 St 116 St 87 Ave 87 Ave Royal Ave 160 132 102 100 M 130 135 112 St P 114 St 133 101 131 103 134 86 Ave Oliver Ave 106 C P R P2 165 P5 107 104 105 108 ? 136 Garneau 85 Ave Griesbach Ave JBLW P 140 143 118 P3 T 116 110 111 HEALTH P SCIENCES 117 STATION 144 84 Ave Lumsden Ave 113 142 141 P 84 115 152 P4 170 111 St 114 83 Ave 112 P 83 Ave McDonald Ave 151 P1 P 150 172 174 124 L 112 St P7 114 St 82 Ave > to Campus Saint-Jean Whyte Ave 123 156 120 P 117 St P6 155 110 St University Ave L 111 St University Ave 125 81 Ave Anderson Ave South Ave 115a St 80 Ave St. -

Locum Radiation Oncologist – Cross Cancer Institute, Edmonton AB
Locum Radiation Oncologist – Cross Cancer Institute, Edmonton AB Posting #: 8007-0017LOC Specialty: Radiation Oncology Type: Locum – One Year Term City/Town: Edmonton Location: Cross Cancer Institute Date Posted: Closed Date: AHS Position: AHS is recruiting for this position AHS Sponsorship: This position is not eligible for AHS sponsorship. Responsibilities/Duties: Locum Radiation Oncologist / Fellowship, Cross Cancer Institute One Year Term The Department of Oncology, Division of Radiation Oncology at the Cross Cancer Institute (CCI), with Alberta Health Services is accepting applications for a 1.0FTE Combined Fellowship/Locum Radiation Oncology position. The institute is operated by CancerControl Alberta, Alberta Health Services and affiliated with the University of Alberta. This comprehensive cancer treatment and research facility serves patients in Edmonton, Northern Alberta, and Northwest Territories. Over 4,500 new consultations are seen each year by 23 Radiation Oncologists who work closely with on- site colleagues in Medical Oncology, Surgical Oncology, Gynecological Surgery, Oncological Imaging, Pathology, and Experimental Oncologists. Modern equipment including nine linear accelerators (capable IMRT, VMAT, IGRT, SRS, and SBRT), orthovoltage, MR-guided brachytherapy (HDR and PDR), and Gamma Knife Icon are available. A state-of-the art LINAC-MR is presently under construction. Two CT simulators, 1.5T and 3T MR, and PET/CT with an on-site cyclotron are available and a state-of-the-art PET/MR unit will open in May 2018. Qualifications: The successful applicant will have a M.D. or equivalent degree and Radiation Oncology specialist certification by the Royal College of Physicians and Surgeons of Canada. Candidates must be eligible to obtain licensure in Alberta without restriction and for medical staff appointment at Alberta Health Services. -
Inventory of Major Alberta Projects
Inventory of Major Alberta Projects March 2011 Alberta Finance and Enterprise 4th Floor, Commerce Place Edmonton, Alberta T5J 4L6 Inventory of Major Alberta Projects Introduction The Inventory of Major Alberta Projects is produced by Alberta Finance and Enterprise to assist firms in identifying potential supply opportunities. The Inventory lists construction projects in Alberta valued at $5.0 million or greater that are planned, underway, or have recently been completed. This edition also identifies projects that have been put on hold or deferred due to the economic downturn. Project data is obtained from public and private information sources. Where possible, this data has been verified with the project proponent/developer. However, users of the Inventory may wish to confirm project data with the proponent/developer. While efforts have been made to obtain the most recent information, it should be noted that projects are constantly being re-evaluated by industry. Although the Inventory attempts to be as comprehensive as possible, some information may not be available at the time of printing, or not published due to reasons of confidentiality. The Inventory can be accessed on the Internet at www.albertacanada.com/about-alberta/inventory-of- major-projects.html. New projects and (where possible) updated information on current projects is added to the Internet database on a monthly basis. The print edition of the report is produced quarterly. Projects added to the Inventory between January 1 and March 31, 2011 are noted in upper case in the Project Description column. If you have questions or require additional information about the Inventory, contact Alberta Finance and Enterprise by phone at (780) 427-6787, or by fax at (780) 422-0061. -

I Alberta Health
■I■ Alberta Health AHS Site List - Edmonton Zone (sorted alphabetically) ■ Services If you do not see your specific department listed, please select the closest active treatment hospital SITE NAME SITE CODE ADDRESS POSTAL CODE ACTIVE TREATMENT HOSPITALS Devon General Hospital (Active Treatment Hospital) 398 101 ERIE Street South/Sud, Devon, AB, Canada T9G1A6 Devon General Hospital (Auxiliary Hospital) 399 101 ERIE Street South/Sud, Devon, AB, Canada T9G1A6 Fort Saskatchewan Community Hospital (Active Treatment Hospital) 578 9401 86 Avenue, Fort Saskatchewan, AB, Canada T8L0C6 Fort Saskatchewan Community Hospital (Diagnostic Imaging) 579 DIAGNOSTIC IMAGING, 9401 86 Ave, Fort Saskatchewan, AB, Canada T8L0C6 Glenrose Rehabilitation Hospital (Active Treatment Hospital) 605 10230 111 Avenue North West, Edmonton, AB, Canada T5G0B7 Glenrose Rehabilitation Hospital (Diagnostic Imaging) 606 IMAGING DEPARTMENT, 10230 111 AVE NW, Edmonton, AB, Canada T5G0B7 Grey Nuns Community Hospital (Active Treatment Hospital) 1638 1100 YOUVILLE Drive North West, Edmonton, AB, Canada T6L5X8 RADIOLOGY & DIAGNOSTIC IMAGING, 1100 YOUVILLE DRIVE W, Edmonton, AB, Grey Nuns Community Hospital (Diagnostic Imaging) 1639 T6L5X8 Canada Leduc Community Hospital (Active Treatment Hospital) 824 4210 48 Street, Leduc, AB, Canada T9E5Z3 Leduc Community Hospital (Auxiliary Hospital) 825 4210 48 Street, Leduc, AB, Canada T9E5Z3 Leduc Community Hospital (Diagnostic Imaging) 826 IMAGING DEPARTMENT, 4210 48TH ST, Leduc, AB, Canada T9E5Z3 Misericordia Community Hospital (Active Treatment